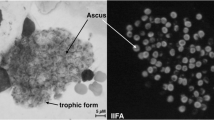
Abstract
Pneumocystis jirovecii is a widespread fungal colonizer of the human lung. Proliferation of the pathogen in the alveoli is controlled by the immune system in healthy individuals. When the immune system is impaired, pneumocystosis can emerge, resulting in a pulmonary infection. Formerly, the disease occurred mainly in acquired immune deficiency syndrome (AIDS) patients, accompanied by a high mortality. Now it is increasingly seen in patients with immunosuppressive treatment. Traditionally, laboratory diagnosis is based on the microscopic detection of cysts and trophic forms of P. jirovecii in respiratory samples. Quantitative PCR-based methods will revolutionize laboratory diagnosis. However, cutoffs have to be established to discriminate between colonization (clinically irrelevant) and infection. Furthermore, the data on the serological detection of (1→3)-ß-D-glucan to diagnose or exclude pneumocystosis is promising.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Walzer DP, Smulian AG. Pneumocystis species. In: Mandel GL, Bennett JE, Dolin R, editors. Principales and practice of infectious diseases. 7th ed. Philadelphia: Churchill Livingstone Elsevier; 2010. p. 3377–90.
Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. http://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf. Accessed 4/5/2014.
Maini R, Henderson KL, Sheridan ES, et al. Increasing Pneumocystis pneumonia, England, UK, 2000–2010. Emerg Infect Dis. 2013;19:386–92. This study shows the significant increase of pneumocystosis in different immunosuppressed patient groups.
Coyle PV, McCaughey C, Nager A, et al. Rising incidence of Pneumocystis jirovecii pneumonia suggests iatrogenic exposure of immune-compromised patients may be becoming a significant problem. J Med Microbiol. 2012;61:1009–15.
De Boer MGJ, De Fijter JW, Kroon FP. Outbreaks and clustering of Pneumocystis pneumonia in kidney transplant recipients: a systematic review. Med Mycol. 2011;49:673–80.
Tasaka S, Tokuda H. Pneumocystis jirovecii pneumonia in non-HIV-infected patients in the era of novel immunosuppressive therapies. J Infect Chemother. 2012;18:793–806.
Ng VL, Yajko DM, Hadley WK. Extrapulmonary pneumocystosis. Clin Microbiol Rev. 1997;10:401–18.
Thomas CF, Limper AH. Current insights into the biology and pathogenesis of Pneumocystis pneumonia. Nat Rev Microbiol. 2007;5:298–308.
Steele C, Shellito JE, Kolls JK. Immunity against the opportunistic fungal pathogen Pneumocystis. Med Mycol. 2005;43:1–19.
Nevez G, Chabé M, Rabodonirina M, Virmaux M, et al. Nosocomial Pneumocystis jirovecii infections. Parasite. 2008;15:359–65.
Martinez A, Halliez MCM, Aliouat EM, et al. Growth and airborne transmission of cell-sorted life cycle stages of Pneumocystis carinii. PLoS One. 2013;8:e79958. This study shows that the cysts are infective.
Peglow SL, Smulian AG, Linke MJ, et al. Serological responses to Pneumocystis carinii antigens in health and disease. J Infect Dis. 1990;161:296–306.
Pifer LL, Huges WT, Stagno S, Woods D. Pneumocystis carinii infection: evidence for high prevalence in normal and immunosuppressed children. Pediatrics. 1978;61:35–41.
Respaldiza N, Medrano FJ, Medrano AC, et al. High seroprevalence of Pneumocystis infection in Spanish children. Clin Microbiol Infect. 2004;10:1029–31.
Vargas SL, Pizarro P, López-Vieyra M, et al. Pneumocystis colonization in older adults and diagnostic yield of single versus paired noninvasive respiratory sampling. Clin Infect Dis. 2010;50:e19–21.
Ponce CA, Gallo M, Bustamante R, Vargas SL. Pneumocystis colonization is highly prevalent in the autopsied lungs of the general population. Clin Infect Dis. 2010;50:347–53.
Vargas SL, Ponce CA, Sanchez CA, et al. Pregnancy and asymptomatic carriage of Pneumocystis jiroveci. Emerg Infect Dis. 2003;9:605–6.
Morris A, Norris KA. Colonization by Pneumocystis jirovecii and its role in disease. Clin Microbiol Rev. 2012;25:297–317. Comprehensive review on different aspects of pneumocystis.
Shipley TW, Kling HM, Morris A, et al. Persistent Pneumocystis colonization leads to the development of chronic obstructive pulmonary disease in a nonhuman primate model of AIDS. J Infect Dis. 2010;202:302–12.
Chagas C. Nova tripanozomiaze humana. Ueber eine neue Trypanosomiasis des Menschen. Mem Inst Oswaldo Cruz. 1909;1:159–218.
Delanoë P, Delanoë M. Sur les rapports des kystes de Carinii du poumon des rates avec Trypanosoma lewisi. CR Acad Sci Gen. 1912;155:658–60.
Vavra J, Kucera K. Pneumocystis carinii Delanoë, its ultrastructure and ultrastructural affinities. J Protozool. 1970;17:463–83.
Edman JC, Kovacs JA, Masur H, et al. Ribosomal RNA sequence shows Pneumocystis carinii to be a member of the fungi. Nature. 1988;334:519–20.
Stringer SL, Stringer JR, Blase MA, et al. Pneumocystis carinii: sequence from ribosomal RNA implies a close relationship with fungi. Exp Parasitol. 1989;68:450–61.
Frenkel JK. Pneumocystis pneumonia, an immunodeficiency-dependent disease (IDD): a critical historical overview. J Eukaryot Microbiol. 1999;46:89S–92.
Stringer JR, Beard CB, Miller RF. Spelling Pneumocystis jirovecii. Emerg Infect Dis. 2009;15:506.
Martinez A, Aliouat EM, Standaert-Vitse A, et al. Ploidy of cell-sorted trophic and cystic forms of Pneumocystis carinii. PLoS One. 2011;6:e20935.
Gigliotti F, Harmsen AG, Haidaris CG, Haidaris PJ. Pneumocystis carinii is not universally transmissible between mammalian species. Infect Immun. 1993;61:2886–90.
Durand-Joly I, Aliouat EM, Recourt C, et al. Pneumocystis carinii f. sp. hominis is not infectious for SCID mice. J Clin Microbiol. 2002;40:1862–5.
Guillot J, Demanche C, Hugot JP, et al. Parallel phylogenies of Pneumocystis species and their mammalian hosts. J Eukaryot Microbiol. 2001;113S-115S.
Keely SP, Fischer JM, Stringer JF. Evolution and speciation of Pneumocystis. J Eukaryot Microbiol. 2003;50:S624–6.
Hauser PM, Burdet FX, Cissé OH, et al. Comparative genomics suggests that the fungal pathogen Pneumocystis is an obligate parasite scavenging amino acids from ist hosts´s lungs. PLoS One. 2010;5:e15152.
Tipirneni R, Daly KR, Jarlsberg LG, et al. Healthcare workers occupation and immune response to Pneumocystis jirovecii. Emerg Infect Dis. 2009;15:1590–7.
Fong S, Daly KR, Tipireni R, et al. Antibody responses against Pneumocystis jirovecii in health care workers over time. Emerg Infect Dis. 2013;19:1612–9.
Miller RF, Ambrose HE, Wakefield AE. Pneumocystis carinii f. sp. hominis DNA in immunocompetent health care workers in contact with patients with P. carinii pneumonia. J Clin Microbiol. 2001;39:3877–82.
Lundgren B, Elvin K, Rothman LP, et al. Transmission of Pneumocystis carinii from patients to hospital staff. Thorax. 1997;52:422–4.
Cruciani M, Marcati P, Malena M, et al. Meta-analysis of diagnostic procedures for Pneumocystis carinii pneumonia in HIV-1-infected patients. Eur Respir J. 2002;20:982–9.
Wang Y, Doucette S, Qian Q, Kirby JE. Yield of primary and repeat induced sputum testing for Pneumocystis jiroveci in human immunodeficiency virus-positive and –negative patients. Arch Pathol Lab Med. 2007;131:1582–4.
To KKW, Wong SCY, Xu T, et al. Use of nasopharyngeal aspirate for diagnosis of Pneumocystis pneumonia. J Clin Microbiol. 2013;51:1570–4.
Finkelman MA. Pneumocystis jirovecii infection: cell wall (1→3)-ß-D-glucan biology and diagnostic utility. Crit Rev Microbiol. 2010;36:271–81.
Onishi A, Sugiyama D, Kogata Y, et al. Diagnostic accuracy of serum 1,3-ß-D-glucan for Pneumocystis jiroveci pneumonia, invasive candidiasis, and invasive aspergillosis: systematic review and meta-analysis. J Clin Microbiol. 2012;50:7–15.
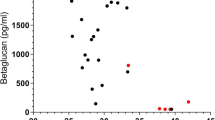
Karageorgopoulos DE, Qu J-M, Korbila IP, et al. Accuracy of ß-D-Glucan for the diagnosis of Pneumocystis jirovecii pneumonia: a meta-analysis. Clin Microbiol Infect. 2013;19:39–49. Up-to-date meta-analysis on the value of ß-D-glucan in pneumocystosis.
Nakamura H, Tateyama M, Tasato D, et al. Clinical utility of serum ß-D-glucan and KL-6 levels in Pneumocystis jirovecii pneumonia. Int Med. 2009;48:195–202.
Limper AH, Offord KP, Smith TF, MartinII WJ. Pneumocystis carinii pneumonia: differences in lung parasite number and inflammation in patients with and without AIDS. Am Rev Respir Dis. 1989;140:1204–9.
Theel ES, Doern CD. ß-D-glucan testing is important for diagnosis of invasive fungal infections. J Clin Microbiol. 2013;51:3478–83.
Chen SC, Kontoyiannis DP. New molecular and surrogate biomarker-based tests in the diagnosis of bacterial and fungal infection in febrile neutropenic patients. Curr Opin Infect Dis. 2010;23:567–77.
Centers of Disease Control and Prevention. DPDx – Laboratory identification of parasitic diseases of public health concern. Diagnosic procedeures. Sputum, induced sputum and bronchoalveolar lavage (BAL) for Pneumocystis jirovecii. http://www.cdc.gov/dpdx/diagnosticProcedures/other/BAL.html Accessed 4/4/2014.
Calderón EJ, Gutiérrez-Rivero S, Durand-Joly I, Dei-Cas E. Pneumocystis infection in humans: diagnosis and treatment. Expert Rev Anti-Infect Ther. 2010;8:683–701.
Harrington BJ. Staining of cysts of Pneumocystis jiroveci (P. carinii) with the fluorescent brighteners Calcofluor white and Uvitex 2B: a review. Labmedicine. 2008;39:731–5.
Procop GW, Haddad S, Quinn J, et al. Detection of Pneumocystis jiroveci in respiratory specimens by four staining methods. J Clin Microbiol. 2004;42:3333–5.
Wakefield AE, Pixley FJ, Banerji S, et al. Detection of Pneumocystis carinii with DNA amplification. Lancet. 1990;336:451–3.
Lu Y, Ling G, Qiang C, et al. PCR diagnosis of Pneumocystis pneumonia: a bivariate meta-analysis. J Clin Microbiol. 2011;49:4361–3.
Fan L-C, Lu H-W, Cheng K-B, et al. Evaluation of PCR in bronchoalveolar lavage fluid for diagnosis of Pneumocystis jirovecii pneumonia: a bivariate meta-analysis and systematic review. PLoS One. 2013;8:e73099. Up-to-date meta-analysis on the value of PCR in pneumocystosis.
McTaggart LR, Wengenack NL, Richardson SE. Validation of the MycAssay Pneumocystis kit for the detection of Pneumocystis jirovecii in bronchoalveolar lavage specimens by comparison to a laboratory standard of direct immunofluorescence microscopy, real-time PCR, or conventional PCR. J Clin Microbiol. 2012;50:1856–9.
Hauser PM, Bille J, Lass-Flörl C, et al. Multicenter, prospective clinical evaluation of respiratory samples from subjects at risk for Pneumocystis jirovecii infection by use of a commercial real-time PCR assay. J Clin Microbiol. 2011;49:1872–8. Multicenter study on a commercially available PCR assay.
Dalpke AH, Hofko M, Zimmermann S. Development and evaluation of a real-time PCR assay for detection of Pneumocystis jirovecii on the fully automated BD MAX platform. J Clin Microbiol. 2013;51:2337–43.
Botterel F, Cabaret O, Foulet F, et al. Clinical significance of quantifying Pneumocystis jirovecii DNA by using real-time PCR in bronchoalveolar lavage fluid from immunocompromised patients. J Clin Microbiol. 2012;50:227–31.
Maillet M, Maubon D, Brion JP, et al. Pneumocystis jirovecii (Pj) quantitative PCR to differentiate Pj pneumonia from Pj colonization in immunocompromised patients. Eur J Clin Microbial Infect Dis. 2014;33:331–6.
Alanio A, Desoubeaux G, Sarfati C, et al. Real-time PCR assay-based strategy for differentiation between active Pneumocystis jirovecii pneumonia and colonization in immunocompromised patients. Clin Microbiol Infect. 2011;17:1531–7.
Damiani C, Le Gal S, Da Costa C, et al. Combined quantification of pulmonary Pneumocystis jirovevii DNA and serum (1→3)-ß-D-glucan for differential diagnosis of Pneumocystis pneumonia and Pneumocystis colonization. J Clin Microbiol. 2013;51:3380–8. This study shows that ß-D-glucan can be used to discriminate pneumonia and colonization in PCR positive patients.
Acknowledgments
We thank Pedrina Gonçalves Vidigal and David Killengray for careful review of the English text.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
PM Rath has received research support from Pfizer and Forest, and a speaker honorarium from Roche Diagnostics.
J Steinmann has received research support from Pfizer and Forest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rath, PM., Steinmann, J. Update on Diagnosis of Pneumocystis Pulmonary Infections. Curr Fungal Infect Rep 8, 227–234 (2014). https://doi.org/10.1007/s12281-014-0188-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12281-014-0188-8