Abstract
Objective
To define the efficacy and safety of low-dose rasburicase in children from south India with hematologic malignancies.
Methods
This study is a retrospective analysis of data on 41 children with hematologic malignacies with laboratory evidence of tumor lysis syndrome (TLS) or clinical features indicating high risk for developing TLS. Patients were treated with rasburicase in doses of 0.1–0.15 mg/kg dose, repeated when necessary.
Results
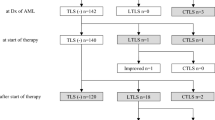
Male : Female ratio was 32:9. Thirty-six children had laboratory evidence of TLS and 5 were at risk for TLS. Diagnoses were T-cell acute lymphoblastic leukemia (ALL), 19; Pre-B ALL, 17; B-non-Hodgkin lymphoma (NHL), 2; T-NHL, 2; and acute myeloid leukemia (AML), 1. Initial plasma uric acid (PUA): median, 8.5 mg/dl (range, 4.3 to 45.5). Six had creatinine levels of >2 mg/dl on admission; and 10 had peak PO4 levels of >10 mg/dl. Dose of rasburicase used: median, 0.12 mg/kg (range, 0.08–0.24). Median reduction of PUA at 6 h: 80 % (range 40 to 98 %). Twenty-seven needed only one dose; 12 needed 2 or 3 doses; and two needed 5 doses each. One child required dialysis. None of the children developed anaphylaxis or hemolysis and there were no deaths from TLS.
Conclusions
Low-dose rasburicase (0.1–0.15 mg/kg) is safe and effective in reducing PUA in Indian children with lymphoid malignancies, and thus it may reduce the risk of renal failure from TLS.
Similar content being viewed by others
References
Saccente SL, Kohaut EC, Berkow RL. Prevention of tumor lysis syndrome using continuous veno-venous hemofiltration. Pediatr Nephrol. 1995;9:569–73.
Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med. 2011;364:1844–54.
Wilson FP, Berns JS. Onco-nephrology: tumor lysis synbdrome. Clin J Am Soc Nephrol. 2012;7:1730–9.
Pui CH, Mahmoud HH, Wiley JM, Woods GM, Leverer G, Camita B, et al. Recombinant urate oxidase for the prophylaxis or treatment of hyperuricemia in patients with leukemia or lymphoma. J Clin Oncol. 2001;19:697–704.
Ueng S. Rasburicase (Elitek): a novel agent for tumor lysis syndrome. BUMC Proc. 2005;18:275–9.
Diwedi P, Bhosle B, Chinnaswamy G, Aurora B, Banavali SD. Single dose/single vial rasburicase in the management of pediatric patients with hyperuricemia/tumor lysis syndrome. Pediatr Blood Cancer. 2013. ASPHO abstracts. Poster 766
Cairo MS, Bishop M. Tumor lysis syndrome: new therapeutic strategies and classification. Br J Haematol. 2004;127:3–11.
Patte C, Sakiroglu O, Ansoborlo S, Baruchel A, Plouvier E, Pacquement H, et al. Urate oxidase in the prevention and treatment of metabolic complications in patients with lymphoma and leukemia treated in the Societe Francaise d’ oncolgie Pediatric LMB 89 protocol. Ann Oncol. 2002;13:789–95.
Atra A, Gerrard M, Hobson R, Imeson JD, Ashley S, Pinkerton CR. Improved cure rate in children with B-cell acute lymphoblastic leukaemia (B-ALL) and stage IV B-cell non-Hodgkin’s lymphoma (B-NHL)– results of the UKCCSG 9003 protocol. Br J Cancer. 1998;77:2281–5.
Bowman WP, Shuster JJ, Cook B, Griffin T, Behm F, Pullen J, et al. Improved survival for children with B-cell acute lymphoblastic leukemia and stage IV small noncleaved-cell lymphoma: a pediatric oncology group study. J Clin Oncol. 1996;14:1252–61.
Cheuk DKL, Chiang AKS, Chan GCF, Ha SY. Urate oxidase for the prevention and treatment of tumor lysis syndrome in children with cancer. Cochrane Database Syst Rev. 2010;6: CD006945
Vadhan-Raj S, Fayad LE, Fanale MA, Pro B, Rodriquez A, Hagemeister FB, et al. A randomized triali of a single-dose rasburicase versus five-daily doses in patients at risk for tumor lysis syndrome. Ann Oncol. 2012;23:1640–5.
McBride A, Lathon SC, Boehmer L, Augustin KM, Butler, Westervelt P. Comparative evaluation of single fixed dosing and weight based dosing of rasburicase for tumor lysis syndrome. Pharmacotherapy. 2013;33:295–303.
Feng X, Dong K, Pence S, Inciardi J, Bhutada NS. Efficacy and cost of single dose rasburicase in prevention and treatment of adult tumor lysis syndrome: a meta-analysis. J Clin Pharm Ther. 2013;38:301–8.
Bosley A, Sonet A, Pinkerton CR, McCowage G, Bron D, Sanz MA, et al. Rasburicase (recombinant urate oxidase) for management of hyperuricemia in patients with cancer. Cancer. 2003;98:1048–54.
Bakhshi S, Bikramjit Singh R, Munot K, Pathania S. Complications of “very high” leukocytosis in pediatric acute leukemia patients managed without rasburicase or leukopheresis. Indian J Pediatr. 2014;81:817–20.
Allen KC, Champlain AH, Cotliar J, Belknap SM, West DP, Mehta J, et al. Anaphylaxis associated with administration of repeated courses of rasburicase: a research on adverse drug events and reports (RADAR) Project. Am Soc Hematol. Annual meeting 2012. Abstract# 1351.
Cheah CY, Lew TA, Seymour JF, Burbury K. Rasburicase causing severe oxidative hemolysis and methemoglobinemia in a patient with previously unrecognized glucose-6 phosphate dehydrogenase deficiency. Acta Haematol. 2013;130:254–9.
Sonbol MB, Yadav H, Vaidya R, Rana V, Witzig TE. Methemoglobinemia and hemolysis in a patient with G6PD deficiency treated with rasburicase. Am J Hematol. 2013;88:152–4.
Nair H. Neonatal screening program for G6PD deficiency in India: need and feasibility. Indian Pediatr. 2009;46:1045–9.
Contributions
SJ: Analyzing data and manuscript writing; VK and RD: Data collection and literature searching; PR: Primary patient care and review of manuscript; KR: Manuscript writing and revision; TKV: Manuscript writing and revision. SJ will act as guarantor for this paper.
Conflict of Interest
None.
Source of Funding
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jayabose, S., Kumar, V., Dhanabalan, R. et al. Low-dose Rasburicase in Hematologic Malignancies. Indian J Pediatr 82, 458–461 (2015). https://doi.org/10.1007/s12098-014-1606-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-014-1606-1