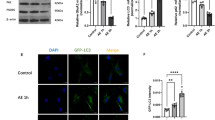
Abstract
Succinobucol (succinyl ester of probucol) is a lipid-lowering compound with anti-inflammatory and antioxidant properties. Recent experimental evidence has highlighted the potential neuroprotective effects of succinobucol. In the present study, cultured neuroblastoma (SH-SY5Y) cells were used to investigate mechanisms mediating the potential protective effect of succinobucol against mitochondrial metabolic impairment and oxidative stress induced by 3-nitropropionic acid (3-NP), a succinate dehydrogenase inhibitor that has been used in experimental models of the Huntington disease (HD). 3-NP decreased cellular viability after 24 h of incubation. This decline in cellular viability was preceded by (i) reduced mitochondrial complex II activity, (ii) increased reactive species generation, (iii) decreased mitochondrial membrane potential (ΔΨm), and (iv) diminished glutathione (GSH) levels. Succinobucol pretreatment (6 days) significantly prevented 3-NP-induced loss of cellular viability, generation of reactive oxygen species, and decrease of ΔΨm. However, succinobucol pretreatment did not protect against 3-NP-induced inhibition of mitochondrial complex II activity, pointing to the mitigation of secondary events resultant from mitochondrial complex II inhibition. Succinobucol pretreatment (6 days) significantly increased (50 %) the levels of GSH in SH-SY5Y cells, and this event was paralleled by significant increases in glutamate cysteine ligase messenger RNA (mRNA) expression and activity (GCL; the first enzyme in the GSH biosynthesis). The present findings are the first to show that succinobucol increases GSH levels via upregulation of GCL activity (possibly through the activation of the nuclear (erythroid-derived 2)-related factor (Nrf2)/antioxidant response element (ARE) pathway), displaying protective effects against mitochondrial dysfunction-derived oxidative stress.








Similar content being viewed by others
References
Roos RA (2010) Huntington’s disease: a clinical review. Orphanet J Rare Dis 5:40. doi:10.1186/1750-1172-5-40
Southwell AL, Patterson PH (2011) Gene therapy in mouse models of huntington disease. Neuroscientist 17:153–162. doi:10.1177/1073858410386236
Reiner A, Albin RL, Anderson KD, D’Amato CJ, Penney JB, Young AB (1988) Differential loss of striatal projection neurons in Huntington disease. Proc Natl Acad Sci U S A 85:5733–5737
Vonsattel JP, DiFiglia M (1998) Huntington disease. J Neuropathol Exp Neurol 57:369–384
Heinsen H, Strik M, Bauer M, Luther K, Ulmar G, Gangnus D, Jungkunz G, Eisenmenger W, Gotz M (1994) Cortical and striatal neurone number in Huntington’s disease. Acta Neuropathol 88:320–333
Mann DM, Oliver R, Snowden JS (1993) The topographic distribution of brain atrophy in Huntington’s disease and progressive supranuclear palsy. Acta Neuropathol 85:553–559
Damiano M, Galvan L, Deglon N, Brouillet E (2010) Mitochondria in Huntington’s disease. Biochim Biophys Acta 1802:52–61. doi:10.1016/j.bbadis.2009.07.012
Mochel F, Haller RG (2011) Energy deficit in Huntington disease: why it matters. J Clin Invest 121:493–499. doi:10.1172/JCI45691
Oliveira JM (2010) Nature and cause of mitochondrial dysfunction in Huntington’s disease: focusing on huntingtin and the striatum. J Neurochem 114:1–12. doi:10.1111/j.1471-4159.2010.06741.x
Ramaswamy S, McBride JL, Kordower JH (2007) Animal models of Huntington’s disease. ILAR J 48:356–373
Beal MF, Brouillet E, Jenkins BG, Ferrante RJ, Kowall NW, Miller JM, Storey E, Srivastava R, Rosen BR, Hyman BT (1993) Neurochemical and histologic characterization of striatal excitotoxic lesions produced by the mitochondrial toxin 3-nitropropionic acid. J Neurosci 13:4181–4192
Tunez I, Tasset I, Perez-De La Cruz V, Santamaria A (2010) 3-Nitropropionic acid as a tool to study the mechanisms involved in Huntington’s disease: past, present and future. Molecules 15:878–916. doi:10.3390/molecules15020878
Kumar P, Kumar A (2009) Possible role of sertraline against 3-nitropropionic acid induced behavioral, oxidative stress and mitochondrial dysfunctions in rat brain. Prog Neuropsychopharmacol Biol Psychiatry 33:100–108. doi:10.1016/j.pnpbp.2008.10.013
Bizat N, Hermel JM, Humbert S, Jacquard C, Creminon C, Escartin C, Saudou F, Krajewski S, Hantraye P, Brouillet E (2003) In vivo calpain/caspase cross-talk during 3-nitropropionic acid-induced striatal degeneration: implication of a calpain-mediated cleavage of active caspase-3. J Biol Chem 278:43245–43253. doi:10.1074/jbc.M305057200
Kumar P, Kalonia H, Kumar A (2010) Huntington’s disease: pathogenesis to animal models. Pharmacol Rep 62:1–14
Lee WT, Itoh T, Pleasure D (2002) Acute and chronic alterations in calcium homeostasis in 3-nitropropionic acid-treated human NT2-N neurons. Neuroscience 113:699–708
Lee WT, Yin HS, Shen YZ (2002) The mechanisms of neuronal death produced by mitochondrial toxin 3-nitropropionic acid: the roles of N-methyl-D-aspartate glutamate receptors and mitochondrial calcium overload. Neuroscience 112:707–716
Montilla P, Espejo I, Munoz MC, Bujalance I, Munoz-Castaneda JR, Tunez I (2004) Effect of red wine on oxidative stress and hypercholesterolemia induced by feeding a high-cholesterol diet in rat. J Physiol Biochem 60:259–264
Perez-De La Cruz V, Gonzalez-Cortes C, Pedraza-Chaverri J, Maldonado PD, Andres-Martinez L, Santamaria A (2006) Protective effect of S-allylcysteine on 3-nitropropionic acid-induced lipid peroxidation and mitochondrial dysfunction in rat brain synaptosomes. Brain Res Bull 68:379–383. doi:10.1016/j.brainresbull.2005.09.013
Perez-De La Cruz V, Elinos-Calderon D, Carrillo-Mora P, Silva-Adaya D, Konigsberg M, Moran J, Ali SF, Chanez-Cardenas ME, Perez-De La Cruz G, Santamaria A (2010) Time-course correlation of early toxic events in three models of striatal damage: modulation by proteases inhibition. Neurochem Int 56:834–842. doi:10.1016/j.neuint.2010.03.008
Elinos-Calderon D, Robledo-Arratia Y, Perez-De La Cruz V, Maldonado PD, Galvan-Arzate S, Pedraza-Chaverri J, Santamaria A (2010) Antioxidant strategy to rescue synaptosomes from oxidative damage and energy failure in neurotoxic models in rats: protective role of S-allylcysteine. J Neural Transm 117:35–44. doi:10.1007/s00702-009-0299-5
Ayala-Pena S (2013) Role of oxidative DNA damage in mitochondrial dysfunction and Huntington’s disease pathogenesis. Free Radic Biol Med 62:102–110. doi:10.1016/j.freeradbiomed.2013.04.017
Chaturvedi RK, Flint Beal M (2013) Mitochondrial diseases of the brain. Free Radic Biol Med 63:1–29. doi:10.1016/j.freeradbiomed.2013.03.018
Liu J, Wang LN (2014) Mitochondrial enhancement for neurodegenerative movement disorders: a systematic review of trials involving creatine, coenzyme Q10, idebenone and mitoquinone. CNS Drugs 28:63–68. doi:10.1007/s40263-013-0124-4
Cusin C, Franco FB, Fernandez-Robles C, DuBois CM, Welch CA (2013) Rapid improvement of depression and psychotic symptoms in Huntington’s disease: a retrospective chart review of seven patients treated with electroconvulsive therapy. Gen Hosp Psychiatry 35(678):e3–e5. doi:10.1016/j.genhosppsych.2013.01.015
Gallina P, Paganini M, Lombardini L, Giordano G, Mascalchi M, Romoli AM, Ghelli E, Porfirio B, Vannelli GB, Di Lorenzo N (2011) Progress in restorative neurosurgery: human fetal striatal transplantation in Huntington’s disease. Review. J Neurosurg Sci 55:371–381
Colle D, Santos DB, Moreira EL, Hartwig JM, dos Santos AA, Zimmermann LT, Hort MA, Farina M (2013) Probucol increases striatal glutathione peroxidase activity and protects against 3-nitropropionic acid-induced pro-oxidative damage in rats. PLoS One 8:e67658. doi:10.1371/journal.pone.0067658
Colle D, Hartwig JM, Soares FA, Farina M (2012) Probucol modulates oxidative stress and excitotoxicity in Huntington’s disease models in vitro. Brain Res Bull 87:397–405. doi:10.1016/j.brainresbull.2012.01.003
Colle D, Santos DB, Hartwig JM, Godoi M, Braga AL, Farina M (2013) Succinobucol versus probucol: higher efficiency of succinobucol in mitigating 3-NP-induced brain mitochondrial dysfunction and oxidative stress in vitro. Mitochondrion. doi:10.1016/j.mito.2013.01.005
Yamashita S, Matsuzawa Y (2009) Where are we with probucol: a new life for an old drug? Atherosclerosis 207:16–23. doi:10.1016/j.atherosclerosis.2009.04.002
Buckley MM, Goa KL, Price AH, Brogden RN (1989) Probucol. A reappraisal of its pharmacological properties and therapeutic use in hypercholesterolaemia. Drugs 37:761–800
Yamashita S, Hbujo H, Arai H, Harada-Shiba M, Matsui S, Fukushima M, Saito Y, Kita T, Matsuzawa Y (2008) Long-term probucol treatment prevents secondary cardiovascular events: a cohort study of patients with heterozygous familial hypercholesterolemia in Japan. J Atheroscler Thromb 15:292–303
Browne KF, Prystowsky EN, Heger JJ, Cerimele BJ, Fineberg N, Zipes DP (1984) Prolongation of the QT interval induced by probucol: demonstration of a method for determining QT interval change induced by a drug. Am Heart J 107:680–684
Tardif JC, Gregoire J, L’Allier PL (2002) Prevention of restenosis with antioxidants: mechanisms and implications. Am J Cardiovasc Drugs 2:323–334
Stocker R (2009) Molecular mechanisms underlying the antiatherosclerotic and antidiabetic effects of probucol, succinobucol, and other probucol analogues. Curr Opin Lipidol 20:227–235. doi:10.1097/MOL.0b013e32832aee68
Kunsch C, Luchoomun J, Grey JY, Olliff LK, Saint LB, Arrendale RF, Wasserman MA, Saxena U, Medford RM (2004) Selective inhibition of endothelial and monocyte redox-sensitive genes by AGI-1067: a novel antioxidant and anti-inflammatory agent. J Pharmacol Exp Ther 308:820–829. doi:10.1124/jpet.103.059733
Midwinter RG, Maghzal GJ, Dennis JM, Wu BJ, Cai H, Kapralov AA, Belikova NA, Tyurina YY, Dong LF, Khachigian L, Neuzil J, Kagan VE, Stocker R (2012) Succinobucol induces apoptosis in vascular smooth muscle cells. Free Radic Biol Med 52:871–879. doi:10.1016/j.freeradbiomed.2011.11.029
Weingarten DM (2004) Process of preparing esters and ethers of probucol and derivatives thereof. Patent WO2004062622
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Petegnief V, Friguls B, Sanfeliu C, Sunol C, Planas AM (2003) Transforming growth factor-alpha attenuates N-methyl-D-aspartic acid toxicity in cortical cultures by preventing protein synthesis inhibition through an Erk1/2-dependent mechanism. J Biol Chem 278:29552–29559. doi:10.1074/jbc.M300661200
Ali SF, LeBel CP, Bondy SC (1992) Reactive oxygen species formation as a biomarker of methylmercury and trimethyltin neurotoxicity. Neurotoxicology 13:637–648
Fischer JC, Ruitenbeek W, Berden JA, Trijbels JM, Veerkamp JH, Stadhouders AM, Sengers RC, Janssen AJ (1985) Differential investigation of the capacity of succinate oxidation in human skeletal muscle. Clin Chim Acta 153:23–36
Reers M, Smith TW, Chen LB (1991) J-aggregate formation of a carbocyanine as a quantitative fluorescent indicator of membrane potential. Biochemistry 30:4480–4486
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Cooper AJL (1998) Role of astrocytes in maintaining cerebral glutathione homeostasis and in protecting the brain against xenobiotics and oxidative stress. In: Francis Ta (ed) The role of glutathione in the nervous system, Washington, pp 91–115
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–333
Seelig GF, Meister A (1985) Glutathione biosynthesis; gamma-glutamylcysteine synthetase from rat kidney. Methods Enzymol 113:379–390
Engel D, Zomkowski AD, Lieberknecht V, Rodrigues AL, Gabilan NH (2013) Chronic administration of duloxetine and mirtazapine downregulates proapoptotic proteins and upregulates neurotrophin gene expression in the hippocampus and cerebral cortex of mice. J Psychiatr Res 47:802–808. doi:10.1016/j.jpsychires.2013.02.013
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Liot G, Bossy B, Lubitz S, Kushnareva Y, Sejbuk N, Bossy-Wetzel E (2009) Complex II inhibition by 3-NP causes mitochondrial fragmentation and neuronal cell death via an NMDA- and ROS-dependent pathway. Cell Death Differ 16:899–909. doi:10.1038/cdd.2009.22
Lubos E, Loscalzo J, Handy DE (2011) Glutathione peroxidase-1 in health and disease: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal 15:1957–1997. doi:10.1089/ars.2010.3586
Dringen R, Hirrlinger J (2003) Glutathione pathways in the brain. Biol Chem 384:505–516. doi:10.1515/BC.2003.059
Farina M, Campos F, Vendrell I, Berenguer J, Barzi M, Pons S, Sunol C (2009) Probucol increases glutathione peroxidase-1 activity and displays long-lasting protection against methylmercury toxicity in cerebellar granule cells. Toxicol Sci 112:416–426. doi:10.1093/toxsci/kfp219
Lu SC (2013) Glutathione synthesis. Biochim Biophys Acta 1830:3143–3153. doi:10.1016/j.bbagen.2012.09.008
Satoh T, McKercher SR, Lipton SA (2013) Nrf2/ARE-mediated antioxidant actions of pro-electrophilic drugs. Free Radic Biol Med 65:645–657. doi:10.1016/j.freeradbiomed.2013.07.022
Jung KA, Kwak MK (2010) The Nrf2 system as a potential target for the development of indirect antioxidants. Molecules 15:7266–7291. doi:10.3390/molecules15107266
Cao Z, Hallur S, Qiu HZ, Peng X, Li Y (2004) Induction of endogenous glutathione by the chemoprotective agent, 3H-1,2-dithiole-3-thione, in human neuroblastoma SH-SY5Y cells affords protection against peroxynitrite-induced cytotoxicity. Biochem Biophys Res Commun 316:1043–1049. doi:10.1016/j.bbrc.2004.02.156
Muldrew KM, Franks AM (2009) Succinobucol: review of the metabolic, antiplatelet and cardiovascular effects. Expert Opin Investig Drugs 18:531–539. doi:10.1517/13543780902849244
Santos DB, Peres KC, Ribeiro RP, Colle D, dos Santos AA, Moreira EL, Souza DO, Figueiredo CP, Farina M (2012) Probucol, a lipid-lowering drug, prevents cognitive and hippocampal synaptic impairments induced by amyloid beta peptide in mice. Exp Neurol 233:767–775. doi:10.1016/j.expneurol.2011.11.036
Ribeiro RP, Moreira EL, Santos DB, Colle D, Dos Santos AA, Peres KC, Figueiredo CP, Farina M (2013) Probucol affords neuroprotection in a 6-OHDA mouse model of Parkinson’s disease. Neurochem Res. doi:10.1007/s11064-012-0965-0
Solesio ME, Saez-Atienzar S, Jordan J, Galindo MF (2013) 3-Nitropropionic acid induces autophagy by forming mitochondrial permeability transition pores rather than activating the mitochondrial fission pathway. Br J Pharmacol 168:63–75. doi:10.1111/j.1476-5381.2012.01994.x
Uo T, Veenstra TD, Morrison RS (2009) Histone deacetylase inhibitors prevent p53-dependent and p53-independent Bax-mediated neuronal apoptosis through two distinct mechanisms. J Neurosci 29:2824–2832. doi:10.1523/JNEUROSCI. 6186-08.2009
Tabrizi SJ, Cleeter MW, Xuereb J, Taanman JW, Cooper JM, Schapira AH (1999) Biochemical abnormalities and excitotoxicity in Huntington’s disease brain. Ann Neurol 45:25–32
Procaccio V, Bris C, Chao de la Barca JM, Oca F, Chevrollier A, Amati-Bonneau P, Bonneau D, Reynier P (2014) Perspectives of drug-based neuroprotection targeting mitochondria. Rev Neurol (Paris) 170:390–400. doi:10.1016/j.neurol.2014.03.005
Lambert AJ, Brand MD (2004) Superoxide production by NADH:ubiquinone oxidoreductase (complex I) depends on the pH gradient across the mitochondrial inner membrane. Biochem J 382:511–517. doi:10.1042/BJ20040485
Adam-Vizi V (2005) Production of reactive oxygen species in brain mitochondria: contribution by electron transport chain and non-electron transport chain sources. Antioxid Redox Signal 7:1140–1149. doi:10.1089/ars.2005.7.1140
Witting PK, Wu BJ, Raftery M, Southwell-Keely P, Stocker R (2005) Probucol protects against hypochlorite-induced endothelial dysfunction: identification of a novel pathway of probucol oxidation to a biologically active intermediate. J Biol Chem 280:15612–15618. doi:10.1074/jbc.M414256200
Franklin CC, Backos DS, Mohar I, White CC, Forman HJ, Kavanagh TJ (2009) Structure, function, and post-translational regulation of the catalytic and modifier subunits of glutamate cysteine ligase. Mol Aspects Med 30:86–98. doi:10.1016/j.mam.2008.08.009
Forman HJ, Zhang H, Rinna A (2009) Glutathione: overview of its protective roles, measurement, and biosynthesis. Mol Aspects Med 30:1–12. doi:10.1016/j.mam.2008.08.006
Lu SC (2009) Regulation of glutathione synthesis. Mol Aspects Med 30:42–59. doi:10.1016/j.mam.2008.05.005
Liu RM, Gao L, Choi J, Forman HJ (1998) Gamma-glutamylcysteine synthetase: mRNA stabilization and independent subunit transcription by 4-hydroxy-2-nonenal. Am J Physiol 275:L861–L869
McMahon M, Itoh K, Yamamoto M, Hayes JD (2003) Keap1-dependent proteasomal degradation of transcription factor Nrf2 contributes to the negative regulation of antioxidant response element-driven gene expression. J Biol Chem 278:21592–21600. doi:10.1074/jbc.M300931200
Anderson ME (1998) Glutathione: an overview of biosynthesis and modulation. Chem Biol Interact 111–112:1–14
Acknowledgments
The financial supports by (i) the FINEP Research Grant “Rede Instituto Brasileiro de Neurociência (IBN-Net)” no. 01.06.0842-00, (ii) Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), (iii) Fundação de Apoio à Pesquisa do Estado de Santa Catarina (FAPESC-TO 1346/2010-3), (iv) Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and (v) INCT-CNPq-Excitotoxicity and Neuroprotection are gratefully acknowledged. M.F. is a recipient of the CNPq fellowship.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Figure S1
Effects of succinobucol treatment on Nrf-2 nuclear translocation in SH-SY5Y cells. Cells were treated with succinobucol (3 μM) or vehicle (DMSO 0.05 %) for 6 hours or 6 days. Tert-butylhydroquinone (TBQ) (10 μM) for 6 hours was used as a positive control. The nuclear translocation of Nrf-2 was evaluated by immunofluorescence (green). Nuclear counterstaining was made with Hoechst 33258 (blue). A representative merge of the two fluorescence images (green and blue) is shown (Magnification 100 ×). (GIF 126 kb)
Rights and permissions
About this article
Cite this article
Colle, D., Santos, D.B., Hartwig, J.M. et al. Succinobucol, a Lipid-Lowering Drug, Protects Against 3-Nitropropionic Acid-Induced Mitochondrial Dysfunction and Oxidative Stress in SH-SY5Y Cells via Upregulation of Glutathione Levels and Glutamate Cysteine Ligase Activity. Mol Neurobiol 53, 1280–1295 (2016). https://doi.org/10.1007/s12035-014-9086-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9086-x