Abstract
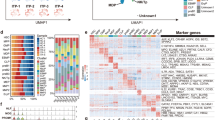
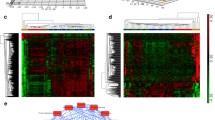
Anagrelide is a treatment option for patients with essential thrombocythemia. Although the clinical efficacy of anagrelide has been established, there is limited knowledge of the molecular mechanism underlying its effect. Here, we evaluated the effect of anagrelide on primary megakaryocytic progenitors from cord blood-derived CD34-positive cells. Anagrelide treatment reduced the expression of megakaryocytic markers (CD41 and CD61). Microarray analysis was performed to characterize gene profiles altered by exposure to anagrelide. The analysis demonstrated upregulation and downregulation (>2-fold) of eight and 34 genes, respectively, in anagrelide-treated megakaryocyte progenitors. This included genes encoding prototypical megakaryocytic proteins, such as PPBP, PF4, and GP6. Gene ontology analysis of genes suppressed by anagrelide treatment revealed significant enrichment of genes involved in platelet activation and degranulation. Expression levels of transcription factors involved in megakaryocyte commitment/differentiation were further evaluated by quantitative RT-PCR, demonstrating significant downregulation of FLI1 and TAL1 in anagrelide-treated megakaryocyte progenitors. Knockdown of TAL1 in primary megakaryocyte progenitors confirmed significant downregulation of FLI1 and megakaryocytic genes. Anagrelide had no significant effect on the surface expression of erythroid markers or on the expression of transcription factors involved in erythroid commitment/differentiation. In conclusion, anagrelide suppresses megakaryocytic differentiation, partly through decreasing the expression of megakaryocytic transcription factors.





Similar content being viewed by others
References
Passamonti F, Rumi E, Arcaini L, Boveri E, Elena C, Pietra D, et al. Prognostic factors for thrombosis, myelofibrosis, and leukemia in essential thrombocythemia: a study of 605 patients. Haematologica. 2008;93:1645–51.
Palandri F, Catani L, Testoni N, Ottaviani E, Polverelli N, Fiacchini M, et al. Long-term follow-up of 386 consecutive patients with essential thrombocythemia: safety of cytoreductive therapy. Am J Hematol. 2009;84:215–20.
Harrison CN, Campbell PJ, Buck G, Wheatley K, East CL, Bareford D, et al. Hydroxyurea compared with anagrelide in high-risk essential thrombocythemia. N Engl J Med. 2005;353:33–45.
Gisslinger H, Gotic M, Holowiecki J, Penka M, Thiele J, Kvasnicka HM, et al. Anagrelide compared with hydroxyurea in WHO-classified essential thrombocythemia: the ANAHYDRET Study, a randomized controlled trial. Blood. 2013;121:1720–8.
Fleming JS, Buyniski JP. A potent new inhibitor of platelet aggregation and experimental thrombosis, anagrelide (BL-4162A). Thromb Res. 1979;15:373–88.
Mazur EM, Rosmarin AG, Sohl PA, Newton JL, Narendran A. Analysis of the mechanism of anagrelide-induced thrombocytopenia in humans. Blood. 1992;79:1931–7.
Solberg LA Jr, Tefferi A, Oles KJ, Tarach JS, Petitt RM, Forstrom LA, et al. The effects of anagrelide on human megakaryocytopoiesis. Br J Haematol. 1997;99:174–80.
Tomer A. Effects of anagrelide on in vivo megakaryocyte proliferation and maturation in essential thrombocythemia. Blood. 2002;99:1602–9.
McCarty JM, Melone PD, Simanis JP, Kanamori D, Dessypris EN, Warshamana-Greene GS. A preliminary investigation into the action of anagrelide: thrombopoietin-c-Mpl receptor interactions. Exp Hematol. 2006;34:87–96.
Lane WJ, Hattori K, Dias S, Peerschke EI, Moore MA, Blanset DL, et al. Anagrelide metabolite induces thrombocytopenia in mice by inhibiting megakaryocyte maturation without inducing platelet aggregation. Exp Hematol. 2001;29:1417–24.
Ahluwalia M, Donovan H, Singh N, Butcher L, Erusalimsky JD. Anagrelide represses GATA-1 and FOG-1 expression without interfering with thrombopoietin receptor signal transduction. J Thromb Haemost. 2010;8:2252–61.
Ahluwalia M, Butcher L, Donovan H, Killick-Cole C, Jones PM, Erusalimsky JD. The gene expression signature of anagrelide provides an insight into its mechanism of action and uncovers new regulators of megakaryopoiesis. J Thromb Haemost. 2015;13:1103–12.
Fujiwara T, Alqadi YW, Okitsu Y, Fukuhara N, Onishi Y, Ishizawa K, et al. Role of transcriptional corepressor ETO2 in erythroid cells. Exp Hematol. 2013;41:303–15.
Fujiwara T, Saitoh H, Inoue A, Kobayashi M, Okitsu Y, Katsuoka Y, et al. 3-Deazaneplanocin A (DZNep), an inhibitor of S-adenosylmethionine-dependent methyltransferase, promotes erythroid differentiation. J Biol Chem. 2014;289:8121–34.
Shaw PH, Gilligan D, Wang XM, Thall PF, Corey SJ. Ex vivo expansion of megakaryocyte precursors from umbilical cord blood CD34 cells in a closed liquid culture system. Biol Blood Marrow Transpl. 2003;9:151–6.
Crispino JD, Weiss MJ. Erythro-megakaryocytic transcription factors associated with hereditary anemia. Blood. 2014;123:3080–8.
Gewirtz AM, Zhang J, Ratajczak J, Ratajczak M, Park KS, Li C, et al. Chemokine regulation of human megakaryocytopoiesis. Blood. 1995;86:2559–67.
Stevenson WS, Rabbolini DJ, Beutler L, Chen Q, Gabrielli S, Mackay JP, et al. Paris-Trousseau thrombocytopenia is phenocopied by the autosomal recessive inheritance of a DNA-binding domain mutation in FLI1. Blood. 2015;126:2027–30.
Joo JH, Ueda E, Bortner CD, Yang XP, Liao G, Jetten AM. Farnesol activates the intrinsic pathway of apoptosis and the ATF4-ATF3-CHOP cascade of ER stress in human T lymphoblastic leukemia Molt4 cells. Biochem Pharmacol. 2015;97:256–68.
Luís A, Martins JD, Silva A, Ferreira I, Cruz MT, Neves BM. Oxidative stress-dependent activation of the eIF2α–ATF4 unfolded protein response branch by skin sensitizer 1-fluoro-2,4-dinitrobenzene modulates dendritic-like cell maturation and inflammatory status in a biphasic manner [corrected]. Free Radic Biol Med. 2014;77:217–29.
Roy L, Bikorimana E, Lapid D, Choi H, Nguyen T, Dahl R. MiR-24 is required for hematopoietic differentiation of mouse embryonic stem cells. PLoS Genet. 2015;11:e1004959.
Chan MC, Hilyard AC, Wu C, Davis BN, Hill NS, Lal A, et al. Molecular basis for antagonism between PDGF and the TGFbeta family of signalling pathways by control of miR-24 expression. EMBO J. 2010;29:559–73.
Porcher C, Swat W, Rockwell K, Fujiwara Y, Alt FW, Orkin SH. The T cell leukemia oncoprotein SCL/tal-1 is essential for development of all hematopoietic lineages. Cell. 1996;86:47–57.
Chan MC, Nguyen PH, Davis BN, Ohoka N, Hayashi H, Du K, et al. A novel regulatory mechanism of the bone morphogenetic protein (BMP) signaling pathway involving the carboxyl-terminal tail domain of BMP type II receptor. Mol Cell Biol. 2007;27:5776–89.
Tao S, Cai Y, Sampath K. The Integrator subunits function in hematopoiesis by modulating Smad/BMP signaling. Development. 2009;136:2757–65.
Watson DK, Smyth FE, Thompson DM, Cheng JQ, Testa JR, Papas TS, et al. The ERGB/Fli-1 gene: isolation and characterization of a new member of the family of human ETS transcription factors. Cell Growth Differ. 1992;3:705–13.
Seth A, Robinson L, Thompson DM, Watson DK, Papas TS. Transactivation of GATA-1 promoter with ETS1, ETS2 and ERGB/Hu-FLI-1 proteins: stabilization of the ETS1 protein binding on GATA-1 promoter sequences by monoclonal antibody. Oncogene. 1993;8:1783–90.
Songdej N, Rao AK. Hematopoietic transcription factor mutations and inherited platelet dysfunction. F1000prime Rep. 2015;7:66.
Kawada H, Ito T, Pharr PN, Spyropoulos DD, Watson DK, Ogawa M. Defective megakaryopoiesis and abnormal erythroid development in Fli-1 gene-targeted mice. Int J Hematol. 2001;73:463–8.
Hart A, Melet F, Grossfeld P, Chien K, Jones C, Tunnacliffe A, et al. Fli-1 is required for murine vascular and megakaryocytic development and is hemizygously deleted in patients with thrombocytopenia. Immunity. 2000;13:167–77.
Fujiwara T, O’Geen H, Keles S, Blahnik K, Linnemann AK, Kang YA, et al. Discovering hematopoietic mechanisms through genome-wide analysis of GATA factor chromatin occupancy. Mol Cell. 2009;36:667–81.
Wilson NK, Foster SD, Wang X, Knezevic K, Schütte J, Kaimakis P, et al. Combinatorial transcriptional control in blood stem/progenitor cells: genome-wide analysis of ten major transcriptional regulators. Cell Stem Cell. 2010;7:532–44.
Tijssen MR, Cvejic A, Joshi A, Hannah RL, Ferreira R, Forrai A, et al. Genome-wide analysis of simultaneous GATA1/2, RUNX1, FLI1, and SCL binding in megakaryocytes identifies hematopoietic regulators. Dev Cell. 2011;20:597–609.
Moriguchi T, Yamamoto M. A regulatory network governing Gata1 and Gata2 gene transcription orchestrates erythroid lineage differentiation. Int J Hematol. 2014;100:417–24.
Acknowledgments
We thank Dr. Hozumi Motohashi (Tohoku University) for the helpful advice. We acknowledge the members of the Biomedical Research Core of Tohoku University School of Medicine for their support. We also thank Ms. Megumi Katoh (Tohoku University) for the excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Drs. Fujiwara and Harigae received a research grant from Chugai Pharmaceutical Co., Ltd. Other authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Sakurai, K., Fujiwara, T., Hasegawa, S. et al. Inhibition of human primary megakaryocyte differentiation by anagrelide: a gene expression profiling analysis. Int J Hematol 104, 190–199 (2016). https://doi.org/10.1007/s12185-016-2006-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-016-2006-2