Abstract
Objective
To determine the extent and nature of off-label drug use in children admitted to a pediatric general ward in a tertiary health care centre
Methods
Consecutive patients aged 1 mo-12 years admitted to the general wards in a tertiary care center in Mumbai over a two-month period were prospectively enrolled in the study. British National Formulary [BNF] version 2005 was used to ascertain if the drug use was “off-label”. The off-label use was categorized as: administration of a greater/lesser dose, administration at a higher/lower frequency than indicated, administration for indications not described, administration of a drug not licensed for use in that age group and/ or use of alternative routes of administration. Descriptive statistics was used for calculating the off-label drug use.
Results
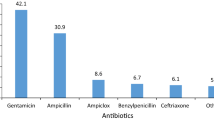
Two thousand prescriptions received by 600 subjects (M:F= 1.47:1) were analyzed. One thousand and forty-five (50.62%) prescriptions were off-label. The off-label drug use rate was 1.74+/−1.56 per patient. The maximum rate of off-label drugs was in infants (2.33/patient). ‘alteration in dosage’ was by far the commonest reason for off-label use; followed by ‘age’ and ‘indication’. Furosemide (i. v.), diazepam (i.v), cefotaxime (i.v), ethambutol (tab) and prednisolone (tab) were the five commonest off-label drugs used in the study population.
Conclusions
Off-label drug use was highly prevalent in general pediatric ward of a tertiary care hospital in India
Similar content being viewed by others
References
Conroy S, Choonara I, Impicciatore P, Mohn A, Arnell H, Rane A et al. Survey of unlicensed and off label drug use in paediatric wards in European countries. BMJ 2000; 320: 79–82
BNF for Children 2005. London, BMJ Publishing Group, 2005
Mudur G. Indian Medical Association wants off-label prescribing. BMJ 2004; 328: 974.
Sharma DC. Indian experts protest against off-label drug use. Lancet 2004; 363: 1372.
Jhaj R, Bhargava VK, Uppal R, Lekha S, Reeta K, Kaur N. Use of cold medications for upper respiratory tract infections in children. Pharmacoepidemiol Drug Saf 2001; 10: 323–327.
Turner S, Longworth A, Nunn A, Choonara I. Unlicensed and off label drug use in paediatric wards: prospective study. BMJ 1998; 316: 313–315.
Pandolfini C, Impicciatore P, Provasi DP, Rocchi F, Campi R, Bonati M, The Italian Pediatric Off-label Collaborative Group. Off-label use of drugs in Italy: a prospective, observational and multicentre study. Acta Paediatr 2002; 91: 339–347.
’t Jong GW, Vulto AG, De Hoog M, Schemmel KJ, Tibboel D, van den Anker JN. Unapproved and off-label use of drugs in a children’s hospital. New Engl J Med 2000; 343:1125
Gavrilov V, Lifshitz M, Levy J, Gorodischer R. Unlicensed and off-label medication use in a general pediatric ambulatory hospital unit in Israel. Isr Med Assoc J 2000; 2: 595–597.
Craig JS, Henderson CR, Magee FA. The extent of unlicensed and off-label drug use in the pediatric ward of a district general hospital in Northern Ireland. Ir Med J 2001; 94:237–240 [abstract]
Conroy S, Peden V. Unlicensed and off-label analgesic use in pediatric pain management. Paediatr Anaesthesia 2001; 11: 431–436.
Turner S, Nunn AJ, Fielding K, Choonara I. Adverse drug reactions to unlicensed and off-label drugs in pediatric wards: a prospective study. Acta Paediatr 1999; 88: 965–968.
McIntyre J, Conroy S, Avery A, Corns H, Choonara I. Unlicensed and off-label prescribing of drugs in general practice. Arch Dis Child 2000; 83: 498–501.
Conroy S, McIntyre J, Choonara I. Unlicensed and off-label drug use in neonates. Arch Dis Child Fetal Neonatal Ed 1999; 80: F142–F145.
Cuzzolin L, Zaccaron A, Fanos V. Unlicensed and off-label uses of drugs in paediatrics: a review of the literature. Fundamental and Clinical Pharmacology 2003; 17:125–131
American Academy of Pediatrics. Committee on Drugs 2000–2001. Pediatrics 2002; 110: 181–183.
Hill P. Off license and off label prescribing in children: litigation fears for physicians. Arch Dis Child 2005; 90: 17–18.
Boos J. Off label use-label off use? Ann Oncol 2003; 14: 1–5.
Bavdekar SB, Gogtay NJ. Unlicensed and off-label drug use in children. J Postgrad Med 2005; 51:249–252.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jain, S.S., Bavdekar, S.B., Gogtay, N.J. et al. Off-label drug use in children. Indian J Pediatr 75, 1133–1136 (2008). https://doi.org/10.1007/s12098-008-0188-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-008-0188-1