Abstract
Objective
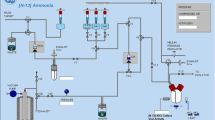
The GE FASTlab radiosynthesis module is routinely used for the production of [18F]FDG, utilizing the commercially available phosphate cassettes. Because of the observation of a white precipitate in the product vial before the product expiry time, we re-examined the quality of the produced [18F]FDG solution.
Methods
Phosphate buffered [18F]FDG solution was synthesized on the FASTlab and analyzed at both National Taiwan University Hospital (NTUH) of Taiwan and Royal Brisbane and Women’s Hospital (RBWH) of Australia. In addition to the standard product quality control (QC), the concentration of aluminum (Al3+) as probable cause of the precipitations in the [18F]FDG solution was analyzed by inductively coupled plasma mass spectrometry (ICP-MS at RBWH) and inductively coupled plasma optical emission spectrometry (ICP-OES at NTUH), and using three semi-quantitative methods at NTUH, Advantec® Alumi Check Test Strip, Quantofix® Aluminum Test Strip and MColortest™ Aluminum Test kit.
Results
The precipitates were observed in the [18F]FDG solution within 24 (NTUH) and 6 (RBWH) hours after the end of synthesis in 38–100 % of the batches, dependent on the batch of the FASTlab cassettes. Addition of metal-free HCl(aq) to aliquots of [18F]FDG containing precipitate, followed by ICP-MS analysis revealed Al3+ concentrations of 70–80 ppm. Al3+ concentrations of 10–12 ppm were detected in [18F]FDG batches that did not show any precipitation. In contrast, less than 5 ppm of the residual Al3+ was detected by semi-quantitative methods in all batches.
Conclusion
The US (USP), British (BP), European (EP) and Japanese (JP) pharmacopeias demand that [18F]FDG for injection should be clear and particulate free within the given shelf-life/expiration time. To avoid Al-phosphate precipitation within the product expiry time, FASTlab citrate cassettes, rather than phosphate cassettes, should be used for [18F]FDG production. Although testing for Al3+ is not listed in the [18F]FDG monographs of the USP, BP and EP, residual Al3+ levels should be considered in the interests of patient safety.


Similar content being viewed by others
References
Ido T, Wan CN, Wan CN, Casella V, Fowler J, Wolf A, Wolf A, Reivich M. Labeled 2‐deoxy‐d‐glucose analogs. 18F‐labeled 2‐deoxy‐2‐fluoro‐d‐glucose, 2‐deoxy‐2‐fluoro‐d‐mannose and 14C‐2‐deoxy‐2‐fluoro‐d‐glucose. J Label Cpd Radiopharm. 1978;14(2):175–83.
Shiue CY, Fowler JS, Wolf AP, Alexoff D, Macgregor RR. Gas-liquid chromatographic determination of relative amounts of 2-deoxy-2-flouro-d-glucose and 2-deoxy-2-fluoro-d-mannose synthesized from various methods. J Label Cpd Radiopharm. 1985;22(5):503–8.
Shiue CY, Salvadori PA, Wolf AP, Fowler JS, MacGregor RR. A new improved synthesis of 2-deoxy-2-[18F]fluoro-d-glucose from 18F-labeled acetyl hypofluorite. J Nucl Med. 1982;23(10):899–903.
Diksic M, Jolly D. New high-yield synthesis of 18F-labelled 2-deoxy-2-fluoro-d-glucose. Int J Appl Radiat Isot. 1983;34(6):893–6.
Bida GT, Satyamurthy N, Barrio JR. The synthesis of 2-[18F]fluoro-2-deoxy-d-glucose using glycals: a reexamination. J Nucl Med. 1984;25(12):1327–34.
Adam MJ. A rapid, stereoselective, high yielding synthesis of 2-deoxy-2-fluoro-d-hexopyranoses: reaction of glycals with acetyl hypofluorite. J Chem Soc Chem Commun. 1982;13:730–1.
Shiue CY, Wolf AP. A rapid synthesis of 2-deoxy-2-fluoro-d-glucose from xenon difluoride suitable for labelling with 18F. J Label Cpd Radiopharm. 1983;20(2):157–62.
Sood S, Firnau G, Garnett E. Radiofluorination with xenon difluoride: a new high yield synthesis of [18F]2-fluoro-2-deoxy-d-glucose. Int J Appl Radiat Isot. 1983;34(4):743–5.
Ehrenkaufer R, Potocki J, Jewett D. Simple synthesis of F-18-labeled 2-fluoro-2-deoxy-d-glucose: concise communication. J Nucl Med. 1984;25(3):333–7.
Tewson TJ. Synthesis of no-carrier-added fluorine-18 2-fluoro-2-deoxy-d-glucose. J Nucl Med. 1983;24(8):718–21.
Levy S, Elmaleh D, Livni E. A new method using anhydrous [18F]fluoride to radiolabel 2-[18F]fluoro-2-deoxy-d-glucose. J Nucl Med. 1982;23(10):918–22.
Hamacher K, Coenen H, Stöcklin G. Efficient stereospecific synthesis of no-carrier-added 2-[18F]-fluoro-2-deoxy-d-glucose using aminopolyether supported nucleophilic substitution. J Nucl Med. 1986;27(2):235–8.
Fowler JS, Ido T. Initial and subsequent approach for the synthesis of 18F-FDG. Semin Nucl Med. 2002;32(1):6–12.
Yu S. Review of 18F-FDG synthesis and quality control. Biomed Imaging Interv J. 2006;2(4):e57.
Saha GB. Fundamentals of nuclear pharmacy. 5th ed. New York: Springer-Verlag; 2004.
Krasikova R. PET radiochemistry automation: state of the art and future trends in 18F-nucleophilic fluorination. Curr Org Chem. 2013;17(19):2097–107.
GE Healthcare. FASTlab synthesizer FDG-phosphate application manual. Waukesha, WI: GE Healthcare; 2012.
GE Healthcare. FASTlab synthesizer FDG-citrate application manual. Waukesha, WI: GE Healthcare; 2012.
USP36/NF31. Fludeoxyglucose F18 injection. In: The United States Pharmacopeia, 37th edn, and The National Formulary, 32nd edn. Rockville: United States Pharmacopeia Convention, Inc.; 2014, p. 3026.
British Pharmacopoeia. Monographs: radiopharmaceutical preparation, fludeoxyglucose [18F]injection. London: British Pharmacopoeia Commission; 2015.
Radionuclides SoMAoC-P. Standards of compounds labeled with positron nuclides approved as established techniques for medical use (2001 revision). Japan Radioisotope Association. 2001.
GE Healthcare. TRACERlab MXFDG operator manual. Waukesha, WI: GE Healthcare; 2003.
GE Healthcare. TRACERlab MXFDG consumable manual. Waukesha, WI: GE Healthcare; 2003.
Fawdry RM. Radiolysis of 2-[18F] fluoro-2-deoxy-d-glucose(FDG) and the role of reductant stabilisers. Appl Radiat Isot. 2007;65(11):1193–201.
Ogawa T, Miyajima M, Wakiyama N, Terada K. Effects of phosphate buffer in parenteral drugs on particle formation from glass vials. Chem Pharm Bull (Tokyo). 2013;61(5):539–45.
FNCA. Guidline for quality assurance and quality control of 18F-FDG (2-deoxy-2-fluoro-d-glucose). Japan: FNCA Association; 2011.
Nakao R, Kida T, Suzuki K. Factors affecting quality control of [18F] FDG injection: bacterial endotoxins test, aluminum ions test and HPLC analysis for FDG and CIDG. Appl Radiat Isot. 2005;62(6):889–95.
Nandy S, Rajan M. Fully automated and simplified radiosynthesis of [18F]-3′-deoxy-3′-fluorothymidine using anhydro precursor and single neutral alumina column purification. J Radioanal Nucl Chem. 2010;283(3):741–8.
Nandy S, Rajan M, Korde A, Krishnamurthy N. The possibility of a fully automated procedure for radiosynthesis of fluorine-18-labeled fluoromisonidazole using a simplified single, neutral alumina column purification procedure. Appl Radiat Isot. 2010;68(10):1937–43.
Lide DR. CRC handbook of chemistry and physics. 85th ed. Boca Raton: CRC Press; 2004.
Walters LR, Martin KJ, Jacobson MS, Hung JC, Mosman EA. Stability evaluation of 18F-FDG at high radioactive concentrations. J Nucl Med Technol. 2012;40(1):52–6.
Long JZ, Jacobson MS, Hung JC. Comparison of FASTlab 18F-FDG production using phosphate and citrate buffer cassettes. J Nucl Med Technol. 2013;41(1):32–4.
Gard DR. Phosphoric acids and phosphates. In: Kirk-Othmer Encyclopedia of Chemical Technology. Hoboken, NJ: Wiley; 2000.
Schrödter K, Bettermann G, Staffel T, Wahl F, Klein T, Hofmann T, editors. Phosphoric acid and phosphates. Wiley-VCH Verlag GmbH & Co. KGaA; 2000.
Daydé S, Filella M, Berthon G. Aluminum speciation studies in biological fluids. Part 3. Quantitative investigation of aluminum-phosphate complexes and assessment of their potential significance in vivo. J Inorg Biochem. 1990;38(3):241–59.
Berthon G, Daydé S. Why aluminum phosphate is less toxic than aluminum hydroxide. J Am Coll Nutr. 1992;11(3):340–8.
Ogawa T, Miyajima M, Wakiyama N, Terada K. Aluminum elution and precipitation in glass vials: effect of pH and buffer species. Drug Dev Ind Pharm. 2013;41(2):315–21.
Motekaitis RJ, Martell AE. Complexes of aluminum (III) with hydroxy carboxylic acids. Inorg Chem. 1984;23(1):18–23.
Jacobson MS, Dankwart HR, Mahoney DW. Radiolysis of 2-[18F]fluoro-2-deoxy-d-glucose ([18F]FDG) and the role of ethanol and radioactive concentration. Appl Radiat Isot. 2009;67(6):990–5.
Dantas NM, Nascimento JE, Santos-Magalhães NS, Oliveira ML. Radiolysis of 2-[18F]fluoro-2-deoxy-d-glucose ([18F]FDG) and the role of ethanol, radioactive concentration and temperature of storage. Appl Radiat Isot. 2013;72:158–62.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, YY., Taylor, S., Koziorowski, J. et al. A two‐center study for the quality control of [18F]FDG using FASTlab phosphate cassettes. Ann Nucl Med 30, 563–571 (2016). https://doi.org/10.1007/s12149-016-1097-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-016-1097-4