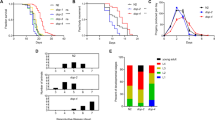
Abstract
Lifespan extension is an all systems encompassing event. Involvement of reduced insulin/IGF1 signalling is well worked out, first in the model organism Caenorhbaditis elegans followed by other systems including humans. But the role of neuronal component in lifespan extension is not well understood due to the refractory nature of neurons to small RNA interference (sRNAi) in C. elegans. Earlier, we have demonstrated that an antihypertensive drug, reserpine, extends lifespan through modulation of neurotransmitter release, especially, acetylcholine, in C. elegans. Intriguingly, the reserpine mediated lifespan extension (RMLE) does not happen through the known longevity pathways. Here, we report that the D2-type dopamine receptor (DOP-3), which acts through the inhibitory G-protein coupled (Gαi) pathway mediated signalling is partly required for RMLE. In the dop-3 loss of function mutant RMLE is shortened. DOP-3 acts through Gαo (goa-1). One of the downstream targets of G protein signalling is the transcription factor, jun-1. MRP-1, an ATP binding cassette transporter, belonging to the multidrug resistance protein family is one of the genes turned on by JUN-1. RMLE is shortened in dop-3→goa-1→jun1→mrp-1 loss of function mutants, elucidating the contribution of dop-3 signalling. The dop-3 receptor system is known to inhibit acetylcholine release. This suggests dopamine receptor, dop-3 could be contributing to the modulation of acetylcholine release by reserpine. ERI-1 is a 3′-5′ exoribonuclease, one of the negative regulators of sRNAi, whose loss of function makes neurons amenable to siRNA. In the absence of eri-1, RMLE is shortened. In the dop-3 loss-of-function background, lack of eri-1 completely abolishes RMLE. This suggests that dop-3 and eri-1 act in independent parallel pathways for RMLE and these two pathways are essential and sufficient for the longevity enhancement by reserpine in C. elegans.





Similar content being viewed by others
References
Allen AT, Maher KN, Wani KA, Betts KE and Chase DL 2011 Co expressed D1- and D2-like dopamine receptors antagonistically modulate acetylcholine release in Caenorhabditis elegans. Genetics 188 579–590
Ansel KM, Pastor WA, Rath N, Lapan AD, Glasmacher E, Wolf C, Smith LC, Papadopoulou N, et al. 2008 Mouse Eri-1 interacts with the ribosome and catalyzes 5.8S rRNA processing. Nat. Struct. Mol. Biol. 15 523–30
Arya U, Dwivedi H and Subramaniam JR 2009 Reserpine ameliorates Abeta toxicity in the Alzheimer's disease model in C. elegans. Exp. Gerontol. 44 462–466
Arya U, Das CK and Subramaniam JR 2010 Caenorhabditis elegans for preclinical drug discovery. Curr. Sci. 99(12)
Brenner S 1974 the genetics of Caenorhabditis elegans. Genetics 77 71–94
Chase DL, Pepper JS and Koelle MR 2004 Mechanism of extrasynaptic dopamine signaling in Caenorhabditis elegans. Nat. Neurosci. 7 1096–103
Cripe LD, Gelfanov VM, Smith EA, Spigel DR, Phillips CA, Gabig TG, Jung S-H, Fyffe J, et al. 2002 Role for c-jun N-terminal kinase in treatment-refractory acute myeloid leukemia (AML): signaling to multidrug-efflux and hyperproliferation. Leukemia 16 799–812
Duerr JS, Frisby DL, Gaskin J, Duke A, Asermely K, Huddleston D, Eiden LE and Rand JB 1999 the cat-1 gene of Caenorhabditis elegans encodes a vesicular monoamine transporter required for specific monoamine-dependent behaviours. J. Neurosci. 19 72–84
Gabel HW and Ruvkun G 2008 The exonuclease ERI-1 has a conserved dual role in 5.8S rRNA processing and RNAi. Nat. Struct. Mol. Biol. 15 531–533
Honda Y, Higashibata A, Matsunaga Y, Yonezawa Y, Kawano T, Higashitani A, Kuriyama K, Shimazu T, et al. 2012 Genes down-regulated in spaceflight are involved in the control of longevity in Caenorhabditis elegans. Sci. Rep. 2 487
Huang H, Lu-Bo Y, Haddad GG 2014 A Drosophila ABC Transporter Regulates Lifespan. PLoS Genet 10(12)
Kawasaki M, Hisamoto N, Iino Y, Yamamoto M, Ninomiya-Tsuji J and Matsumoto K 1999 A Caenorhabditis elegans JNK signal transduction pathway regulates coordinated movement via type-D GABAergic motor neurons. EMBO J. 18 3604–3615
Kennedy S, Wang D and Ruvkun G 2004 A conserved siRNA-degrading RNase negatively regulates RNA interference in C. elegans. Nature 427 645–9
Kenyon C 2010 The genetics of aging. Nature. 464 504–512
Kenyon C, Chang J, Gensch E, Rudner A and Tabtiang R 1993 A C. elegans mutant that lives twice as long as wild type. Nature 366 461–464
Knoll J 1992 Deprenyl-medication: a strategy to modulate the age-related decline of the striatal dopaminergic system. J. Am. Geriatr. Soc. 40 839–47
Lee YS, Y-S P, Nam S, Suh SJ, Kaang BK, Cho NJ and Lee JH 2000 Characterization of GAR-2, a novel G protein-linked acetylcholine receptor from Caenorhabditis elegans. J. Neurochem. 75 1800–9
Libina N, Berman JR and Kenyon C 2003 Tissue-specific activities of C. elegans DAF-16 in the regulation of lifespan. Cell 15 489–502
Loer CM and Kenyon CJ 1993 Serotonin-deficient mutants and male mating behavior in the nematode Caenorhabditis elegans. J. Neurosci. 13 5407–17
Saharia K, Arya U, Kumar R, Sahu R, Das CK, Gupta K, Dwivedi H and Subramaniam JR 2012 Reserpine modulates neurotransmitter release to extend lifespan and alleviate age-dependent Aβ proteotoxicity in Caenorhabditis elegans. Exp. Gerontol. 47 188–97
Sawin ER, Ranganathan R and Horvitz HR 2000 C. elegans locomotory rate is modulated by the environment through a dopaminergic pathway and by experience through a serotonergic pathway. Neuron. 26 619–631
Srivastava D, Arya U, SoundaraRajan T, Dwivedi H, Kumar S and Subramaniam JR 2008 Reserpine can confer stress tolerance and lifespan extension in the nematode C. elegans. Biogerontology 9 309–316
Sulston J, Dew M and Brenner S 1975 Dopaminergic neurons in the nematode Caenorhabditis elegans. J. Comp. Neurol. 1633 215–26
Sundaram P, Echalier B, Han W, Hull D and Timmons L 2006 ATP-binding Cassette Transporters Are Required for Efficient RNA Interference in Caenorhabditis elegans. Mol. Biol. Cell 17 3678–3688
Sze JY, Victor M, Loer C, Shi Y and Ruvkun G 2000 Food and metabolic signalling defects in Caenorhabditis elegans serotonin-synthesis mutant. Nature 403 560–4
Acknowledgements
The authors are grateful to Prof K Subramaniam, Indian Institute of Technology, Kanpur & Chennai, India, for the technical help and suggestions. The authors gratefully acknowledge the financial support from Department of Biotechnology, India, and the Caenorhabditis Genetics Center supported by National Institute of Health, USA, and the Gene Knockout Consortium, British Columbia for the C. elegans strains.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Subhash C Lakhotia
[Saharia K, Kumar R, Gupta K, Mishra S and Subramaniam JR 2016 Reserpine requires the D2-type receptor, dop-3, and the exoribonuclease, eri-1, to extend the lifespan in C. elegans. J. Biosci.]
Rights and permissions
About this article
Cite this article
Saharia, K., Kumar, R., Gupta, K. et al. Reserpine requires the D2-type receptor, dop-3, and the exoribonuclease, eri-1, to extend the lifespan in C. elegans . J Biosci 41, 689–695 (2016). https://doi.org/10.1007/s12038-016-9652-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12038-016-9652-7