Abstract
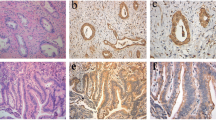
Numerous data suggest that altered expression of tight junction proteins such as occludin and claudins plays important role in carcinogenesis. However, little is known about tricellulin, a transmembrane tight junction protein concentrated where three epithelial cells meet. We aimed to characterize tricellulin expression in normal and cirrhotic liver in comparison to primary hepatic neoplasms. Tricellulin expression of 20 control livers, 12 cirrhotic livers, 32 hepatocellular carcinomas (HCC), and 20 intrahepatic cholangiocarcinomas (iCCC) was investigated by immunohistochemistry and Western blotting. Co-localization of tricellulin with claudin-1, -4, and MRP2 was studied using double immunofluorescence. Scattered tricellulin immunopositivity was restricted to biliary pole of hepatocytes confirmed by co-localization with MRP2. Moreover, spotted-like reaction was observed between bile duct epithelial cells. In 40 % of HCCs marked tricellulin overexpression was measured regardless of tumor grades. In iCCCs, however, tricellulin expression decreased parallel with dedifferentiation. In HCCs high tricellulin expression, in iCCCs low tricellulin expression correlated with poor prognosis. Co-localization with MRP2 might substantiate that tricellulin plays role in blood-biliary barrier. Overexpressed tricellulin in a subset of HCCs correlated with unfavorable prognosis. Similar to ductal pancreatic adenocarcinoma, higher grades of iCCCs were associated with decreased tricellulin expression correlating with poor prognosis.








Similar content being viewed by others
References
Zigler M, Dobroff AS, Bar-Eli M (2010) Cell adhesion: implication in tumor progression. Minerva Med 101:149–162
Turksen K, Troy TC (2011) Junctions gone bad: claudins and loss of the barrier in cancer. Biochim Biophys Acta 1816:73–79
Martin TA, Mason MD, Jiang WG (2011) Tight junctions in cancer metastasis. Front Biosci 16:898–936
Ikenouchi J, Furuse M, Furuse K et al (2005) Tricellulin constitutes a novel barrier at tricellular contacts of epithelial cells. J Cell Biol 171:939–945
Chiba H, Osanai M, Murata M et al (2008) Transmembrane proteins of tight junctions. Biochim Biophys Acta 1778:588–600
Riazuddin S, Ahmed ZM, Fanning AS et al (2006) Tricellulin is a tight-junction protein necessary for hearing. Am J Hum Genet 79:1040–1051
Chishti MS, Bhatti A, Tamim S et al (2008) Splice-site mutations in the TRIC gene underlie autosomal recessive nonsyndromic hearing impairment in Pakistani families. J Hum Genet 53:101–105
Kondoh A, Takano K, Kojima T et al (2011) Altered expression of claudin-1, claudin-7, and tricellulin regardless of human papilloma virus infection in human tonsillar squamous cell carcinoma. Acta Otolaryngol 131:861–868
Korompay A, Borka K, Lotz G et al (2012) Tricellulin expression in normal and neoplastic human pancreas. Histopathology 60(6B):E76–E78
Patonai A, Erdelyi-Belle B, Korompay A et al (2011) Claudins and tricellulin in fibrolamellar hepatocellular carcinoma. Virchows Arch 458:679–688
Parkin DM, Bray F, Ferlay J et al (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Hammill CW, Wong LL (2008) Intrahepatic cholangiocarcinoma: a malignancy of increasing importance. J Am Coll Surg 207:594–603
Nemeth Z, Szasz AM, Tatrai P et al (2009) Claudin-1, -2, -3, -4, -7, -8, and -10 protein expression in biliary tract cancers. J Histochem Cytochem 57:113–121
Lodi C, Szabo E, Holczbauer A et al (2006) Claudin-4 differentiates biliary tract cancers from hepatocellular carcinomas. Mod Pathol 19:460–469
Liu S, Yang W, Shen L et al (2009) Tight junction proteins claudin-1 and occludin control hepatitis C virus entry and are downregulated during infection to prevent superinfection. J Virol 83:2011–2014
Evans MJ, von Hahn T, Tscherne DM et al (2007) Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry. Nature 446:801–805
Sakaguchi T, Suzuki S, Higashi H et al (2008) Expression of tight junction protein claudin-5 in tumor vessels and sinusoidal endothelium in patients with hepatocellular carcinoma. J Surg Res 147:123–131
Higashi Y, Suzuki S, Sakaguchi T et al (2007) Loss of claudin-1 expression correlates with malignancy of hepatocellular carcinoma. J Surg Res 139:68–76
Huang GW, Ding X, Chen SL et al (2011) Expression of claudin 10 protein in hepatocellular carcinoma: impact on survival. J Cancer Res Clin Oncol 137:1213–1218
Cheung ST, Leung KL, Ip YC et al (2005) Claudin-10 expression level is associated with recurrence of primary hepatocellular carcinoma. Clin Cancer Res 11:551–556
Shinozaki A, Shibahara J, Noda N et al (2011) Claudin-18 in biliary neoplasms. Its significance in the classification of intrahepatic cholangiocarcinoma. Virchows Arch 459:73–80
Kojima T, Yamamoto T, Murata M et al (2003) Regulation of the blood-biliary barrier: interaction between gap and tight junctions in hepatocytes. Med Electron Microsc 36:157–164
Meertens L, Bertaux C, Cukierman L et al (2008) The tight junction proteins claudin-1, -6, and -9 are entry cofactors for hepatitis C virus. J Virol 82:3555–3560
Zheng A, Yuan F, Li Y et al (2007) Claudin-6 and claudin-9 function as additional coreceptors for hepatitis C virus. J Virol 81:12465–12471
French AD, Fiori JL, Camilli TC et al (2009) PKC and PKA phosphorylation affect the subcellular localization of claudin-1 in melanoma cells. Int J Med Sci 6:93–101
Terry S, Nie M, Matter K et al (2010) Rho signaling and tight junction functions. Physiology (Bethesda) 25:16–26
Gonzalez-Mariscal L, Lechuga S, Garay E (2007) Role of tight junctions in cell proliferation and cancer. Prog Histochem Cytochem 42:1–57
Lal-Nag M, Morin PJ (2009) The claudins. Genome Biol 10:235
de Oliveira SS, de Oliveira IM, De Souza W et al (2005) Claudins upregulation in human colorectal cancer. FEBS Lett 579:6179–6185
Yoshida T, Kinugasa T, Akagi Y et al (2011) Decreased expression of claudin-1 in rectal cancer: a factor for recurrence and poor prognosis. Anticancer Res 31:2517–2525
Shin HI, Kim BH, Chang HS et al (2011) Expression of claudin-1 and -7 in clear cell renal cell carcinoma and its clinical significance. Korean J Urol 52:317–322
Acknowledgments
This work was supported by grants from the Hungarian Scientific Research Found (OTKA)# K101435.
Disclosure Statement
The authors have no financial or personal conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Somorácz, Á., Korompay, A., Törzsök, P. et al. Tricellulin Expression and its Prognostic Significance in Primary Liver Carcinomas. Pathol. Oncol. Res. 20, 755–764 (2014). https://doi.org/10.1007/s12253-014-9758-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9758-x