Abstract
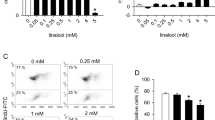
In the current study, macrolactin compounds, macrolactin A (MA) and 7-O-succinyl macrolactin A (SMA), were investigated for their anti-angiogenic activities and action mechanism. MA and SMA inhibited in vitro and in vivo angiogenesis induced by three different classes of pro-angiogenic factors, VEGF, IL-8, and TNF-α. SMA exhibited stronger anti-angiogenic activity than MA, and such anti-angiogenic activity of SMA was consistently observed in MDA-MB-231 human breast cancer cell-inoculated CAM assay showing dose-dependent suppression of tumor growth and tumor-induced angiogenesis. In an in vitro PI3K competitive activity assay, SMA induced concentration-dependent inhibition of class I PI3K isoforms, p110α, p110β, p110δ, and p110γ. In addition, non-receptor tyrosine kinase c-Src, which is involved in the activation of PI3K heterodimer, was suppressed by MA and SMA. Correspondingly, MA and SMA significantly inhibited the stimulus-induced phosphorylation of Akt, mTOR, p70S6K, and ribosomal S6 in human umbilical vein endothelial cells (HUVECs). At the same time, the stimulus-induced production of reactive oxygen species (ROS) and activation of NF-κB were significantly suppressed by MA and SMA. Moreover, the macrolactins suppressed NF-κB-regulated HSP90 protein expression, which stabilizes phosphorylated Akt and NADPH oxidase. Suppression of NF-κB in macrolactin-treated HUVECs with concurrent inhibition of rS6 indicates that MAs effectively block angiogenesis through down-regulation of genes related to angiogenesis at both transcriptional and translational levels. Taken together, the results demonstrate that anti-angiogenic effect of MA and SMA is mediated through inhibition of PI3K/Akt and NADPH oxidase-derived ROS/NF-κB signaling pathways. These results further indicate that MA and SMA may be applicable for treatment of various diseases associated with angiogenesis.





Similar content being viewed by others
References
Acosta, J.C., A. O’Loghlen, A. Banito, M.V. Guijarro, A. Augert, S. Raguz, M. Fumagalli, et al. 2008a. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133(6): 1006–1018.
Acosta, J.C., A. O’Loghlen, A. Banito, S. Raguz, and J. Gil. 2008b. Control of senescence by CXCR2 and its ligands. Cell Cycle 7(19): 2956–2959.
Akinleye, A., P. Avvaru, M. Furqan, Y. Song, and D. Liu. 2013. Phosphatidylinositol 3-kinase (PI3K) inhibitors as cancer therapeutics. Journal of Hematology and Oncology 6(1): 88.
Andreev, J., M.L. Galisteo, O. Kranenburg, S.K. Logan, E.S. Chiu, M. Okigaki, L.A. Cary, W.H. Moolenaar, and J. Schlessinger. 2001. Src and Pyk2 mediate G-protein-coupled receptor activation of epidermal growth factor receptor (EGFR) but are not required for coupling to the mitogen-activated protein (MAP) kinase signaling cascade. Journal of Biological Chemistry 276(23): 20130–20135.
Backer, J.M., M.G. Myers Jr, S.E. Shoelson, D.J. Chin, X.J. Sun, M. Miralpeix, P. Hu, et al. 1992. Phosphatidylinositol 3′-kinase is activated by association with IRS-1 during insulin stimulation. The EMBO Journal 11(9): 3469–3479.
Bader, A.G., S. Kang, L. Zhao, and P.K. Vogt. 2005. Oncogenic PI3K deregulates transcription and translation. Nature Reviews Cancer 5(12): 921–929.
Banning, A., N. Kurrle, M. Meister, and R. Tikkanen. 2014. Flotillins in receptor tyrosine kinase signaling and cancer. Cells 3(1): 129–149.
Barcelos, L.S., A. Talvani, A.S. Teixeira, L.Q. Vieira, G.D. Cassali, S.P. Andrade, and M.M. Teixeira. 2005. Impaired inflammatory angiogenesis, but not leukocyte influx, in mice lacking TNFR1. Journal of Leukocyte Bioliogy 78(2): 352–358.
Basuroy, S., S. Bhattacharya, C.W. Leffler, and H. Parfenova. 2009. Nox4 NADPH oxidase mediates oxidative stress and apoptosis caused by TNF-alpha in cerebral vascular endothelial cells. American Journal of Physiology-Cell Physiology 296(3): C422–C432.
Bhonde, M.R., R.D. Gupte, S.D. Dadarkar, M.G. Jadhav, A.A. Tannu, P. Bhatt, D.R. Bhatia, et al. 2008. A novel mTOR inhibitor is efficacious in a murine model of colitis. American Journal of Physiology-Gastrointestinal and Liver Physiology 295(6): G1237–G1245.
Carpenter, C.L., K.R. Auger, M. Chanudhuri, M. Yoakim, B. Schaffhausen, S. Shoelson, and L.C. Cantley. 1993. Phosphoinositide 3-kinase is activated by phosphopeptides that bind to the SH2 domains of the 85-kDa subunit. Journal of Biological Chemistry 268(13): 9478–9483.
Condliffe, A.M., K. Davidson, K.E. Anderson, C.D. Ellson, T. Crabbe, K. Okkenhaug, B. Vanhaesebroeck, et al. 2005. Sequential activation of class IB and class IA PI3K is important for the primed respiratory burst of human but not murine neutrophils. Blood 106(4): 1432–1440.
Ferrara, N. 2004. Vascular endothelial growth factor as a target for anticancer therapy. Oncologist 9(Suppl 1): 2–10.
Frey, R.S., X. Gao, K. Javaid, S.S. Siddiqui, A. Rahman, and A.B. Malik. 2006. Phosphatidylinositol 3-kinase gamma signaling through protein kinase Czeta induces NADPH oxidase-mediated oxidant generation and NF-kappaB activation in endothelial cells. Journal of Biological Chemistry 281(23): 16128–16138.
Fukui, Y., and H. Hanafusa. 1989. Phosphatidylinositol kinase activity associates with viral p60src protein. Molecular and Cell Biology 9(4): 1651–1658.
Gorlach, A., R.P. Brandes, K. Nguyen, M. Amidi, F. Dehghani, and R. Busse. 2000. A gp91phox containing NADPH oxidase selectively expressed in endothelial cells is a major source of oxygen radical generation in the arterial wall. Circulation Research 87(1): 26–32.
Graupera, M., J. Guillermet-Guibert, L.C. Foukas, L.K. Phng, R.J. Cain, A. Salpekar, W. Pearce, et al. 2008. Angiogenesis selectively requires the p110alpha isoform of PI3K to control endothelial cell migration. Nature 453(7195): 662–666.
Guillermet-Guibert, J., K. Bjorklof, A. Salpekar, C. Gonella, F. Ramadani, A. Bilancio, S. Meek, A.J. Smith, K. Okkenhaug, and B. Vanhaesebroeck. 2008. The p110beta isoform of phosphoinositide 3-kinase signals downstream of G protein-coupled receptors and is functionally redundant with p110gamma. Proceeding of the National Academy of Sciences of the United States of America 105(24): 8292–8297.
Gustafson, K., M. Roman, and W. Fenical. 1989. The macrolactins, a novel class of antiviral and cytotoxic macrolides from a deep-sea marine bacterium. Journal of the American Chemical Society 111(19): 7519–7524.
Heidemann, J., H. Ogawa, M.B. Dwinell, P. Rafiee, C. Maaser, H.R. Gockel, M.F. Otterson, et al. 2003. Angiogenic effects of interleukin 8 (CXCL8) in human intestinal microvascular endothelial cells are mediated by CXCR2. Journal of Biological Chemistry 278(10): 8508–8515.
Jiang, B.H., and L.Z. Liu. 2009. PI3K/PTEN signaling in angiogenesis and tumorigenesis. Advances in Cancer Research 102: 19–65.
Jung, Y.D., S.A. Ahmad, W. Liu, N. Reinmuth, A. Parikh, O. Stoeltzing, F. Fan, and L.M. Ellis. 2002. The role of the microenvironment and intercellular cross-talk in tumor angiogenesis. Seminars in Cancer Biology 12(2): 105–112.
Kanoh, S., and B.K. Rubin. 2010. Mechanisms of action and clinical application of macrolides as immunomodulatory medications. Clinical Microbiology Review 23(3): 590–615.
Kapeller, R., and L.C. Cantley. 1994. Phosphatidylinositol 3-kinase. BioEssays 16(8): 565–576.
Karar, J., and A. Maity. 2011. PI3K/AKT/mTOR pathway in angiogenesis. Frontiers in Molecular Neuroscience 4: 51.
Keffer, J., L. Probert, H. Cazlaris, S. Georgopoulos, E. Kaslaris, D. Kioussis, and G. Kollias. 1991. Transgenic mice expressing human tumour necrosis factor: A predictive genetic model of arthritis. The EMBO Journal 10(13): 4025–4031.
Kerbel, R.S., and B.A. Kamen. 2004. The anti-angiogenic basis of metronomic chemotherapy. Nature Reviews Cancer 4(6): 423–436.
Laing, K.J., and C.J. Secombes. 2004. Chemokines. Developmental and Comparative Immunology 28(5): 443–460.
Li, A., S. Dubey, M.L. Varney, B.J. Dave, and R.K. Singh. 2003. IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis. The Journal of Immunology 170(6): 3369–3376.
Li, J.M., L.M. Fan, M.R. Christie, and A.M. Shah. 2005. Acute tumor necrosis factor alpha signaling via NADPH oxidase in microvascular endothelial cells: role of p47phox phosphorylation and binding to TRAF4. Molecular and Cell Biology 25(6): 2320–2330.
Li, J.M., and A.M. Shah. 2004. Endothelial cell superoxide generation: regulation and relevance for cardiovascular pathophysiology. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 287(5): R1014–R1030.
Madrigal-Matute, J., C.E. Fernandez-Garcia, C. Gomez-Guerrero, O. Lopez-Franco, B. Munoz-Garcia, J. Egido, L.M. Blanco-Colio, and J.L. Martin-Ventura. 2012. HSP90 inhibition by 17-DMAG attenuates oxidative stress in experimental atherosclerosis. Cardiovascular Research 95(1): 116–123.
Ono, M. 2008. Molecular links between tumor angiogenesis and inflammation: inflammatory stimuli of macrophages and cancer cells as targets for therapeutic strategy. Cancer Science 99(8): 1501–1506.
Park, S., S.C. Regmi, S.Y. Park, E.K. Lee, J.H. Chang, S.K. Ku, D.H. Kim, and J.A. Kim. 2014. Protective effect of 7-O-succinyl macrolactin A against intestinal inflammation is mediated through PI3-kinase/Akt/mTOR and NF-kappaB signaling pathways. European Journal of Pharmacology 735: 184–192.
Pincheira, R., A.F. Castro, O.N. Ozes, P.S. Idumalla, and D.B. Donner. 2008. Type 1 TNF receptor forms a complex with and uses Jak2 and c-Src to selectively engage signaling pathways that regulate transcription factor activity. Journal of Immunology 181(2): 1288–1298.
Puri, K.D., T.A. Doggett, J. Douangpanya, Y. Hou, W.T. Tino, T. Wilson, T. Graf, et al. 2004. Mechanisms and implications of phosphoinositide 3-kinase delta in promoting neutrophil trafficking into inflamed tissue. Blood 103(9): 3448–3456.
Puri, K.D., T.A. Doggett, C.Y. Huang, J. Douangpanya, J.S. Hayflick, M. Turner, J. Penninger, and T.G. Diacovo. 2005. The role of endothelial PI3Kgamma activity in neutrophil trafficking. Blood 106(1): 150–157.
Ribatti, D., B. Nico, A.S. Belloni, A. Vacca, L. Roncali, and G.G. Nussdorfer. 2001. Angiogenic activity of leptin in the chick embryo chorioallantoic membrane is in part mediated by endogenous fibroblast growth factor-2. International Journal of Molecular Medicine 8(3): 265–268.
Rordorf-Nikolic, T., D.J. Van Horn, D. Chen, M.F. White, and J.M. Backer. 1995. Regulation of phosphatidylinositol 3′-kinase by tyrosyl phosphoproteins. Full activation requires occupancy of both SH2 domains in the 85-kDa regulatory subunit. Journal of Biological Chemistry 270(8): 3662–3666.
Ruderman, N.B., R. Kapeller, M.F. White, and L.C. Cantley. 1990. Activation of phosphatidylinositol 3-kinase by insulin. Proceeding of the National Academy of Sciences of the United States of America 87(4): 1411–1415.
Sarma, V., F.W. Wolf, R.M. Marks, T.B. Shows, and V.M. Dixit. 1992. Cloning of a novel tumor necrosis factor-alpha-inducible primary response gene that is differentially expressed in development and capillary tube-like formation in vitro. Journal of Immunology 148(10): 3302–3312.
Sato, S., N. Fujita, and T. Tsuruo. 2000. Modulation of Akt kinase activity by binding to Hsp90. Proceeding of the National Academy of Sciences of the United States of America 97(20): 10832–10837.
Serban, D., J. Leng, and D. Cheresh. 2008. H-ras regulates angiogenesis and vascular permeability by activation of distinct downstream effectors. Circulation Research 102(11): 1350–1358.
Shiojima, I., and K. Walsh. 2002. Role of Akt signaling in vascular homeostasis and angiogenesis. Circulation Research 90(12): 1243–1250.
Stommel, J.M., A.C. Kimmelman, H. Ying, R. Nabioullin, A.H. Ponugoti, R. Wiedemeyer, A.H. Stegh, et al. 2007. Coactivation of receptor tyrosine kinases affects the response of tumor cells to targeted therapies. Science 318(5848): 287–290.
Thomas, S.M., and J.S. Brugge. 1997. Cellular functions regulated by Src family kinases. Annual Review of Cell and Developmental Biology 13: 513–609.
Tojo, T., M. Ushio-Fukai, M. Yamaoka-Tojo, S. Ikeda, N. Patrushev, and R.W. Alexander. 2005. Role of gp91phox (Nox2)-containing NAD(P)H oxidase in angiogenesis in response to hindlimb ischemia. Circulation 111(18): 2347–2355.
Tufan, A.C., and N.L. Satiroglu-Tufan. 2005. The chick embryo chorioallantoic membrane as a model system for the study of tumor angiogenesis, invasion and development of anti-angiogenic agents. Current Cancer Drug Targets 5(4): 249–266.
Ushio-Fukai, M., Y. Tang, T. Fukai, S.I. Dikalov, Y. Ma, M. Fujimoto, M.T. Quinn, P.J. Pagano, C. Johnson, and R.W. Alexander. 2002. Novel role of gp91(phox)-containing NAD(P)H oxidase in vascular endothelial growth factor-induced signaling and angiogenesis. Circulation Research 91(12): 1160–1167.
Wilson, J., and F. Balkwill. 2002. The role of cytokines in the epithelial cancer microenvironment. Seminars in Cancer Biology 12(2): 113–120.
Xie, K. 2001. Interleukin-8 and human cancer biology. Cytokine & Growth Factor Reviews 12(4): 375–391.
Yatsunami, J., Y. Fukuno, M. Nagata, M. Tominaga, S. Aoki, N. Tsuruta, M. Kawashima, S. Taniguchi, and S. Hayashi. 1999. Antiangiogenic and antitumor effects of 14-membered ring macrolides on mouse B16 melanoma cells. Clinical and Experimental Metastasis 17(4): 361–367.
Yatsunami, J., N. Tsuruta, N. Hara, and S. Hayashi. 1998. Inhibition of tumor angiogenesis by roxithromycin, a 14-membered ring macrolide antibiotic. Cancer Letters 131(2): 137–143.
Yoshida, S., M. Ono, T. Shono, H. Izumi, T. Ishibashi, H. Suzuki, and M. Kuwano. 1997. Involvement of interleukin-8, vascular endothelial growth factor, and basic fibroblast growth factor in tumor necrosis factor alpha-dependent angiogenesis. Molecular and Cell Biology 17(7): 4015–4023.
Yoshimoto, T., N. Gochou, N. Fukai, T. Sugiyama, M. Shichiri, and Y. Hirata. 2005. Adrenomedullin inhibits angiotensin II-induced oxidative stress and gene expression in rat endothelial cells. Hypertension Research 28(2): 165–172.
Zachary, I., and G. Gliki. 2001. Signaling transduction mechanisms mediating biological actions of the vascular endothelial growth factor family. Cardiovasular Research 49(3): 568–581.
Zhang, R., Y. Xu, N. Ekman, Z. Wu, J. Wu, K. Alitalo, and W. Min. 2003. Etk/Bmx transactivates vascular endothelial growth factor 2 and recruits phosphatidylinositol 3-kinase to mediate the tumor necrosis factor-induced angiogenic pathway. Journal of Biological Chemistry 278(51): 51267–51276.
Zhao, M., A. Wimmer, K. Trieu, R.G. Discipio, and I.U. Schraufstatter. 2004. Arrestin regulates MAPK activation and prevents NADPH oxidase-dependent death of cells expressing CXCR2. Journal of Biological Chemistry 279(47): 49259–49267.
Acknowledgments
This work was supported by the 2010 Yeungnam University Research Grant.
Conflict of interest
There is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, Y., Regmi, S.C., Kim, M.Y. et al. Anti-angiogenic activity of macrolactin A and its succinyl derivative is mediated through inhibition of class I PI3K activity and its signaling. Arch. Pharm. Res. 38, 249–260 (2015). https://doi.org/10.1007/s12272-014-0535-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-014-0535-x