Abstract
Purpose
Human epithelial growth factor receptor 2 (HER2) is over-expressed in several malignancies and represents an important therapeutic target. Aptamers are oligonucleotides that may potentially serve as tumor-homing ligand with excellent affinity and specificity for targeted cancer therapy. However, aptamers need to have nuclease resistance in order to function in vivo. The aim of this study was to generate a novel HER2 thioaptamer with enhanced nuclease resistance.
Methods
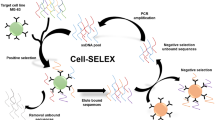
The HER2 thioaptamer is selected in an evolutionary process called systematic evolution of ligands by exponential enrichment.
Results
The thioaptamer could bind to the extracellular domain of HER2 with a K d of 172 nM and had minimal cross reactivity to trypsin or IgG. Moreover, the thioaptamer was found capable of binding with the HER2-positive breast cancer cells SK-BR-3 and MDA-MB-453, but not the HER2-negative cells MDA-MB-231. Notably, the thioaptamer HY6 largely maintained its structural integrity facing the nucleases in serum, while regular DNA aptamers were mostly digested. Additionally, the thioaptamer retained the capability of binding with the HER2-positive cells in the presence of serum, whereas non-thionated HER2 aptamer lost the binding function.
Conclusion
The results indicated that the selected thioaptamer was more resistant to nuclease than regular DNA aptamers and might potentially function as a HER2-targeting ligand in complicated environment.







Similar content being viewed by others
References
Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001;2(2):127–37. doi:10.1038/35052073.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235(4785):177–82.
Tateishi M, Ishida T, Mitsudomi T, Kaneko S, Sugimachi K. Prognostic value of c-erbB-2 protein expression in human lung adenocarcinoma and squamous cell carcinoma. Eur J Cancer. 1991;27(11):1372–5.
Lemoine NR, Jain S, Silvestre F, Lopes C, Hughes CM, McLelland E, et al. Amplification and overexpression of the EGF receptor and c-erbB-2 proto-oncogenes in human stomach cancer. Br J Cancer. 1991;64(1):79–83.
Hu Y, Bandla S, Godfrey TE, Tan D, Luketich JD, Pennathur A, et al. HER2 amplification, overexpression and score criteria in esophageal adenocarcinoma. Mod Pathol. 2011;24(7):899–907. doi:10.1038/modpathol.2011.47.
Cobleigh MA, Vogel CL, Tripathy D, Robert NJ, Scholl S, Fehrenbacher L, et al. Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J Clin Oncol. 1999;17(9):2639–48.
Tuerk C, Gold L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science. 1990;249(4968):505–10.
Ellington AD, Szostak JW. In vitro selection of RNA molecules that bind specific ligands. Nature. 1990;346(6287):818–22. doi:10.1038/346818a0.
Tombelli S, Minunni M, Mascini M. Analytical applications of aptamers. Biosens Bioelectron. 2005;20(12):2424–34. doi:10.1016/j.bios.2004.11.006.
Soontornworajit B, Wang Y. Nucleic acid aptamers for clinical diagnosis: cell detection and molecular imaging. Anal Bioanal Chem. 2011;399(4):1591–9. doi:10.1007/s00216-010-4559-x.
Dassie JP, Liu XY, Thomas GS, Whitaker RM, Thiel KW, Stockdale KR, et al. Systemic administration of optimized aptamer-siRNA chimeras promotes regression of PSMA-expressing tumors. Nat Biotechnol. 2009;27(9):839–49. doi:10.1038/nbt.1560.
Farokhzad OC, Cheng J, Teply BA, Sherifi I, Jon S, Kantoff PW, et al. Targeted nanoparticle-aptamer bioconjugates for cancer chemotherapy in vivo. Proc Natl Acad Sci USA. 2006;103(16):6315–20. doi:10.1073/pnas.0601755103.
Hu Y, Duan J, Zhan Q, Wang F, Lu X, Yang XD. Novel MUC1 aptamer selectively delivers cytotoxic agent to cancer cells in vitro. PLoS One. 2012;7(2):e31970. doi:10.1371/journal.pone.0031970.
Yang X, Wang H, Beasley DW, Volk DE, Zhao X, Luxon BA, et al. Selection of thioaptamers for diagnostics and therapeutics. Ann NY Acad Sci. 2006;1082:116–9. doi:10.1196/annals.1348.065.
Davis KA, Abrams B, Lin Y, Jayasena SD. Use of a high affinity DNA ligand in flow cytometry. Nucleic Acids Res. 1996;24(4):702–6. doi:10.1093/nar/24.4.702.
Shangguan D, Li Y, Tang Z, Cao ZC, Chen HW, Mallikaratchy P, et al. Aptamers evolved from live cells as effective molecular probes for cancer study. Proc Natl Acad Sci USA. 2006;103(32):11838–43. doi:10.1073/pnas.0602615103.
Dakappagari NK, Douglas DB, Triozzi PL, Stevens VC, Kaumaya PT. Prevention of mammary tumors with a chimeric HER-2 B-cell epitope peptide vaccine. Cancer Res. 2000;60(14):3782–9.
Liu Z, Duan JH, Song YM, Ma J, Wang FD, Lu X, et al. Novel HER2 aptamer selectively delivers cytotoxic drug to HER2-positive breast cancer cells in vitro. J Transl Med. 2012;10:148. doi:10.1186/1479-5876-10-148.
Zhao Q, Li XF, Le XC. Aptamer-modified monolithic capillary chromatography for protein separation and detection. Anal Chem. 2008;80(10):3915–20. doi:10.1021/ac702567x.
Bridonneau P, Chang YF, O’Connell D, Gill SC, Snyder DW, Johnson L, et al. High-affinity aptamers selectively inhibit human nonpancreatic secretory phospholipase A2 (hnps-PLA2). J Med Chem. 1998;41(6):778–86. doi:10.1021/jm970579k.
Lewis GD, Figari I, Fendly B, Wong WL, Carter P, Gorman C, et al. Differential responses of human tumor cell lines to anti-p185HER2 monoclonal antibodies. Cancer Immunol Immunother. 1993;37(4):255–63.
Hudziak RM, Lewis GD, Winget M, Fendly BM, Shepard HM, Ullrich A. p185HER2 monoclonal antibody has antiproliferative effects in vitro and sensitizes human breast tumor cells to tumor necrosis factor. Mol Cell Biol. 1989;9(3):1165–72.
Tang Z, Shangguan D, Wang K, Shi H, Sefah K, Mallikratchy P, et al. Selection of aptamers for molecular recognition and characterization of cancer cells. Anal Chem. 2007;79(13):4900–7. doi:10.1021/ac070189y.
Yoshino I, Goedegebuure PS, Peoples GE, Parikh AS, DiMaio JM, Lyerly HK et al. HER2/neu-derived peptides are shared antigens among human non-small cell lung cancer and ovarian cancer. Cancer Res. 1994;54(13):3387–90.
Brabender J, Danenberg KD, Metzger R, Schneider PM, Park J, Salonga D, et al. Epidermal growth factor receptor and HER2-neu mRNA expression in non-small cell lung cancer is correlated with survival. Clin Cancer Res. 2001;7(7):1850–5.
Coombs LM, Pigott DA, Sweeney E, Proctor AJ, Eydmann ME, Parkinson C, et al. Amplification and over-expression of c-erbB-2 in transitional cell carcinoma of the urinary bladder. Br J Cancer. 1991;63(4):601–8.
Famulok M, Hartig JS, Mayer G. Functional aptamers and aptazymes in biotechnology, diagnostics, and therapy. Chem Rev. 2007;107(9):3715–43. doi:10.1021/cr0306743.
Vester B, Wengel J. LNA (locked nucleic acid): high-affinity targeting of complementary RNA and DNA. Biochemistry. 2004;43(42):13233–41. doi:10.1021/bi0485732.
Lin Y, Nieuwlandt D, Magallanez A, Feistner B, Jayasena SD. High-affinity and specific recognition of human thyroid stimulating hormone (hTSH) by in vitro-selected 2′-amino-modified RNA. Nucleic Acids Res. 1996;24(17):3407–14. doi:10.1093/nar/24.17.3407.
Burmeister PE, Lewis SD, Silva RF, Preiss JR, Horwitz LR, Pendergrast PS, et al. Direct in vitro selection of a 2′-O-methyl aptamer to VEGF. Chem Biol. 2005;12(1):25–33. doi:10.1016/j.chembiol.2004.10.017.
Boomer RM, Lewis SD, Healy JM, Kurz M, Wilson C, McCauley TG. Conjugation to polyethylene glycol polymer promotes aptamer biodistribution to healthy and inflamed tissues. Oligonucleotides. 2005;15(3):183–95. doi:10.1089/oli.2005.15.183.
de Smidt PC, Le Doan T, de Falco S, van Berkel TJ. Association of antisense oligonucleotides with lipoproteins prolongs the plasma half-life and modifies the tissue distribution. Nucleic Acids Res. 1991;19(17):4695–700.
Somasunderam A, Ferguson MR, Rojo DR, Thiviyanathan V, Li X, O’Brien WA, et al. Combinatorial selection, inhibition, and antiviral activity of DNA thioaptamers targeting the RNase H domain of HIV-1 reverse transcriptase. Biochemistry. 2005;44(30):10388–95. doi:10.1021/bi0507074.
King DJ, Bassett SE, Li X, Fennewald SA, Herzog NK, Luxon BA, et al. Combinatorial selection and binding of phosphorothioate aptamers targeting human NF-kappa B RelA(p65) and p50. Biochemistry. 2002;41(30):9696–706. doi:10.1021/bi020220k.
Gallo M, Montserrat JM, Iribarren AM. Design and applications of modified oligonucleotides. Braz J Med Biol Res. 2003;36(2):143–51. doi:10.1590/S0100-879X2003000200001.
Kim MY, Jeong S. In vitro selection of RNA aptamer and specific targeting of ErbB2 in breast cancer cells. Nucleic Acid Ther. 2011;21(3):173–8. doi:10.1089/nat.2011.0283.
Dastjerdi K, Tabar GH, Dehghani H, Haghparast A. Generation of an enriched pool of DNA aptamers for an HER2-overexpressing cell line selected by Cell SELEX. Biotechnol Appl Biochem. 2011;58(4):226–30. doi:10.1002/bab.36.
Oh SS, Qian J, Lou X, Zhang Y, Xiao Y, Soh HT. Generation of highly specific aptamers via micromagnetic selection. Anal Chem. 2009;81(13):5490–5. doi:10.1021/ac900759k.
Acknowledgments
We acknowledge the funding support from the Chinese Ministry of Science and Technology (2011CB933504, 2011CB911003), and the 111 project from the Chinese Ministry of Education.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hu, Y., Duan, J., Cao, B. et al. Selection of a novel DNA thioaptamer against HER2 structure. Clin Transl Oncol 17, 647–656 (2015). https://doi.org/10.1007/s12094-015-1292-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1292-0