Abstract
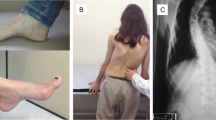
Friedreich’s ataxia (FDRA) is the most common inherited ataxia worldwide, caused by homozygous GAA expansions in the FXN gene. Patients usually have early onset ataxia, areflexia, Babinski sign, scoliosis and pes cavus, but at least 25 % of cases have atypical phenotypes. Disease begins after the age of 25 in occasional patients (late-onset Friedreich ataxia (LOFA)). Little is known about the frequency and clinical profile of LOFA patients. One hundred six patients with molecular confirmation of FDRA and followed in three Brazilian outpatient centers were enrolled. General demographics, GAA expansion size, age at onset, cardiac, endocrine, and skeletal manifestations were evaluated and compared between LOFA and classic FDRA (cFDRA) groups. We used Mann–Whitney and Fisher tests to compare means and proportions between groups; p values <0.05 were considered significant. LOFA accounted for 17 % (18/106) and cFDRA for 83 % (88/106) of the patients. There were 13 and 48 women in each group, respectively. LOFA patients were significantly older and had smaller GAA expansions. Clinically, LOFA group had a tendency toward lower frequency of diabetes/impaired glucose tolerance (5.8 vs. 17 %, p = 0.29) and cardiomyopathy (16.6 vs. 28.4 %, p = 0.38). Skeletal abnormalities were significantly less frequent in LOFA (scoliosis 22 vs. 61 %, p = 0.003, and pes cavus 22 vs.75 %, p < 0.001) as were spasticity and sustained reflexes, found in 22 % of LOFA patients but in none of the cFDRA patients (p = 0.001). LOFA accounts for 17 % of Brazilian FDRA patients evaluated herein. Clinically, orthopedic features and spasticity with retained reflexes are helpful tips to differentiate LOFA from cFDRA patients.
Similar content being viewed by others
References
Campuzano V, Montermini L, Molto MD, Pianese L, Cossée M, Cavalcanti F, et al. Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science. 1996;271:1423–7.
Bhidayasiri R, Perlman SL, Pulst SM, Geschwind DH. Late-onset Friedreich ataxia: phenotypic analysis, magnetic resonance imaging findings, and review of the literature. Arch Neurol. 2005;629:1865–9.
Abrahão A, Pedroso JL, Braga-Neto P, Bor-Seng-Shu E, Aguiar PC, Barsottini OG. Milestones in Friedreich ataxia: more than a century and still learning. Neurogenetics. 2015;16:151–60.
Friedreich N. Über degenerative Atrophie der spinalen Hinterstränge (On degenerative atrophy of the spinal dorsal columns), Virchows Arch. Pathol Anat Physiol Klin Med. 1863;26:391–419.
de Michele G, Filla A, Cavalcanti F, Di Maio L, Pianese L, Castaldo I, et al. Late onset Friedreich’s disease: clinical features and mapping of mutation to the FRDA locus. J Neurol Neurosurg Psychiatry. 1994;57:977–9.
Harding AE. Friedreich’s ataxia: a clinical and genetic study of 90 families with an analysis of early diagnostic criteria and intrafamilial clustering of clinical features. Brain. 1981;104:589–620.
Bayot A, Santos R, Camadro JM, Rustin P. Friedreich’s ataxia: the vicious circle hypothesis revisited. BMC Med. 2011;9:112.
Koeppen AH, Morral JA, McComb RD, Feustel PJ. The neuropathology of late-onset Friedreich’s ataxia. Cerebellum. 2011;10:96–103.
Reetz K, Dogan I, Costa AS, Dafotakis M, Fedosov K, Giunti P, et al. Biological and clinical characteristics of the European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS) cohort: a cross-sectional analysis of baseline data. Lancet Neurol. 2015;14:174–82.
Lecocq C, Charles P, Azulay JP, Meissner W, Rai M, N’Guyen K, et al. Delayed-onset Friedreich’s ataxia revisited. Mov Disord. 2015. doi:10.1002/mds.26382.
Durr A, Cossee M, Agid Y, Campuzano V, Mignard C, Penet C, et al. Clinical and genetic abnormalities in patients with Friedreich’s ataxia. N Engl J Med. 1996;335:1169–75.
Berciano J, Mateo I, De Pablos C, Polo JM, Combarros O. Friedreich ataxia with minimal GAA expansion presenting as adult-onset spastic ataxia. J Neurol Sci. 2002;194:75–82.
Moro A, Martinez ARM, Karuta CVS, Munhoz RP, Moscovich M, Germiniani FMB, et al. Pseudo-Dominant’ inheritance in Friedreich’s ataxia: clinical and genetic study of a Brazilian family. Movmnt Disords Clncl Pract. 2014;1:361–3.
Labelle L, Tohmé S, Duhaime M, Allard P. Natural history of scoliosis in Friedreich’s ataxia. J Bone Joint Surg Am. 1986;68:564–72.
da Silva CB, Chevis CF, D’Abreu A, Lopes-Cendes I, França Jr MC. Fatigue is frequent and multifactorial in Friedreich’s ataxia. Parkinsonism Relat Disord. 2013;19:766–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Martinez, A.R.M., Moro, A., Abrahao, A. et al. Nonneurological Involvement in Late-Onset Friedreich Ataxia (LOFA): Exploring the Phenotypes. Cerebellum 16, 253–256 (2017). https://doi.org/10.1007/s12311-015-0755-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-015-0755-8