Abstract
Purpose
This study aimed at investigating whether the irradiated volume of pelvic bone marrow (PBM) and specific subsites may predict the occurrence of acute hematologic toxicity (HT) in anal cancer patients undergoing concurrent chemo-radiation.
Methods
50 patients, submitted to IMRT and concurrent chemotherapy, were analyzed. Several bony structures were defined on planning-CT: PBM and lumbar-sacral (LSBM), lower pelvis (LPBM) and iliac (IBM) bone marrow. On dose-volume histograms, dosimetric parameters were taken. Endpoints included white blood-cell-count (WBC), absolute-neutrophil-count (ANC), hemoglobin (Hb) and platelet nadirs and acute hematologic toxicity (HT) according to RTOG scoring scale. Generalized linear modeling was used to find correlations between dosimetric variables and blood cell nadirs, while logistic regression analysis was used to test correlation with ≥G3 HT. Receiver Operating Characteristic (ROC) curve analysis was used to evaluate the optimal cut-off points for predictive dosimetric variables with the Youden method.
Results
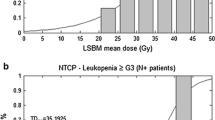
Maximum detected acute HT comprised 38 % of ≥G3 leukopenia and 32 % of ≥G3 neutropenia. Grade 2 anemia was observed in 4 % of patients and ≥G3 thrombocytopenia in 10 %. On multivariate analysis a higher PBM-V 20 was associated with lower WBC nadir. Increased LSBM-V 40 was correlated with a higher likelihood to develop ≥G3 HT. A cut-off point at 41 % for LSBM-V 40 was found. Patients with LSBM-V 40 ≥41 % were more likely to develop ≥G3 HT (55.3 vs. 32.4 %; p < 0.01).
Conclusions
Increased low-dose to pelvic bony structures significantly predicted for WBC decrease. Medium–high dose to specific osseous subsites was associated with a higher probability of HT. LSBM-V 40 was a strong predictor of ≥G3 HT. A threshold at 41 % for LSBM-V 40 could be used to limit HT.




Similar content being viewed by others
References
Franco P, Mistrangelo M, Arcadipane F, Munoz F, Sciacero P, Spadi R, et al. Intensity-modulated radiation therapy with simultaneous integrated boost combined with concurrent chemotherapy for the treatment of anal cancer patients: 4-year results of a consecutive case series. Cancer Invest. 2015;33:259–66.
Gunderson LL, Winter KA, Ajani JA, Pedersen JE, Moughan J, Benson AB 3rd, et al. Long-term update of US GI intergroup RTOG 98-11 phase III trial for anal carcinoma: survival, relapse, and colostomy failure with concurrent chemoradiation involving fluorouracil/mitomycin versus fluorouracil/cisplatin. J Clin Oncol. 2012;30:4344–51.
Franco P, Zeverino M, Migliaccio F, Sciacero P, Cante D, Casanova Borca V, et al. Intensity-modulated adjuvant whole breast radiation delivered with static angle tomotherapy (TomoDirect): a prospective case series. J Cancer Res Clin Oncol. 2013;139:1927–36.
Merlotti A, Alterio D, Vigna-Taglianti R, Muraglia A, Lastrucci L, Manzo R, et al. Technical guidelines for head and neck IMRT on behalf of the Italian association of radiation oncology—head and neck working group. Radiat Oncol. 2014;9:264.
Chuong MD, Freilich JM, Hoffe SE, Fulp W, Weber J, Almhanna K, et al. Intensity-modulated radiation therapy vs 3d conformal radiation therapy for squamous cell carcinoma of the anal canal. Gastrointest Cancer Res. 2013;6:39–45.
Mell LK, Schomas DA, Salama JK, Devisetty K, Aydogan B, et al. Association between bone marrow dosimetric parameters and acute hematologic toxicity in anal cancer patients treated with concurrent chemotherapy and intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2008;70:1431–7.
Kachnic LA, Winter K, Myerson RJ, Goodyear MD, Willins J, Esthappan J, et al. RTOG 0529: a phase 2 evaluation of dose-painted intensity modulated radiation therapy in combination with 5-fluorouracil and mitomycin C for the reduction of acute morbidity in carcinoma of the anal canal. Int J Radiat Oncol Biol Phys. 2013;86:27–33.
Ellis RE. The distribution of active bone marrow in the adult. Phys Med Biol. 1961;5:255–8.
Kachnic LA, Tsai HK, Coen JJ, Blaszkowsky LS, Hartshorn K, Kwak EL, et al. Dose-painted intensity-modulated radiation therapy for anal cancer: a multi-institutional report of acute toxicity and response to therapy. Int J Radiat Oncol Biol Phys. 2012;82:153–8.
Lk Mell, Kochanski JD, Roeske JC, Haslam JJ, Mehta N, Yamada SD, et al. Dosimetric predictors of acute hematologic toxicity in cervical cancer patients with concurrent cisplatin and intensity-modulated pelvic radiotherapy. Int J Radiat Oncol Biol Phys. 2006;66:1356–65.
Radiation Therapy Oncology Group. Acute radiation morbidity scoring criteria. http://www.rtog.org. Accessed 15 Dec 2015.
UKCCCR Anal cancer Trial Working Party. UK Co-ordination Committee on Cancer Research: epidermoid anal cancer: results from the UKCCCR randomized trial of radiotherapy alone versus radiotherapy, 5-fluorouracil, and mitomycin. Lancet. 1996;348:1049–54.
Bartelink H, Roelofsen F, Eschwege F, Rougier P, Bosset JF, Gonzalez DG, et al. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced anal cancer: results of a phase III randomized trial of the European Organization for Research and Treatment of Cancer Radiotherapy and Gastrointestinal Cooperative Group. J Clin Oncol. 1997;15:2040–9.
Flam M, John M, Pajak TF, Petrelli N, Myerson R, Doggett S, et al. Role of mitomycin in combination with fluorouracil and radiotherapy, and salvage chemoradiation in the definitive nonsurgical treatment of epidermoid carcinoma of the anal canal: results of a phase III randomized intergroup study. J Clin Oncol. 1996;14:2527–39.
Ajani JA, Winter KA, Gunderson LL, Pedersen J, Benson AB 3rd, Thomas CR Jr, et al. Fluorouracil, mitomycin, and radiotherapy vs fluorouracil, cisplatin, and radiotherapy for carcinoma of the anal canal: a randomized controlled trial. JAMA. 2008;299:1914–21.
Salama J, Mell LK, Schomas DA, Miller RC, Devisetty K, Jani AB, et al. Concurrent chemotherapy and intensity modulated radiation therapy for anal cancer patients: a multicenter experience. J Clin Oncol. 2007;25:4581–6.
Mauch P, Constine L, Greenberger J, Knospe W, Sullivan J, Liesveld JL, et al. Hematopoietic stem cell compartment: acute and late effects of radiation therapy and chemotherapy. Int J Radiat Oncol Biol Phys. 1995;31:1319–39.
Liang Y, Messer K, Rose BS, Lewis JH, Jiang SB, Yashar CM, et al. Impact of bone marrow radiation dose on acute hematologic toxicity in cervical cancer: principal component analysis on high dimensional data. Int J Radiat Oncol Biol Phys. 2010;78:912–9.
Sacks EL, Goris ML, Glatstein E, Gilbert E, Kaplan HS. Bone marrow regeneration following large field radiation: influence of volume, age, dose, and time. Cancer. 1978;42:1057–65.
Albuquerque K, Giangreco D, Morrison C, Siddiqui M, Sinacore J, Potkul R, et al. Radiation-related predictors of hematologic toxicity after concurrent chemoradiation for cervical cancer and implications for bone marrow-sparing pelvic IMRT. Int J Radiat Oncol Biol Phys. 2011;79:1043–7.
Rose BS, Aydogan B, Liang Y, Yeginer M, Hassalle MD, Dandekar V, et al. Normal tissue complication probability modeling of acute hematologic toxicity in cervical cancer patients treated with chemo radiotherapy. Int J Radiat Oncol Biol Phys. 2011;79:800–7.
Cheng JC, Bazan JG, Wu JK, Koong AC, Chang DT. Lumbosacral spine and marrow cavity modeling of acute hematologic toxicity in patients treated with intensity modulated radiation therapy for squamous cell carcinoma of the anal canal. Pract Radiat Oncol. 2014;4:198–206.
Rose BS, Liang Y, Lau SK, Jensen LG, Yashar CM, Hoh CK, et al. Correlation between radiation dose to 18F-FDG-PET defined active bone marrow subregions and acute hematologic toxicity in cervical cancer patients treated with chemo radiotherapy. Int J Radiat Oncol Biol Phys. 2012;82:1185–91.
Roeske JC, Lujan A, Reba RC, Penney BC, Yamada DS, Mundt AJ. Incorporation of SPECT bone marrow imaging into intensity modulated whole-pelvic radiation therapy treatment planning for gynecologic malignancies. Radiother Oncol. 2005;77:11–7.
Zhu H, Zakeri K, Vaida F, Carmona R, Dadachanji KK, Bair R, et al. Longitudinal study of acute hematologic toxicity in cervical cancer patients treated with chemo radiotherapy. J Med Imaging Radiat Oncol. 2015;59:386–93.
Wan J, Liu K, Li K, Li G, Zhang Z. Can dosimetric parameters predict acute hematologic toxicity in rectal cancer patients treated with intensity-modulated pelvic radiotherapy? Radiat Oncol. 2015;10:162.
Bazan JG, Luxton G, Kozak MM, Anderson EM, Hancock SL, Kapp DS, et al. Impact of chemotherapy on normal tissue complication probability models of acute hematologic toxicity in patients receiving pelvic intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys. 2012;87:983–91.
Franco P, Arcadipane F, Ragona R, Mistrangelo M, Cassoni P, Munoz F, et al. Volumetric modulated arc therapy (VMAT) in the combined modality treatment of anal cancer patients. Br J radiol. 2016;89(1060):20150832.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
We declare that we do not have any conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Franco, P., Ragona, R., Arcadipane, F. et al. Dosimetric predictors of acute hematologic toxicity during concurrent intensity-modulated radiotherapy and chemotherapy for anal cancer. Clin Transl Oncol 19, 67–75 (2017). https://doi.org/10.1007/s12094-016-1504-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-016-1504-2