Abstract
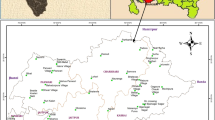
The present study includes detailed hydrochemical assessment of groundwater resources of Saq aquifer. The Saq aquifer covers a large area (about 375,000 km2) and lies in the arid region with low annual rainfall and extremely high evaporation. In the study area, groundwater serves as the major source for agricultural activity and for domestic usages. A total of 295 groundwater samples collected and were analyzed for physico-chemical parameters such as hydrogen ion concentration (pH), total dissolved solids (TDS) and electrical conductivity (EC), sodium (Na+), potassium (K+), magnesium (Mg2+), and calcium (Ca2+), bicarbonate (HCO3) chloride (Cl−), sulfate (SO4 2−) and nitrate (NO3). The goal and challenge for the statistical overview was to delineate chemical distributions in a complex, heterogeneous set of data spanning over a large geographic range. After de-clustering to create a uniform spatial sample distribution with 295 samples, histograms and quantile-quantile (Q-Q) plots were employed to delineate subpopulations that have coherent chemical affinities. The elements showing significantly higher positive correlation are: TDS with EC; Ca with EC, TDS; Mg and EC, TDS, Ca, K; Cl and EC, TDS, Mg, Na, Ca; SO4 and EC, TDS, Ca, Cl, Na, Mg. The distribution of major ions in the groundwater is Na+ > Ca++ > Mg++ > K+ and Cl− > SO4 −2>HCO3 −>NO3 −. Ionic abundance plot of alkalis with Ca and Mg is suggestive of mix type trends of concentrations as evident by moderate correlation (r = 0.57). About 60% of the total samples have alkalis abundance and rests have more Ca + Mg concentrations than alkalis. Taking both results of cluster tree and geochemical features of variables into consideration, the authors classify the elements into two major groups, the first includes TDS, Na, EC, Cl, Ca, SO4, and Mg, where the relationship within the group are strong. The second group includes K, HCO3, pH, and NO3. This group has close relationship with group 1 demonstrate that, the increase in the concentration to some elements could be the same. Some of the analyzed parameters approach a normal distribution, as both their skewnesses and kurtoses reach close to “0”. The study revealed that, all of the element pairs exhibit positive relations.
Similar content being viewed by others
References
Al-Amry, A.S., 2008, Hydrogeochemistry and Groundwater Quality Assessment in an Arid Region: A Case Study from Al Salameh Area, Shabwah, Yemen. In: The 3rd International Conference on Water Resources and Arid Environments, the 1st Arab Water Forum, p. 1–19.
Al-Bassam, A.M., Hussein, T., and Sharaf, M.A., 1997, Hydrochemical evaluation of the Umm-er-Radhuma aquifer system, Saudi Arabia, Hydrochemistry. Proceedings of the Rabat Symposium, April 1997, IAHS Publication, 244, 13–26.
American public Health association (APHA), 1995, Standard methods for the examination of water and wastewater, 19th edition. APHA, Washington DC, 45 p.
Nas, B., Cay, T., Iscan, F., and Berktay, A., 2010, Selection of MSW landfill site for Konya, Turkey using GIS and multi-criteria evaluation. Environmental Monitoring Assessment, 160, 491–500.
BUREAU DE RECHERCHES GÉOLOGIQUES ET MINIÈRES (BRGM), 1985, Water, agriculture and soil studies of Saq and overlying aquifers. Unpublished report on file, Ministry of Agriculture, Kingdom of Saudi Arabia, 279 p.
Cloutier, V., Lefebvre, R., Therrien, R., and Savard, M., 2008, Multivariate statistical analysis of geochemical data as indicative of the hydrogeochemical evolution of groundwater in a sedimentary rock aquifer system. Hydrogeology Journal, 353, 294–313.
Freeze, R.A. and Cherry, J.A., 1979, Groundwater. Prentice Hall, New Jersey, 123 p.
Garcia, M.G., Del Hidalgo, M., and Blesa, M., 2001, Geochemistry of groundwater in the alluvial plain of Tucuman province, Argentina. Hydrogeology Journal, 9, 597–610.
Ministry of Water and Electricity, Kingdom of Saudi Arabia (MoWE), 2008, Investigations for updating the groundwater mathematical models of the Saq and overlying aquifers. Unpublished report on file, Ministry of Water and Electricity, Kingdom of Saudi Arabia, 302 p.
Sall, J. and Lehman, A., 1996, A Guide to Statistic and Data Analysis Using JMP and JMP IN Software. Duxbury Press, Belmont, 521 p.
Sawyer, C.N. and McCarty, P.L., 1967, Chemistry for sanitary, 2nd edition. McGraw Hill, New York, 518 p.
Schoeller, H., 1965, Hydrodynamique lans le karst. Actes du Colloque de Dubrovnik, IAHS, UNESCO, p. 3–20.
Sinclair, A.J., 1974, Selection of threshold values in geochemical data using probability graphs. Journal Geochemical Exploration, 2, 129–149.
Soltan, M.E., 1998, Characterization, classification and evaluation of some groundwater samples in upper Egypt. Journal Chemosphere, 37, 735–745.
Subba Rao, N., Surya Rao, P., Venktram Reddy, G., Nagamani, M., Vidyasagar, G., and Satyanarayana, N.L.V.V., 2012, Chemical characteristics of groundwater and assessment of groundwater quality in Varaha River Basin, Visakhapatnam District, Andhra Pradesh, India. Environmental Monitoring and Assessment, 184, 5189–5214.
Todd, D.K., 1980, Groundwater hydrology, 3rd edition. Wiley, New York, 656 p.
World Health Organization (WHO), 2011, Guidelines for drinkingwater quality, fourth edition. ISBN: 9789241548151, 564 p.
Zhang, C.S., Fay, D., McGrath, D., Grennan, E., and Carton, O.T., 2008, Statistical analysis of geochemical variables in soils of Ireland. Geoderma, 146, 378–390.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nazzal, Y., Ahmed, I., Al-Arifi, N.S.N. et al. A combined hydrochemical-statistical analysis of Saq aquifer, northwestern part of the Kingdom of Saudi Arabia. Geosci J 19, 145–155 (2015). https://doi.org/10.1007/s12303-014-0016-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12303-014-0016-8