Abstract
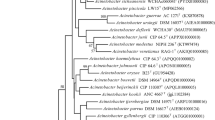
A non-motile and rod shaped bacterium, designated strain B1T, was isolated from forest soil at Mt. Baekwoon, Republic of Korea. Cells were Gram-negative, catalase-positive, and oxidase-negative. The major fatty acids were 9-octadecenoic acid (C18:1 ω9c; 42%) and hexadecanoic acid (C16:0; 25.9%) and summed feature 3 (comprising iso-C15:0 2-OH and/or C16:1 ω7c; 10.0%). The DNA G+C content was 44.1 mol%. A phylogenetic tree based on 16S rRNA gene sequences showed that strain B1T formed a lineage within the genus Acinetobacter and was closely related to A. baylyi DSM 14961T (98.6% sequence similarity), followed by A. baumannii DSM 30007T (97.4%), A. calcoaceticus DSM 30006T (97.0%) and 3 genomic species (96.8∼7.6%). Phenotypic characteristics, gyrB gene sequence analysis and DNA-DNA relatedness data distinguished strain B1T from type strains of A. baylyi, A. baumannii, and A. calcoaceticus. On the basis of the evidence presented in this study, strain B1T represents a novel species of the genus Acinetobacter, for which the name Acinetobacter soli sp. nov. is proposed. The type strain is B1T (= KCTC 22184T= JCM 15062T).
Similar content being viewed by others
References
Baik, K.S., Y.D. Park, C.N. Seong, E.M. Kim, K.S. Bae, and J. Chun. 2006. Glaciecola nitratireducens sp. nov. isolated from sea water. Int. J. Syst. Evol. Microbiol. 56, 2185–2188.
Bouvet, P.J.M. and P.A.D. Grimont. 1986. Taxonomy of the genus Acinetobacter with the recognition of Acinetobacter baumannii sp. nov., Acinetobacter haemolyticus sp. nov., Acinetobacter johnsonii sp. nov., and Acinetobacter junii sp. nov. and emended descriptions of Acinetobacter calcoaceticus and Acinetobacter lwoffii. Int. J. Syst. Bacteriol. 36, 228–240.
Bouvet, P.J.M. and S. Jeanjean. 1989. Delineation of new proteolytic genomic species in the genus Acinetobacter. Res. Microbiol. 140, 291–299.
Carr, E.L., P. Kämpfer, B.K.C. Patel, V. Gürtler, and R.J. Seviour. 2003. Seven novel species of Acinetobacter isolated from activated sludge. Int. J. Syst. Evol. Microbiol. 53, 953–963.
Chun, J., K.S. Bae, E.Y. Moon, S.O. Jung, H.K. Lee, and S.J. Kim. 2000. Nocardiopsis kunsanensis sp. nov., a moderately halophilic actinomycete isolated from a saltern. Int. J. Syst. Evol. Microbiol. 50, 1909–1913.
Chun, J. and M. Goodfellow. 1995. A phylogenetic analysis of the genus Nocardia with 16S rRNA gene sequences. Int. J. Syst. Bacteriol. 45, 240–245.
Chun, J., J.H. Lee, Y. Jung, M. Kim, S. Kim, B.K. Kim, and Y.W. Lim. 2007. EzTaxon: a web-based tool for the identification of prokaryotes based on 16S ribosomal RNA gene sequences. Int. J. Syst. Evol. Microbiol. 57, 2259–2261.
CLSI. 2003. Performance standards for antimicrobial disk susceptibility tests, 8th ed. Approved Standard M2-A8, Clinical Laboratory Standards Institute, Wayne, PA, USA.
Collins, M.D. and D. Jones. 1981. Distribution of isoprenoid quinone structural types in bacteria and their taxonomic implications. Microbiol. Rev. 45, 316–354.
Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39, 783–791.
Fitch, W.M. and E. Margoliash. 1967. Construction of phylogenetic trees. Science 155, 279–284.
Gerner-Smidt, P. and I. Tjernberg. 1993. Acinetobacter in Denmark: II. Molecular studies of the Acinetobacter calcoaceticus-Acinetobacter baumannii complex. APMIS 101, 826–832.
Gordon, R.E. and J.M. Mihm. 1962. Identification of Nocardia caviae (Erikson) nov. comb. Ann. N. Y. Acad. Sci. 98, 628–636.
Jukes, T.H. and C.R. Cantor. 1969. Evolution of protein molecules, p. 21–132. In H.N. Munro (ed.), Mammalian protein metabolism. Academic Press. New York, N.Y., USA.
Kovacs, N. 1956. Identification of Pseudomonas pyocyanea by the oxidase reaction. Nature 178, 703.
Marmur, J. and P. Doty. 1962. Determination of the base composition of deoxyribonucleic acid from its thermal denaturation temperature. J. Mol. Biol. 5, 109–118.
MIDI. 1999. Sherlock Microbial Identification System Operating Manual, version 3.0. MIDI Inc., Newark, DE, USA.
Minnikin, D.E., A.G. O’Donnell, M. Goodfellow, G. Alderson, M. Athalye, A. Schaal, and J.H. Parlett. 1984. An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J. Microbiol. Methods 2, 233–241.
Nemec, A., T. De Baere, I. Tjernberg, M. Vaneechoutte, T.J.K. Van Der Reijden, and L. Dijkshoorn. 2001. Acinetobacter ursingii sp. nov. and Acinetobacter schindleri sp. nov., isolated from human clinical specimens. Int. J. Syst. Evol. Microbiol. 51, 1891–1899.
Nemec, A., L. Dijkshoorn, I. Cleenwerck, T. De Baere, D. Janssens, T.J.K. Van Der Reijden, P. Jezek, and M. Vaneechoutte. 2003. Acinetobacter parvus sp. nov., a small-colony-forming species isolated from human clinical specimens. Int. J. Syst. Evol. Microbiol. 53, 1563–1567.
Nishimura, Y., T. Ino, and H. Iizuka. 1988. Acinetobacter radioresistens sp. nov. isolated from cotton and soil. Int. J. Syst. Bacteriol. 38, 209–211.
Richard, C. and M. Kiredjian. 1995. Laboratory Methods for the Identification of Strictly Aerobic Gram-negative Bacilli. Institut Pasteur, Paris, France.
Saitou, N. and M. Nei. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425.
Seldin, L. and D. Dubnau. 1985. Deoxyribonucleic acid homology among Bacillus polymyxa, Bacillus macerans, Bacillus azotofixans, and other nitrogen-fixing Bacillus strains. Int. J. Syst. Bacteriol. 35, 151–154.
Skerman, V.B.D. 1967. A Guide to the Identification of the Genera of Bacteria, 2nd ed. Williams and Wilkins, Baltimore, USA.
Smibert, R.M. and N.R. Krieg. 1994. Phenotypic characterization, p. 607–654. In P. Gerhardt, R.G.E. Murray, W.A. Wood, and N.R. Krieg (eds.), Methods for General and Molecular Bacteriology. American Society for Microbiology, Washington, D.C., USA.
Swofford, D.L. 1998. Phylogenetic analysis using parsimony (PAUP). Version 4. Sinauer Associates, Suderland, MA, USA.
Thompson, J.D., D.G. Higgins, and T.J. Gibson. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680.
Tjernberg, I. and J. Ursing. 1989. Clinical strains of Acinetobacter classified by DNA-DNA hybridization. APMIS 97, 595–605.
Vaneechoutte, M., I. Tjernberg, F. Baldi, M. Pepi, R. Fani, E.R. Sullivan, J. Van Der Toorn, and L. Dijkshoorn. 1999. Oil-degrading Acinetobacter strain RAG-1 and strains described as ‘Acinetobacter venetianus sp. nov.’ belong to the same genomic species. Res. Microbiol. 150, 69–73.
Wayne, L.G., D.J. Brenner, R.R. Colwell, P.A.D. Grimont, O. Kandler, M.I. Krichevsky, L.H. Moore, W.E.C. Moore, R.G.E. Murray, E. Stackebrandt, M.P. Starr, and H.G. Trüper. 1987. International Committee on Systematic Bacteriology. Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int. J. Syst. Bacteriol. 37, 463–464.
Yamamoto, S. and S. Harayama. 1995. PCR Amplification and direct sequencing of gyrB genes with universal primers and their application to the detection and taxonomic analysis of Pseudomonas putida strains. Appl. Environ. Microbiol. 61, 1104–1109.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, D., Baik, K.S., Kim, M.S. et al. Acinetobacter soli sp. nov., isolated from forest soil. J Microbiol. 46, 396–401 (2008). https://doi.org/10.1007/s12275-008-0118-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-008-0118-y