Abstract
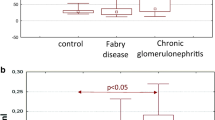
We evaluated the heat shock system 70 (HSP70) in patients with chronic glomerulonephritis (CGN). Seventy-six patients with CGN patients were included in our study. Ten patients with mild proteinuria (median 0.48 [0.16–0.78] g/24 h) and ten healthy subjects served as positive and negative controls, respectively. Urinary levels of HSP70, interleukin-10, and serum levels of anti-HSP70 were measured by ELISA. The immunohistochemical peroxidase method was used to study the expression of HSP70 and Foxp3+ in kidney biopsies. TregFoxP3+ cells in the interstitium were determined morphometrically. Median urinary HSP70 levels in patients with nephrotic syndrome (NS) [6.57 (4.49–8.33) pg/mg] and subnephrotic range proteinuria [5.7 (4.12–6.9) pg/mg] were higher (p < 0.05) than in positive [3.7 (2.5–4.82) pg/mg] and negative [3.78 (2.89–4.84) pg/mg] controls. HSP70 expression index in tubular cells positively correlated with urinary HSP70 (Rs = 0.948, р < 0.05) and proteinuria (Rs = 0.362, p < 0.05). The number of TregFoxp3+ cells in the kidney interstitium and interleukin-10 excretion were lower in patients with NS. Anti-HSP70 antibody serum levels in patients with NS [21.1 (17.47–29.72) pg/ml] and subnephrotic range proteinuria [24.9 (18.86–30.92) pg/ml] were significantly higher than in positive [17.8 (12.95–23.03) pg/ml] and negative [18.9 (13.5–23.9) pg/ml] controls. In patients with CGN, increasing proteinuria was associated with higher HSP70 renal tissue and urinary levels. However, activation of HSP70 in patients with nephrotic syndrome did not lead to an increase in tissue levels of TregFoxp3+ cells or to the release of IL-10.



Similar content being viewed by others
Abbreviations
- HSPs:
-
Heat shock proteins
- CGN:
-
Chronic glomerulonephritis
- Tregs:
-
Regulatory T cells
- Foxp3:
-
Transcription factor Forkhead box P3
- IL-10:
-
IL-1, IL-6 - interleukin -10, -1, and -6
- CKD-EPI:
-
Chronic Kidney Disease Epidemiology Collaboration
- eGFR:
-
Estimated glomerular filtration rate
- ELISA:
-
Enzyme-linked immunosorbent assay
- MCD:
-
Minimal change disease
- FSGS:
-
Focal segmental glomerular sclerosis
- MN:
-
Membranous nephropathy
- MPGN:
-
Membranoproliferative glomerulonephritis
- CMV:
-
Cytomegalovirus
References
Asadullah K, Sterry W, Volk HD (2010) Interleukin-10 therapy – review of a new approach. Pharmacol Rev 55:241–269. https://doi.org/10.1124/pr.55.2.4
Beck F-X, Neuhofer W, Muller E (2000) Molecular chaperones in the kidney: distribution, putative roles and regulation. Am J Physiol Ren Physiol 279:203–215
Birnbaum G, Kotilinek L, Miller SD, Raine CS, Gao YL, Lehmann PV et al (1998) Heat shock proteins and experimental autoimmune encephalomyelitis. II: environmental infection and extra-neuraxial inflammation after the course of chronic relapsing encephalomyelitis. J Neuroimmunol 90:149–161. https://doi.org/10.1016/S0165-5728(98)00141-6
de Waal Malefyt R, Abrams J, Bennett B, Figdor CG, de Vries JE (1991) Interleukin 10(IL-10) inhibits cytokine synthesis by human monocytes: an autoregulatory role of IL-10 produced by monocytes. J Exp Med 174(5):1209–1220
Dhillon V, Latchman D, Isenberg D (1991) Review: heat shock proteins and systemic lupus erythematosus. Lupus 1:3–8. https://doi.org/10.1177/096120339100100102
Dodd SM, Martin JE, Swash M, Mather K (1993) Expression of heat shock protein epitopes in renal disease. Clin Nephrol 39(5):239–244
Dokka S, Shi X, Leonard S, Wang L, Castranova V, Rojanasakul Y (2001) Interleukin-10-mediated inhibition of free radical generation in macrophages. Am J Phys Lung Cell Mol Phys 280(6):1196–1202
Fontenot JD, Gavin MA, Rudensky AY (2003) Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol 4:330–336. https://doi.org/10.1038/ni904
Georgopoulos C, McFarland H (1993) Heat shock protein in multiple sclerosis and other autoimmune diseases. Immunol Today 14:373–375. https://doi.org/10.1016/0167-5699(93)90135-8
Guo Q, Du X, Zhao Y, Zhang D, Yue L, Wang Z (2014) Ischemic postconditioning prevents renal ischemia reperfusion injury through the induction of heat shock proteins in rats. Mol Med Rep 10:2875–2881. https://doi.org/10.3892/mmr.2014.2641
Harrison EM, Sharpe E, Bellamy CO, McNally SJ, Devey L, Garden OJ et al (2008) Heat shock protein 90-binding agents protect renal cells from oxidative stress and reduce kidney ischemia reperfusion injury. Am J Physiol Ren Physiol 295:397–405. https://doi.org/10.1152/ajprenal.00361.2007
Hori S, Nomura T, Sakaguchi S (2003) Control of regulatory T cell development by the transcription factor Foxp3. Science 299:1057–1061. https://doi.org/10.1126/science.1079490
Kampinga HH, Craig EA (2010) The HSP70 chaperone machinery: J proteins as drivers of functional specificity. Nat Rev Mol Cell Biol 11:579–592. https://doi.org/10.1038/nrm2941
Kim M-G, Cho EG, Lee JW, Ko YS, Lee HY, Jo S-K et al (2014) The heat-shock protein-70-induced renoprotective effect is partially mediated by CD4+ CD25+ Foxp3 + regulatory T cells in ischemia/reperfusion-induced acute kidney injury. Kidney Int 85(1):62–71. https://doi.org/10.1038/ki.2013.277
Kitamura M, Fine LG (1999) The concept of glomerular self-defense. Kidney Int 55:1639–1671. https://doi.org/10.1046/j.1523-1755.1999.00425.x
Komatsuda A, Wakui H, Imai H, Nakamoto Y, Miura AB, Itoh H (1992) Renal localization of the constitutive 73-kDa heat shock protein in normal and PAN rats. Kidney Int 41:1204–1212. https://doi.org/10.1038/ki.1992.182
Multhoff G, Hightower LE (1996) Cell surface expression of heat shock proteins and the immune response. Cell Stress Chaperones 1:167–176
O’Neill S, Ingman TG, Wigmore SJ, Harrison EM, Bellamy CO (2013) Differential expression of heat shock proteins in healthy and diseased human renal allografts. Ann Transplant 18:550–557. https://doi.org/10.12659/AOT.889599
Pockley AG (2003) Heat shock proteins as regulators of the immune response. Lancet 362:469–476. https://doi.org/10.1016/S0140-6736(03)14075-5
van Eden W, Tholet JER, van der Zee R, Noordzij A, van Embden JDA, Hensen EJ et al (1988) Cloning of the mycobacterial epitope recognized by T lymphocyte in adjuvant arthritis. Nature 331:171–173. https://doi.org/10.1038/331171a0
Venkataseshan V, Marque S (1996) Heat shock protein 72/73 in normal and diseased kidneys. Nephron 73(3):442–449
Wang Z, Gall JM, Bonegio RG, Havasi A, Hunt CR, Sherman MY et al (2011) Induction of heat-shock protein 70 inhibits ischemic renal injury. Kidney Int 79:861–870. https://doi.org/10.1038/ki.2010.527
Wu T, Tanguay RM (2006) Antibodies against heat shock proteins in environmental stresses and diseases: friend or foe? Cell Stress Chaperones 11:1–12. https://doi.org/10.1379/CSC-155R.1
Acknowledgements
The authors thank Professor Vladimir Varshavsky for morphological analysis. The authors also thank all patients and site staff who participated in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the Ethics Committee of the Sechenov University. All subjects provided informed written consent before participation in the study that conformed with the Declaration of Helsinki.
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
ESM 1
(DOCX 2832 kb)
Rights and permissions
About this article
Cite this article
Chebotareva, N., Bobkova, I., Lysenko, L. et al. Heat shock protein 70 and anti-heat shock protein 70 antibodies in patients with chronic glomerulonephritis. Cell Stress and Chaperones 23, 1229–1235 (2018). https://doi.org/10.1007/s12192-018-0928-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-018-0928-8



