Abstract
B and T lymphocyte attenuator (BTLA) is a lymphocyte inhibitory receptor mainly expressed on several subsets of T and B lymphocytes. In the current retrospective study, BTLA expression is examined for the first time on formalin fixed and paraffin embedded (FFPE) tissue sections of mature B cell non-Hodgkin's lymphomas (253 cases), using a newly developed monoclonal antibody. BTLA is highly expressed in chronic lymphocytic leukemia/small lymphocytic lymphoma. In contrast, BTLA expression is nearly absent in mantle cell lymphoma and is not expressed in any of the follicular lymphoma cases of all grades. In marginal zone lymphoma, diffuse large B cell lymphoma, and Burkitt's lymphoma, a minority of the cases exhibit expression of BTLA. This study demonstrates the value of BTLA expression as an additional tool in the panel of immunochemical markers used for the differential diagnosis of mature B cell lymphomas in FFPE tissue sections.
Similar content being viewed by others
Introduction
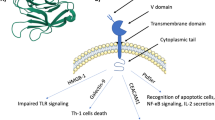
B and T lymphocyte attenuator (BTLA) (CD272) is a transmembrane glycoprotein, acting as a negative regulator of lymphocyte activation. BTLA is a member of the CD28-like immune regulators family that takes part in the coordination of immune responses through the T cell receptor (TCR) and the B cell receptor (BCR)-mediated signaling [1–4]. BTLA is expressed on anergic CD4+ T cells, subsets of activated T cells, resting and memory B cells, and has a lower expression on macrophages, dendritic cells, and natural killer cells. BTLA expression was not detected in non-immune cells [1, 5, 6]. Other members of the family, such as CD28, programmed cell death protein 1 (PD-1), inducible T cell co-stimulator, and cytotoxic T lymphocyte antigen 4 (CTLA-4), are also co-receptors that serve as either positive or negative effectors of the immune response. A primary co-stimulatory signal is delivered through the CD28-like receptors after engagement of B7-like family members, which are expressed on antigen-presenting cells and act as their ligands. BTLA exerts its inhibitory signals through interaction with herpesvirus entry mediator (HVEM), a member of the tumor necrosis factor (TNF) superfamily [7, 8]. The binding of BTLA to HVEM induces suppression of responses to self-antigens, thereby preventing autoimmune diseases and regulating responses to non-self molecules in adaptive immunity [3]. In the human reactive lymphoid tissue, BTLA is expressed by subsets of interfollicular T lymphocytes, mostly T regulator cells, by B cells in the mantle and marginal zones of the lymphoid follicle and by follicular T helper cells in the germinal center (GC) [9]. The expression of BTLA in human malignant lymphomas was previously described mainly in blood and bone marrow cells [10, 11] as well as in a limited number of cases of fresh lymphoid tissue frozen sections [9]. To the best of our knowledge, our study is the first performed on routine formalin fixed and paraffin embedded (FFPE) sections to analyze BTLA expression by neoplastic B cells in a large group of human mature B cell lymphomas.
Materials and methods
Tissue sampling
A total of 253 FFPE samples from biopsies of mature B cell lymphomas were studied. All the cases were classified according to the last World Health Organization classification (2008) [12] into chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), mantle cell lymphoma (MCL), follicular lymphoma (FL) of all grades, marginal zone lymphoma (MZL), diffuse large B cell lymphoma (DLBCL) and Burkitt's lymphoma (BL). One hundred thirteen samples were obtained from cases diagnosed between 2001 and 2010, based on morphologic examination and immunophenotyping, and stored at the Tissue Diagnosis and Cancer Research Department in Ha’Emek Medical Center, Afula, Israel. These biopsies were examined using conventional whole FFPE sections. The additional 140 cases were obtained from microarrays representative FFPE tissue core of lymphomas, kindly provided by Dr. Giovanna Roncador, head of the Monoclonal Antibodies Unit, Centro Nacional de Investigaciones Oncologicas (CNIO), Madrid, Spain (see Table 1 for summary of cases). A control group of FFPE non-neoplastic tissues from reactive lymphoid tissue (the tonsil and spleen) showing common features of hyperplasia (n = 16) was also tested. Control group biopsies were obtained from the Tissue Diagnosis and Cancer Research Department in Ha’Emek Medical Center, Afula, Israel. The study was approved by the institutional ethical committee.
Single immunohistochemical staining
Immunostaining of FFPE sections was performed in an automated stainer (Benchmark; Ventana Systems, Phoenix, AZ, USA). Slide-mounted 2 μm sections were subjected to Tris based buffer antigen retrieval followed by incubation with primary antibody for 32 min. Sections of the reactive tonsils were used as positive control samples, while negative control samples were prepared by using secondary antibody only. Single immunostaining was performed using the new anti-BTLA monoclonal antibody (clone FLO67B, kindly provided by Dr. Giovanna Roncador, CNIO, Madrid, Spain), PAX-5 (clone RM-9133, Thermo Scientific, Waltham, MA, USA), BCL-1 (clone RM-9104, Thermo Scientific, USA), BCL-6 (clone GI 191E/A8, kindly provided by Dr. Giovanna Roncador, CNIO, Madrid, Spain) and CD20 (clone M-0755, Dako, Glostrup, Denmark) antibodies. iVIEW DAB detection kit (760–091, Ventana Systems, USA) was used for reaction detection according to the manufacturer-recommended protocol. Hematoxylin counterstain was used for color development. Pictures of relevant sections were acquired with a DP70 camera on a BX51 microscope (both from Olympus Optical Ltd, Tokyo, Japan).
Double immunohistochemical (double IHC) staining
The selected positively and negatively stained biopsy sections were also studied using two-color immunostaining of BTLA and PAX-5, which was used for BTLA expression in B cells. BTLA/Bcl-6 immunostaining was used for identifying neoplastic B cells in FL and BL, and BTLA/Bcl-1 in MCL. The staining was performed on 2-μm FFPE sections in BenchMark automated stainer with serial use of iView DAB detection kit (760–091, Ventana Systems, USA) which stains brown with DAB reagent and ultraView universal alkaline phosphatase red detection kit (760–501, Ventana Systems, USA) which stains red with fast red. Hematoxylin counterstain was used for color development.
Double immunofluorescence (double IF) staining
Two μm sections of FFPE samples were mounted onto electrostatically charged microscope slides (VWR, Lutterworth, UK), dried at 60 °C for 1 h, dewaxed, and rehydrated using conventional methods. Heat-induced antigen retrieval was performed by placing slides in a reveal decloaker solution (RV1000MMRTU, Biocare Medical, Concord, CA, USA) preheated to 95 °C for 30 min followed by cooling for 10 min. The slides were transferred to DDW preheated to 95 °C for 10 min followed by additional 10 min of cooling and washed twice with DDW at room temperature. Blocking was performed using PBS with 10 % normal rabbit serum (HK114-5 K, Biogenex, Fremont, CA, USA) for 1 h at room temperature. For BTLA/CD20 double staining, primary antibodies were incubated overnight at 4 °C. The secondary antibodies used, goat anti-mouse Alexa Fluor 488 (A21121, Invitrogen, Grand Island, NY, USA) and goat anti-mouse IgG2a TRITC (1080–03, Southern Biotech, Birmingham, AL, USA), both anti-mouse-recognizing different isotopes, were incubated for 1 h. Primary and secondary antibodies were diluted in blocking solution. All washes carried out between antibody incubations were in PBS and repeated three times for 5 min each. Slides were counterstained for 1 min with DAPI (CS-2010-06, Biogenex, USA), mounted with fluorescence mounting medium (S3023, Dako, Denmark), and kept in the dark at 4 °C. The slides were viewed by motorized epifluorescent BX61 microscope (Olympus Optical Ltd, Japan), and pictures were taken using Spot RT-KE charge coupled device digital camera with monochrome fluorescence channel (Diagnostic Instrument Inc., Sterling Heights, MI, USA). The merged images were obtained using Spot Advanced v4.0.1 software (Diagnostic Instrument Inc., USA).
Immunohistochemistry and immunofluorescence staining analysis
Assessment of the BTLA expression in lymphoma B cells, as negative or positive, was conducted by two independent observers (P. T. and H. K. S). BTLA expression was evaluated as positive in sections in which more than 30 % of the neoplastic B cells were stained.
Statistics
Pearson's 2-tailed correlation χ2 test was used to calculate differences in frequencies of positive vs. negative BTLA expression in neoplastic B cells in mature B cell lymphomas.
Results
The pattern of BTLA expression in the reactive lymphoid tissue was studied on the tonsil and spleen sections, as a control group (Fig. 1). A strong expression of BTLA was localized in the marginal and mantle zone of the reactive follicles around the GC, and few interfollicular cells displayed a weak expression (Fig. 1a, b). The B cells in the GC did not express BTLA, as confirmed by double IHC with BTLA and Bcl-6 (Fig. 1b). We found, as well, a similar pattern of BTLA expression in sections of normal spleen (Fig. 1c). These findings corroborate the previous descriptions of BTLA expression in fresh frozen tissue of reactive lymph nodes [9].
BTLA expression in reactive tonsil lymphoid tissue and normal spleen. a BTLA is strongly expressed by the marginal and mantle zone lymphocytes, (single IHC; original magnification, ×400), while in the germinal center (g.c.) only few small cells consistent with T cells are stained (arrowheads). b The Bcl-6 positive GC B cells do not express BTLA, in contrast to the mantle and marginal zone Bcl-6 negative cells (double IHC of BTLA-red and Bcl-6-brown; original magnification, ×400). c In the normal spleen, the strongest BTLA expression is in the marginal and mantle zone of the white pulp B follicles, similar to the pattern seen in tonsil (single IHC; original magnification, ×200)
In the current study, we examined BTLA expression in 253 cases of non-Hodgkin's mature B cell lymphomas including CLL/SLL, MCL, FL, MZL, DLBCL, and BL. All FFPE sections were subjected to single IHC with BTLA monoclonal antibody. BTLA expression was examined by double IHC and double IF in FFPE sections of several positive and negative cases of specific lymphomas in order to confirm the results of the single BTLA IHC staining. Double IHC examinations consisted of BTLA and PAX-5 in CLL/SLL, MZL, and DLBCL samples; BTLA and Bcl-6 in FL and BL samples and BTLA and Bcl-1 in MCL samples. Double IF examinations consisted of BTLA and CD2O staining and included all the cases of CLL/SLL and the positive cases of MCL, MZL, BL, and DLBCL.
CLL/SLL showed a significant difference in frequency of positive vs. negative BTLA staining (52/55 positive cases; χ2(1) = 43.65, p < 0.0001). BTLA expression in CLL/SLL is shown in Fig. 2a–c. Of note, a stronger staining of BTLA is commonly seen in the prolymphocytes and paraimmunoblasts of the proliferation center (Fig. 2a inset). BTLA expression by CLL/SLL cells was confirmed in all CLL/SLL samples by double staining (Fig. 2b, c). BTLA expression is absent in almost all but three samples of MCL (3/85 positive cases), showing significant difference in BTLA expression frequency (χ2(1) = 73.42, p < 0.0001). The pattern of expression in MCL is displayed in Fig. 2d–f. BTLA expression by the lymphoma B cells was confirmed in the positive cases of MCL by double IF (Fig. 2f).
BTLA expression in CLL/SLL and in MCL. a In nearly all the CLL/SLL cases, the neoplastic cells exhibit a strong labeling of BTLA (single IHC; original magnification, ×200). Inset: the large cells of the proliferation centers (arrowheads) often display a stronger staining (original magnification, ×400). b Most of the PAX-5 positive neoplastic B cells also express BTLA (double IHC with BTLA-brown and PAX-5-red; original magnification, ×400). c By double IF on FFPE sections, the CLL/SLL cells are labeled by CD20 (lower right inset) and BTLA (upper right inset) (CD20-red and BTLA-green; original magnification, ×400). d In most of MCL cases, the neoplastic cells are not stained with BTLA, while scattered reactive cells are labeled by BTLA (arrow) (single IHC; original magnification, ×200). e, f The three MCL cases expressing BTLA by single IHC are confirmed by double IF on FFPE sections with co-expression of CD20 and BTLA (e, original magnification, ×200; f, CD20-red and BTLA-green; original magnification, ×400)
Most DLBCL cases (29/35) did not express BTLA (Fig. 3a). Six cases (17 %) of DLBCL exhibited a faint to strong BTLA labeling of the lymphoma B cells (Fig. 3b, c). This difference in BTLA expression frequency was shown to be significant (χ2(1) = 15.11, p < 0.0001) (Table 1). Regarding BL, only 3 out of 18 cases showed strong expression of BTLA (Fig. 3d–g). BTLA expression by the lymphoma B cells was confirmed by double IHC and double IF in all the BTLA-positive cases of DLBCL (Fig. 3c) and BL (Fig. 3f, g). Of the 20 cases of MZL, 15 were negative for BTLA expression; however, in five cases the neoplastic B cells expressed BTLA (χ2(1) = 5.0, p = 0.025) as confirmed by double staining (data not shown). BTLA expression was not detected in any neoplastic cells of the FL samples of all grades (40 cases, data not shown). Summary of results is shown in Table 1.
BTLA expression in DLBCL and BL. a More than 80 % of the 35 samples of DLBCL studied do not express BTLA. Only some reactive small cells are stained (arrows) (single IHC; original magnification, ×200). b, c Six cases (17 %) exhibit a faint to strong BTLA labeling of the lymphoma cells (b, single IHC; original magnification, ×200; c, double IF; original magnification, ×400). d Regarding BL, most of the cases do not express BTLA and few reactive cells are positive (arrows) (single IHC original magnification, ×200). e–g Out of 18 cases, only 3 show strong expression of BTLA confirmed by double staining (e, single IHC; original magnification, ×200; f, double IHC of BTLA-brown and PAX-5-red; original magnification, ×400; g, double IF of CD20-red and BTLA-green; original magnification, ×600)
Discussion
The current study describes the pattern of expression of BTLA by neoplastic B cells in FFPE sections of human tissue in 253 samples of mature B cell lymphomas, using a recently developed anti-BTLA monoclonal antibody. To the best of our knowledge, it is the largest study examining BTLA expression in human B cell lymphomas and the first to report about BTLA expression in histopathology routine FFPE biopsy material. Previous findings regarding BTLA expression in lymphocytes were obtained in studies of mice [1, 5, 13–15] as well as human blood and bone marrow cells [10, 11, 16]. A limited number of human lymphoma fresh frozen tissue sections were also examined [9]. However, the later involved a technique rarely available for the diagnosis of lymphoma in daily practice in most medical centers.
The BTLA–HVEM complex is a key factor in the fine tuning of the immune response. BTLA inhibits the response of T lymphocytes by decreasing proliferation and cytokines production after TCR engagement and its activity is essential to prevent the development of autoimmune disease and for the regulation of the adaptive immunity, including antitumoral response [3, 17, 18].
The pattern of BTLA activity was extensively studied in T cells, but to a lesser extent in B lymphocytes. BTLA was found to be associated with the BCR complex-activating members IgM and CD79a/b. BTLA attenuates B cells activation by reducing downstream signaling of BCR complex-associated molecules, like spleen tyrosine kinase (Syk) and B cell linker protein, whose phosphorylation was shown to be reduced upon BTLA binding to HVEM after BCR engagement [4].
CLL/SLL is a disease with a highly variable clinical course related to the mutated or unmutated BCR status, among other factors, which corresponds to the pre- or post-GC cell of origin of the neoplasm [19]. The current study confirms the high expression of BTLA in CLL/SLL (Fig. 2), which is also found by M'hidi et al. [9]. BTLA, as an attenuator of the Syk-related BCR pathway activation, may play an important role in the course of CLL/SLL progression, especially as Syk signaling was found to enhance CLL/SLL neoplastic cells survival, adhesion, and chemotaxis [20].
The neoplastic B cells of the CLL/SLL were found to co-express PD-1 along with BTLA, in a very similar pattern [21]. The presence of proliferation centers is one of the histological hallmarks of the disease. In the current study, we observed a stronger expression of BTLA in the paraimmunoblasts and prolymphocytes of the CLL/SLL proliferation centers than in small neoplastic cells (Fig. 2a, inset), a feature that was also observed in PD-1 expression in CLL/SLL [21]. The simultaneous expression of BTLA and PD-1 has been recently shown to play a critical role in the mechanism responsible for immune escape that causes dysfunction of tumor-specific CD8(+) T cell [22, 23]. The role of the immune microenvironment is crucial for the response of B neoplastic cells to therapy and for long-term prognosis in mature B cell lymphomas [24–27]; For instance, it was shown the gene expression signature of the tumor-infiltrating and immune-reactive cells in FL is more predictive at diagnosis than the tumor cells properties [24, 25], and has a more crucial impact on the response to therapy than any other factor [26]. Concurrent targeting of BTLA and PD-1 pathways may play a crucial role, especially in preventing tumoral immune escape. It was demonstrated that tumor-specific T cell response can be restored by interference with the BTLA–HVEM pathway in combination with vaccination therapy in melanoma patients [28].
Finally, our study shows BTLA expression in 17 % of DLBCL cases, as confirmed by double IF (Fig. 3). DLBCL encompasses a heterogeneous group of diseases sharing the same morphologic features, yet having diverse responses to treatment protocols and overall survival [12, 16]. It is conceivable that the pattern of BTLA expression in DLBCL may distinguish subsets of patients. BCR status in DLBCL, which is related to BTLA function, as previously discussed [4, 29, 30], has been shown to have a prognostic impact through the Syk pathway.
Our results show a significant difference in the frequency of positive vs. negative BTLA expression in CLL/SLL, MCL, and DLBCL (Table 1), which indicates that the use of BTLA monoclonal antibody may be beneficial in the antibodies panel utilized for the differential diagnosis in routinely examined FFPE sections of mature B cell lymphomas. Further studies that will include larger number of cases and will relate to other prognostic factors may elucidate BTLA function in B cell lymphomas as a possible marker of specific disease subgroups as well as evaluate the potential use of BTLA as a therapeutic target.
References
Watanabe N, Gavrieli M, Sedy JR, Yang J, Fallarino F, Loftin SK, Hurchla MA, Zimmerman N, Sim J, Zang X, Murphy TL, Russel JH, Allison JP, Murphy KM (2003) BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat Immunol 4:670–679
Riley JL, June CH (2005) The CD28 family: a T-cell rheostat for therapeutic control of T-cell activation. Blood 105:13–21
Sakaguchi S, Ono M, Setoguchi R, Yagi H, Hori S, Fehevari Z, Shimizu J, Takahashi T, Nomura T (2006) Foxp3+ CD25+ CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol Rev 212:8–27
Vendel AC, Calemine-Fenaux J, Izrael-Tomasevic A, Chauhan V, Arnott D, Eaton DL (2009) B and T lymphocyte attenuator regulates B cell receptor signaling by targeting Syk and BLNK. J Immunol 182:1509–1517
Han P, Goularte OD, Rufner K, Wilkinson B, Kaye J (2004) An inhibitory Ig superfamily protein expressed by lymphocytes and APCs is also an early marker of thymocytes positive selection. J Immunol 172:5931–5939
Hurchla MA, Sedy JR, Gavrieli M, Drake CG, Murphy TL, Murphy KM (2005) B and T lymphocyte attenuator exhibits structural and expression polymorphisms and is highly induced in anergic CD4+ T cells. J Immunol 174:3377–3385
Compaan DM, Gonzalez LC, Tom I, Loyet KM, Eaton D, Hymowitz SG (2005) Attenuating lymphocyte activity: the crystal structure of the BTLA-HVEM complex. J Biol Chem 280:39553–39561
Sedy JR, Gavrieli M, Potter KG, Hurchla MA, Coleman L, Hildner K, Scheu S, Pfeffer K, Ware CF, Murphy TL, Murphy KM (2005) B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat Immunol 6:90–98
M'Hidi H, Thibult ML, Chetaille B, Rey F, Bouadallah R, Nicollas R, Olive D, Xerri L (2009) High expression of the inhibitory receptor BTLA in T-follicular helper cells and in B-cell small lymphocytic lymphoma/chronic lymphocytic leukemia. Am J Clin Pathol 132:589–596
Gonzalez LC, Loyet KM, Calemine-Fenaux J, Chauhan V, Wranik B, Ouyang W, Eaton DL (2005) A coreceptor interaction between the CD28 and TNF receptor family members B and T lymphocyte attenuator and herpesvirus entry mediator. PNAS 102(4):1116–1121
Hobo W, Norde WJ, Schaap N, Fredrix H, Maas F, Schellens K, Falkenburg JHF, Korman AJ, Olive D, van der Voort R, Dolstra H (2012) B and T lymphocyte attenuator mediates inhibition of tumor-reactive CD8+ T cells in patients after allogeneic stem cell transplantation. J Immunol 189(1):39–49
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW (2008) WHO classification of tumours of haematopoietic and lymphoid tissues. IARC, Lyon
Krieg C, Han P, Stone R, Goularte OD, Kaye J (2005) Functional analysis of B and T lymphocyte attenuator engagement on CD4+ and CD8+ T cells. J Immunol 175:6420–6427
Tao R, Wang L, Murphy KM, Fraser CC, Hancock WW (2008) Regulatory T cell expression of herpes virus entry mediator suppresses the function of B and T lymphocyte attenuator-positive effector T cells. J Immunol 180:6649–6655
Steinberg MW, Turovskaya O, Shaikh RB, Kim G, McCole DF, Pfeffer K, Murphy KM, Ware CF, Kronenberg M (2008) A crucial role for HVEM and BTLA in preventing intestinal inflammation. J Exp Med 205(6):1463–1476
Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A, Boldrick JC, Sabet H, Tran T, Yu X, Powell JI, Yang L, Marti GE, Moore T, Hudson J Jr, Lu L, Lewis DB, Tibshirani R, Sherlock G, Chan WC, Greiner TC, Weisenburger DD, Armitage JO, Warnke R, Levy R, Wilson W, Grever MR, Byrd JC, Botstein D, Brown PO, Staudt LM (2000) Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 403:503–511
Iwata A, Watanabe N, Oya Y, Owada T, Ikeda K, Suto A, Kagami S, Hirose K, Kanari H, Kawashima S, Nakayama T, Taniguchi M, Iwamoto I, Nakajima H (2010) Protective roles of B and T lymphocyte attenuator in NKT cell-mediated experimental hepatitis. J Immunol 184:127–133
Oki M, Watanabe N, Owada T, Oya Y, Ikeda K, Saito Y, Matsamura R, Seto Y, Iwamoto I, Nakajima H (2011) A functional polymorphism in B and T lymphocyte attenuator is associated with susceptibility to rheumatoid arthritis. Clin Dev Immunol. doi:10.1155/2011/305656
Hervé M, Xu K, Ng Y-S, Wardemann H, Albesiano E, Messmer BT, Chiorazzi N, Meffre E (2005) Unmutated and mutated chronic lymphocytic leukemias derives from self-reactive B cell precursors despite expressing different antibody reactivity. J Clin Invest 15:1636–1643
Quiroga MP, Balakrishnan K, Kurtova AV, Sivina M, Keating MJ, Wierda WG, Gandhi V, Burger JA (2009) B-cell antigen receptor signaling enhances chronic lymphocytic leukemia cells migration and survival: specific targeting with a novel spleen tyrosine kinase inhibitor, R406. Blood 114(5):1029–1037
Xerri L, Chetaille B, Seriari N, Atias C, Guillaume Y, Arnoulet C, Olive D (2008) Programmed death 1 is a marker of angioimmunoblastic T-cell lymphoma and B-cell small lymphocytic lymphoma/chronic lymphocytic leukemia. Human Pathol 39:1050–1058
Fourcade J, Sun Z, Pagliano O, Guillaume P, Luescher IF, Sander C, Kirkwood JM, Olive D, Kuchroo V, Zarour HM (2012) CD8+ T cells specific for tumor antigens can be rendered dysfunctional by the tumor microenvironment through upregulation of the inhibitory receptors BTLA and PD-1. Cancer Res 72:1–10
Norde WI, Hobo W, van der Voort R, Dolstra H (2012) Coinhibitory molecules in hematologic malignancies: targets for therapeutic intervention. Blood 120(4):728–736
Dave SS, Wright G, Tan B, Rosenwald A, Gascoyne RD, Chan WC, Fisher RI, Braziel RM, Rimsza LM, Grogan TM, Miller TP, LeBlanc M, Greiner TC, Weisenburger DD, Lynch JC, Julie Vose J, Armitage JO, Smeland EB, Kvaloy S, Harald Holte H, Delabie J, Connors JM, Lansdorp PM, Ouyang Q, Lister TA, Davies AJ, Norton AJ, Muller-Hermelink HK, Ott G, Campo E, Montserrat E, Wilson WH, Jaffe ES, Simon R, Yang L, Powell J, Zhao H, Goldschmidt N, Chiorazzi M, Staudt LM (2004) Prediction of survival in follicular lymphoma based on molecular features of tumor-infiltrating immune cells. N Engl J Med 351:2159–2169
Lejeune M, Alvaro T (2009) Clinicobiological prognostic and therapeutic implications of the tumor microenvironment in follicular lymphoma. Haematologica 94(1):16–21
De Jong D, Koster A, Hagenbeek A, Raemaekers J, Veldhuizen D, Heisterkamp S (2009) Impact of the tumor microenvironment on prognosis in follicular lymphoma is dependent on specific treatment protocols. Haematologica 94(1):70–77
Glas AM, Knoops L, Delahaye L, Kersten MJ, Kibbelaar RE, Wessels LA, van Laar R, van Krieken JH, Baars JW, Raemaekers J, Kluin PM, van't Veer LJ, de Jong D (2007) Gene-expression and immunohistochemical study of specific T-cell subsets and accessory cell types in the transformation and prognosis of follicular lymphoma. J Clin Oncol 25:390–398
Derré L, Rivals JP, Jandus C, Pastor S, Rimoldi D, Romero P, Michielin O, Olive D, Speiser DE (2010) BTLA mediates inhibition of human tumor-specific CD8+ T cells that can be partially reversed by vaccination. J Clin Invest 120(1):157–167
Burger JA, Ghia P, Rosenwald A, Caligaris-Cappio F (2009) The microenvironment in mature B-cell malignancies: a target for new treatment strategies. Blood 114:3367–3375
Chen L, Monti S, Juszczynski P, Daley J, Chen W, Witzig TE, Habermann TM, Kutok JL, Shipp MA (2008) SYK-dependent tonic B-cell receptor signaling is a rational treatment target in diffuse large B-cell lymphoma. Blood 111:2230–223
Acknowledgments
We would like to acknowledge Dr. Giovanna Roncador, head of the Monoclonal Antibodies Unit, CNIO (Madrid, Spain), for the provision of the BTLA monoclonal antibody and her very helpful advices. We are also very grateful to Dr. Avi Avital for his assistance in the statistical analysis and his review of this manuscript. Many thanks for the support of Dr. Irit Elmalah and Shulamit Goez from the Tissue Diagnosis and Cancer Research department at the Ha’Emek Medical Center (Afula, Israel).
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trougouboff, P., Kreizman Shefer, H. B and T lymphocyte attenuator expression in mature B cell lymphomas. J Hematopathol 6, 57–63 (2013). https://doi.org/10.1007/s12308-012-0173-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-012-0173-5