Abstract
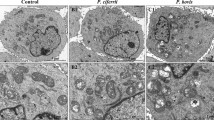
Brucella, which is regarded as an intracellular pathogen responsible for a zoonotic disease called brucellosis, survives and proliferates within several types of phagocytic and non-phagocytic cells. Brucella infects not only their preferred hosts but also other domestic and wild animal species, inducing abortion and infertility. Therefore, the interaction between uterine cells and Brucella is important for understanding the pathogenesis of this disease. In this study, we describe the Brucella suis vaccine strain S2 (B.suis.S2) infection and replication in the immortalized caprine endometrial epithelial cell line hTERT-EECs and the induced cellular and molecular response modulation in vitro. We found that B.suis S2 was able to infect and replicate to high titers and inhibit the proliferation of EECs and induce non-apoptotic pathways, as determined by B.suis.S2 detection using MTT and acridine orange/ethidium bromide (AO/EB) staining and flow cytometry. We explored the evidence of non-apoptotic pathways using real-time quantitative RT-PCR and by western blot analysis. Finally, we discovered the over-expression of GRP78, ATF4, ATF6, PERK, eIF2α, CHOP, and cytochrome c (Cyt-c) but not IRE1, xbp-1, and caspase-3 in B.suis.S2 (HK)-attacked and B.suis.S2-infected cells, suggesting that the molecular mechanism of ER stress sensor activation by B.suis.S2 is basically concomitant with that by B.suis.S2 (HK) and that ER stress, especially the PERK pathway, plays an important role in the process of B.suis.S2 infecting EEC, which may, in part, explain the role of the uterus in the pathogenesis of B.suis.S2.






Similar content being viewed by others
Abbreviations
- T4SS:
-
Type IV secretion system
- ER:
-
Endoplasmic reticulum
- EEC:
-
Caprine endometrial epithelial cell
- B.suis.S2 :
-
Brucella suis vaccine strain S2
- PERK:
-
Protein kinase RNA-like kinase
- IRE1α:
-
Inositol-requiring enzyme 1α
- ATF6:
-
Activating transcription factor 6
- GRP78:
-
78-kDa glucose-regulated protein
- CHOP:
-
C/EBP homologus protein
- CFU:
-
Colony-forming units
- Tm:
-
Tunicamycin
- xbp-1:
-
X-box binding protein 1
- MTT-3-[4:
-
5-Dimethylthiazol-2-yl]-2
- 5:
-
Diphenyltetrazolium bromide assay
- eIF2α:
-
α Subunit of eukaryotic initiation factor 2
References
Baud D, Greub G (2011) Intracellular bacteria and adverse pregnancy outcomes. Clin Microbiol Infect 17:1312–1322
Celli J, de Chastellier C, Franchini DM, Pizarro-Cerda J et al (2003) Brucella evades macrophage killing via VirB-dependent sustained interactions with the endoplasmic reticulum. J Exp Med 198:545–556
de Jong MF, Starr T, Winter MG, den Hartigh AB et al (2013) Sensing of bacterial Type IV secretion via the unfolded protein response. Mbio 4(1):e00418-12
Díaz Aparicio E (2013) Epidemiology of brucellosis in domestic animals caused by Brucella melitensis, Brucella suis and Brucella abortus. Rev Sci Tech 32(1):43–51, 53-60
Endo M, Oyadomari S, Suga S, Mori M et al (2005) The ER stress pathway involving CHOP is activated in the lungs of LPS-treated mice. J Biochem 138:501–507
Endo M, Mori M, Akira S, Gotoh T (2006) C/EBP homologous protein (CHOP) is crucial for the induction of caspase-11 and the pathogenesis of lipopolysaccharide-induced inflammation. J Immunol 176:6245–6253
Gotoh T, Oyadomari S, Mori K, Mori M (2002) Nitric oxide-induced apoptosis in RAW 264.7 macrophages is mediated by endoplasmic reticulum stress pathway involving ATF6 and CHOP. J Biol Chem 277:12343–12350
Gross A, Terraza A, Ouahrani-Bettache S, Liautard JP et al (2000) In vitro Brucella suis infection prevents the programmed cell death of human monocytic cells. Infect Immun 68(1):342–351
Harding HP, Zhang Y, Bertolotti A, Zeng H et al (2000) Perk is essential for translational regulation and cell survival during the unfolded protein response. Mol Cell 5:897–904
Li J, Lee B, Lee AS (2006) Endoplasmic reticulum stress-induced apoptosis. J Biol Chem 281:7260–7270
Ogata M, Hino S, Saito A, Morikawa K et al (2006) Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol Cell Biol 26:9220–9231
Oyadomari S, Mori M (2004) Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ 11:381–389
Pei J, Turse JE, Wu Q, Ficht TA (2006) Brucella abortus rough mutants induce macrophage oncosis that requires bacterial protein synthesis and direct interaction with the macrophage. Infect Immun 74:2667–2675
Posadas DM, Ruiz‐Ranwez V, Bonomi HR, Martín FA et al (2012) BmaC, a novel autotransporter of Brucella suis, is involved in bacterial adhesion to host cells. Cell Microbiol 14(6):965–982
Qin QM, Pei J, Ancona V, Shaw BD et al (2008) RNAi screen of endoplasmic reticulum-associated host factors reveals a role for IRE1a in supporting Brucella replication. PLoS Pathog 4(7):e1000110
Qiu M, Quan F, Han C, Wu B et al (2013) Effects of granulosa cells on steroidogenesis, proliferation and apoptosis of stromal cells and theca cells derived from the goat ovary. J Steroid Biochem Mol Biol 138:325–333
Roop RM II, Gaines JM, Anderson ES, Caswell CC et al (2009) Survival of the fittest: how Brucella strains adapt to their intracellular niche in the host. Med Microbiol Immunol 198(4):221–238
Salcedo SP, Chevrier N, Lacerda TLS, Harms JS et al (2013) Pathogenic brucellae replicate in human trophoblasts. J Infect Dis 207(7):1075–1083
Sano R, Reed JC (2013) ER stress-induced cell death mechanisms. Biochim Biophys Acta 1833(12):3460–3470
Santi-Rocca J, Smith S, Weber C, Pineda E et al (2012) Endoplasmic reticulum stress-sensing mechanism is activated in Entamoeba histolytica upon treatment with nitric oxide. PLoS One 7(2):e31777
Scholz HC, Nöckler K, Göllner C, Bahn P et al (2010) Brucella inopinata sp. nov., isolated from a breast implant infection. Int J Syst Evol Microbiol 60:801–808
Schro¨der M, Kaufman RJ (2005) ER stress and the unfolded protein response. Mutat Res 569:29–63
Schröder M, Kaufman RJ (2005) The mammalian unfolded protein response. Annu Rev Biochem 74:739–789
Shang DQ, Xiao DL, Yin JM (2002) Epidemiology and control of brucellosis in China. Vet Microbiol 90:165–182
Silva APC, Costa EA, Macêdo AA, Martins TDM et al (2012) Transcription of pattern recognition receptors and abortive agents induced chemokines in the bovine pregnant uterus. Vet Immunol immunopathol 145(1):248–256
Smith JA, Khan M, Magnani DD, Harms JS et al (2013) Brucella induces an unfolded protein response via TcpB that supports intracellular replication in macrophages. PLoS Pathog 9(12):e1003785
Yoichiro N, Motoyoshi E, Hiroto T, Masataka M et al (2010) Molecular mechanisms of the LPS-induced non-apoptotic ER stress-CHOP pathway. J Biochem 147(4):471–483
Zhang YY, Wang AH, Wu QX, Sheng HX et al (2010) Establishment and characteristics of immortal goat endometrial epithelial cells and stromal cells with hTERT. J Anim Vet Adv 9:2738–2747
Acknowledgments
We thank the Shaanxi Provincial Institute for Veterinary Drug Control for vaccine strains.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Lin, P., Yin, Y. et al. Brucella suis vaccine strain S2-infected immortalized caprine endometrial epithelial cell lines induce non-apoptotic ER-stress. Cell Stress and Chaperones 20, 399–409 (2015). https://doi.org/10.1007/s12192-014-0564-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-014-0564-x