Abstract
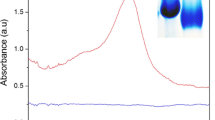
A facile and low-cost method to prepare periodic Au@metal–organic framework (MOF) (MIL-100(Fe)) nanoparticle arrays was developed. The arrays were fabricated in situ using monolayer colloidal crystals as templates, followed by Au deposition on substrates, and annealing. MIL-100(Fe) coatings were applied on the nanospheres using a simple solvent thermal process. The prepared periodic Au@MIL-100(Fe) nanoparticle (NP) arrays were characterized by two peaks in the visible spectra. The first peak represented the surface plasmon resonance (SPR) of the Au nanospheres, and the other peak, or the diffraction peak, originated from the periodic structure in the NP array. After modification with 3-aminophenylboronic acid hemisulfate (PBA), the Au@MIL-100(Fe) NP arrays exhibited sensitive responses to different glucose concentrations with good selectivity. These responses could be due to the strong interaction between PBA and glucose molecules. The diffraction peak was sensitive at low glucose concentrations (less than 12 mM), whereas the SPR peak rapidly responded at high concentrations. The peaks thus demonstrated satisfactory complementary sensitivity for glucose detection in different concentration regions. These results can be used to develop a dual-channel biosensor. We also created a standard diagram, which can be used to efficiently monitor blood glucose levels. The proposed strategy can be extended to develop different dual-channel sensors using Au@MIL-100(Fe) NP arrays functionalized with different recognition agents.

Similar content being viewed by others

References
Som, T.; Karmakar, B. Core-shell Au-Ag nanoparticles in dielectric nanocomposites with plasmon-enhanced fluorescence: A new paradigm in antimony glasses. Nano Res. 2009, 2, 607–616.
Li, P.; Wei, Z.; Wu, T.; Peng, Q.; Li, Y. D. Au-ZnO hybrid nanopyramids and their photocatalytic properties. J. Am. Chem. Soc. 2011, 133, 5660–5663.
Park, B.; Kim, S. J.; Sohn, J. S.; Nam, M. S.; Kang, S.; Jun, S. C. Surface plasmon enhancement of photoluminescence in photo-chemically synthesized graphene quantum dot and Au nanosphere. Nano Res. 2016, 9, 1866–1875.
Ding, D. W.; Liu, K.; He, S. N.; Gao, C. B.; Yin, Y. D. Ligand-exchange assisted formation of Au/TiO2 schottky contact for visible-light photocatalysis. Nano Lett. 2014, 14, 6731–6736.
Liu, H. P.; Liu, T. Z.; Zhang, L.; Han, L.; Gao, C. B.; Yin, Y. D. Etching-free epitaxial growth of gold on silver nanostructures for high chemical stability and plasmonic activity. Adv. Funct. Mater. 2015, 25, 5435–5443.
Li, C. C.; Shuford, K. L.; Chen, M. H.; Lee, E. J.; Cho, S. O. A facile polyol route to uniform gold octahedra with tailorable size and their optical properties. ACS Nano 2008, 2, 1760–1769.
Liu, D. L.; Zhou, F.; Li, C. C.; Zhang, T.; Zhang, H. H.; Cai, W. P.; Li, Y. Black gold: Plasmonic colloidosomes with broadband absorption self-assembled from monodispersed gold nanospheres by using a reverse emulsion system. Angew. Chem., Int. Ed. 2015, 54, 9596–9600.
Zhou, Q.; Yang, Y.; Ni, J.; Li, Z. C.; Zhang, Z. J. Rapid recognition of isomers of monochlorobiphenyls at trace levels by surface-enhanced Raman scattering using Ag nanorods as a substrate. Nano Res. 2010, 3, 423–428.
Luo, M.; Ruditskiy, A.; Peng, H. C.; Tao, J.; Figueroa- Cosme, L.; He, Z. K.; Xia, Y. N. Penta-twinned copper nanorods: Facile synthesis via seed-mediated growth and their tunable plasmonic properties. Adv. Funct. Mater. 2016, 26, 1209–1216.
Pyykkö, P. Theoretical chemistry of gold. III. Chem. Soc. Rev. 2008, 37, 1967–1997.
Dodson, S. L.; Cao, C.; Zaribafzadeh, H.; Li, S. Z.; Xiong, Q. H. Engineering plasmonic nanorod arrays for colon cancer marker detection. Biosens. Bioelectron. 2015, 63, 472–477.
Xue, B.; Chen, P.; Hong, Q.; Lin, J. Y.; Tan, K. L. Growth of Pd, Pt, Ag and Au nanoparticles on carbon nanotubes. J. Mater. Chem. 2001, 11, 2378–2381.
Qian, L. H.; Mookherjee, R. Convective assembly of linear gold nanoparticle arrays at the micron scale for surface enhanced Raman scattering. Nano Res. 2011, 4, 1117–1128.
Ruditskiy, A.; Xia, Y. N. Toward the synthesis of sub-15 nm Ag nanocubes with sharp corners and edges: The roles of heterogeneous nucleation and surface capping. J. Am. Chem. Soc. 2016, 138, 3161–3167.
Men, D. D.; Zhou, F.; Hang, L. F.; Li, X. Y.; Duan, G. T.; Cai, W. P.; Li, Y. A functional hydrogel film attached with a 2D Au nanosphere array and its ultrahigh optical diffraction intensity as a visualized sensor. J. Mater. Chem. C 2016, 4, 2117–2122.
Kelly, K. L.; Coronado, E.; Zhao, L. L.; Schatz, G. C. The optical properties of metal nanoparticles: The influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668–677.
Zhou, F.; Liu, Y.; Cai, W. P. Huge local electric field enhancement in hybrid plasmonic arrays. Opt. Lett. 2014, 39, 1302–1305.
Meek, S. T.; Greathouse, J. A.; Allendorf, M. D. Metalorganic frameworks: A rapidly growing class of versatile nanoporous materials. Adv. Mater. 2011, 23, 249–267.
He, K.; Cao, Z.; Liu, R. R.; Miao, Y.; Ma, H. Y.; Ding, Y. In situ decomposition of metal-organic frameworks into ultrathin nanosheets for the oxygen evolution reaction. Nano Res. 2016, 9, 1856–1865.
Li, H. L.; Eddaoudi, M.; O’Keeffe, M.; Yaghi, O. M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279.
Kitagawa, S.; Kitaura, R.; Noro, S. I. Functional porous coordination polymers. Angew. Chem., Int. Ed. 2004, 43, 2334–2375.
Zhang, L.; Wu, H. B.; Lou, X. W. Metal–organicframeworks- derived general formation of hollow structures with high complexity. J. Am. Chem. Soc. 2013, 135, 10664–10672.
Zhang, W.; Hu, Y. L.; Ge, J.; Jiang, H. L.; Yu, S. H. A facile and general coating approach to moisture/waterresistant metal–organic frameworks with intact porosity. J. Am. Chem. Soc. 2014, 136, 16978–16981.
D’Alessandro, D. M.; Smit, B.; Long, J. R. Carbon dioxide capture: Prospects for new materials. Angew. Chem., Int. Ed. 2010, 49, 6058–6082.
Li, J. R.; Yu, J. M.; Lu, W. G.; Sun, L. B.; Sculley, J.; Balbuena, P. B.; Zhou, H. C. Porous materials with predesigned single-molecule traps for CO2 selective adsorption. Nat. Commun. 2013, 4, 1538–1544.
Zhang, B.; Asakura, H.; Zhang, J.; Zhang, J. G.; De, S.; Yan, N. Stabilizing a platinum1 single-atom catalyst on supported phosphomolybdic acid without compromising hydrogenation activity. Angew. Chem., Int. Ed. 2016, 55, 8319–8323.
Liu, Y.; Xuan, W. M.; Cui, Y. Engineering homochiral metalorganic frameworks for heterogeneous asymmetric catalysis and enantioselective separation. Adv. Mater. 2010, 22, 4112–4135.
Sadakiyo, M.; Yamada, T.; Kitagawa, H. Rational designs for highly proton-conductive metal-organic frameworks. J. Am. Chem. Soc. 2009, 131, 9906–9907.
Rowsell, J. L. C.; Millward, A. R.; Park, K. S.; Yaghi, O. M. Hydrogen sorption in functionalized metal-organic frameworks. J. Am. Chem. Soc. 2004, 126, 5666–5667.
Xia, B. Y.; Yan, Y.; Li, N.; Wu, H. B.; Lou, X. W.; Wang, X. A metal-organic framework-derived bi-functional oxygen electrocatalyst. Nat. Energy 2016, 1, 15006.
Tang, J.; Salunkhe, R. R.; Liu, J.; Torad, N. L.; Imura, M.; Furukawa, S.; Yamauch, Y. Thermal conversion of core-shell metal-organic frameworks: A new method for selectively functionalized nanoporous hybrid carbon. J. Am. Chem. Soc. 2015, 137, 1572–1580.
He, L. C.; Liu, Y.; Liu, J. Z.; Xiong, Y. S.; Zheng, J. Z.; Liu, Y. L.; Tang, Z. Y. Core-shell noble-metal@metal-organicframework nanoparticles with highly selective sensing property. Angew. Chem., Int. Ed. 2013, 52, 3741–3745.
Zhou, J. J.; Wang, P.; Wang, C. X.; Goh, Y. T.; Fang, Z.; Messersmith, P. B.; Duan, H. W. Versatile core–shell nanoparticle@metal–organic framework nanohybrids: Exploiting mussel-inspired polydopamine for tailored structural integration. ACS Nano 2015, 9, 6951–6960.
Li, L. M.; Jiao, X. L.; Chen, D. R.; Lotsch, B. V.; Li, C. Facile fabrication of ultrathin metal-organic frameworkcoated monolayer colloidal crystals for highly efficient vapor sensing. Chem. Mater. 2015, 27, 7601–7609.
Cai, J. G.; Ye, J. F.; Chen, S. Y.; Zhao, X. W.; Zhang, D. Y.; Chen, S.; Ma, Y. R.; Jin, S.; Qi, L. M. Self-cleaning, broadband and quasi-omnidirectional antireflective structures based on mesocrystalline rutile TiO2 nanorod arrays. Energy Environ. Sci. 2012, 5, 7575–7581.
Kleinman, S. L.; Sharma, B.; Blaber, M. G.; Henry, A. I.; Valley, N.; Freeman, R. G.; Natan, M. J.; Schatz, G. C.; Van Duyne, R. P. Structure enhancement factor relationships in single gold nanoantennas by surface-enhanced raman excitation spectroscopy. J. Am. Chem. Soc. 2013, 135, 301–308.
Liu, B.; Wang, D. Y. High-throughput transformation of colloidal polymer spheres to discs simply via magnetic stirring of their dispersions. Langmuir 2012, 28, 6436–6440.
Liu, Y. D.; Goebl, J.; Yin, Y. D. Templated synthesis of nanostructured materials. Chem. Soc. Rev. 2013, 42, 2610–2653.
Li, Y.; Koshizaki, N.; Cai, W. P. Periodic one-dimensional nanostructured arrays based on colloidal templates, applications, and devices. Coord. Chem. Rev. 2011, 255, 357–373.
Li, Y.; Duan, G. T.; Liu, G. Q.; Cai, W. P. Physical processes-aided periodic micro/nanostructured arrays by colloidal template technique: Fabrication and applications. Chem. Soc. Rev. 2013, 42, 3614–3627.
Cong, H. L.; Yu, B.; Tang, J. G.; Li, Z. J.; Liu, X. S. Current status and future developments in preparation and application of colloidal crystals. Chem. Soc. Rev. 2013, 42, 7774–7800.
Zhu, D. F.; Huang, H.; Zhang, G.; Zhang, X.; Li, X.; Zhang, X. M.; Wang, T. Q.; Yang, B. Fabrication of heterogeneous double-ring-like structure arrays by combination of colloidal lithography and controllable dewetting. Langmuir 2012, 28, 2873–2880.
Hang, L. F.; Zhao, Y.; Zhang, H. H.; Liu, G. Q.; Cai, W. P.; Li, Y.; Qu, L. T. Copper nanoparticle@graphene composite arrays and their enhanced catalytic performance. Acta. Mater. 2016, 105, 59–67.
Mikrajuddin Iskandar, F.; Okuyanma, K. Single route for producing organized metallic domes, dots, and pores by colloidal templating and over-sputtering. Adv. Mater. 2002, 14, 930–933.
Férey, G.; Serre, C.; Mellot-Draznieks, C.; Millange, F.; Surblé, S.; Dutour, J.; Margiolaki, I. A hybrid solid with giant pores prepared by a combination of targeted chemistry, simulation, and powder diffraction. Angew. Chem., Int. Ed. 2004, 116, 6456–6461.
Férey, G.; Mellot-Draznieks, C.; Serre, C.; Millange, F.; Dutour, J.; Surblé, S.; Margiolaki, I. A chromium terephthalate-based solid with unusually large pore volumes and surface area. Science 2005, 309, 2040–2042.
Bhattachaijee, S.; Yang, D. A.; Ahn, W. S. A new heterogeneous catalyst for epoxidation of alkenesvia one-step postfunctionalization of IRMOF-3 with a manganese(II) acetylacetonate complex. Chem. Commun. 2011, 47, 3637–3639.
Wu, C. D.; Hu, A. G.; Zhang, L.; Lin, W. B. A homochiral porous metal-organic framework for highly enantioselective heterogeneous asymmetric catalysis. J. Am. Chem. Soc. 2005, 127, 8940–8941.
Dai, Z. F.; Li, Y.; Duan, G. T.; Jia, L. C.; Cai, W. P. Phase diagram, design of monolayer binary colloidal crystals, and their fabrication based on ethanol-assisted self-assembly at the air/water interface. ACS Nano 2012, 6, 6706–6716.
Zhang, H. H.; Liu, M.; Zhou, F.; Liu, D. L.; Liu, G. Q.; Duan, G. T.; Cai, W. P.; Li, Y. Physical deposition improved SERS stability of morphology controlled periodic micro/nanostructured arrays based on colloidal templates. Small 2015, 11, 844–853.
Zhang, C. J.; Losego, M. D.; Braun, P. V. Hydrogel-based glucose sensors: Effects of phenylboronic acid chemical structure on response. Chem. Mater. 2013, 25, 3239–3250.
Zhang, Y. J.; Guan, Y.; Zhou, S. Q. Synthesis and volume phase transitions of glucose-sensitive microgels. Biomacromolecules 2006, 7, 3196–3201.
Updike, S. J.; Hicks, G. P. The enzyme electrode. Nature 1967, 214, 986–988.
Shafer-Peltier, K. E.; Haynes, C. L.; Glucksberg, M. R.; Van Duyne, R. P. Toward a glucose biosensor based on surface-enhanced Raman scattering. J. Am. Chem. Soc. 2003, 125, 588–593.
Matsui, J.; Akamatsu, K.; Hara, N.; Miyoshi, D.; Nawafune, H.; Tamaki, K.; Sugimoto, N. SPR sensor chip for detection of small molecules using molecularly imprinted polymer with embedded gold nanoparticles. Anal. Chem. 2005, 77, 4282–4285.
ToghilI, K. E.; Compton, R. G. Electrochemical nonenzymatic glucose sensors: A perspective and an evaluation. Int. J. Electrochem. Sci. 2010, 5, 1246–1301.
Lu, G.; Li, S. Z.; Guo, Z.; Farha, O. K.; Hauser, B. G.; Qi, X. Y.; Wang, Y.; Wang, X.; Han, S. Y.; Liu, X. G. et al. Imparting functionality to a metal–organic framework material by controlled nanoparticle encapsulation. Nat. Chem. 2012, 4, 310–316.
Gu, Z. Z.; Horie, R.; Kubo, S.; Yamada, Y.; Fujishima, A.; Sato, O. Fabrication of a metal-coated three-dimensionally ordered macroporous film and its application as a refractive index sensor. Angew. Chem., Int. Ed. 2002, 41, 1153–1156.
Palik, E. D. Handbook of Optical Constants of Solids; Academic Press: New York, 1997.
Kanai, T.; Sawada, T.; Kitamura K. Optical determination of the lattice constants of colloidal crystals without use of the refractive index. Langmuir 2003, 19, 1984–1986.
Li, J. L.; Zheng, T. S. A comparison of chemical sensors based on the different ordered inverse opal films. Sensor. Actuat. B-Chem. 2008, 131, 190–195.
Lu, G.; Hupp, J. T. Metal-organic frameworks as sensors: A ZIF-8 based Fabry-Pérot device as a selective sensor for chemical vapors and gases. J. Am. Chem. Soc. 2010, 132, 7832–7833.
Acknowledgements
The authors acknowledge the financial support from the National Basic Research Program of China (No. 2012CB932303), the National Natural Science Foundation of China (Nos. 51371165 and 51571189), the State Key Program of National Natural Science Foundation of China (No. 51531006), the Anhui Provincial Natural Science Foundation (No. 1508085JGD07), the Cross-disciplinary Collaborative Teams Program in CAS, and the CAS/SAFEA International Partnership Program for Creative Research Teams.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
12274_2016_1414_MOESM1_ESM.pdf
Functionalized periodic Au@MOFs nanoparticle arrays as biosensors for dual-channel detection through the complementary effect of SPR and diffraction peaks
Rights and permissions
About this article
Cite this article
Hang, L., Zhou, F., Men, D. et al. Functionalized periodic Au@MOFs nanoparticle arrays as biosensors for dual-channel detection through the complementary effect of SPR and diffraction peaks. Nano Res. 10, 2257–2270 (2017). https://doi.org/10.1007/s12274-016-1414-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-016-1414-1