Abstract
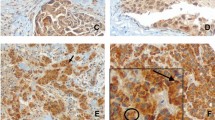
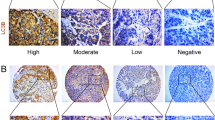
Microtubule-associated protein 1 light chain 3B (LC3B) was involved in autophagosome formation and had been as a marker of autophagy which played an important role in the development of breast cancer. The purpose of this study was to explore the level of LC3B expression in four stages of triple-negative breast cancer (TNBC) and to evaluate the prognostic significance of LC3B expression in TNBC. The ultimate aim was to identify the new factor that could be useful in predicting clinical behavior of TNBC. We evaluated the expression level of LC3B protein in four kinds of TNBC tissue samples, including intraductal carcinoma in situ (DCIS), invasive ductal carcinoma (IDC) without metastases, IDC with lymph node metastases (LNM), and IDC with distant metastases (DM). Hundred and four primary TNBC patients were involved in present study, and the expression level of LC3B protein was assessed by immunohistochemistry. Medical records of these patients were reviewed, and the clinicopathological analysis was performed. High expression of LC3B was observed in 7.7 % of DCIS (1 of 13 cases), 16.2 % of IDC (6 of 37 cases), 35.7 % of LNM (15 of 42 cases), and 58.3 % of DM (7 of 12 cases). LC3B high expression was significantly associated with tumor size (P = 0.028), lymph node metastasis (P = 0.002), and Ki-67 expression (P = 0.047). LC3B high expression patients showed poorer DFS and OS rates compared with LC3B low expression patients (P = 0.024, and P = 0.047, respectively). Multivariate analyses confirmed that high LC3B expression was an independent and significant factor for predicting the poor outcome of TNBC patients. These preliminary results demonstrated that high LC3B expression was associated with lymph node and distant metastasis. Furthermore, high LC3B protein expression was correlated with shorter survival in patients with triple-negative breast carcinoma. These findings provided preliminary evidence for the function of LC3B on the progression of TNBC and suggested LC3B was a useful marker in prognostic evaluation for patients with TNBC.



Similar content being viewed by others
References
Zhao J, Liu H, Wang M, et al. Characteristics and prognosis for molecular breast cancer subtypes in Chinese women. J Surg Oncol. 2009;100:89–94.
Reis-Filho JS, Tutt AN. Triple negative tumours: a critical review. Histopathology. 2008;52:108–18.
Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290:1717–21.
Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell. 2004;6:463–77.
Qu X, Yu J, Bhagat G, et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest. 2003;112:1809–20.
Kuma A, Mizushima N. Physiological role of autophagy as an intracellular recycling system: with an emphasis on nutrient metabolism. Semin Cell Dev Biol. 2010;21:683–90.
Degenhardt K, Mathew R, Beaudoin B, et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell. 2006;10:51–64.
Eisenberg-Lerner A, Kimchi A. The paradox of autophagy and its implication in cancer etiology and therapy. Apoptosis. 2009;14:376–91.
Fung C, Lock R, Gao S, et al. Induction of autophagy during extracellular matrix detachment promotes cell survival. Mol Biol Cell. 2008;19:797–806.
Karantza-Wadsworth Vassiliki, White Eileen. Role of autophagy in breast cancer. Autophagy. 2007;3(6):610–3.
Meijer Alfred J, Codogno Patrice. Regulation and role of autophagy in mammalian cells. Int J Biochem Cell Biol. 2004;36:2445–62.
Cheong H, Klionsky DJ. Biochemical methods to monitor autophagy-related processes in yeast. Methods Enzymol. 2008;451:1–26.
Tanida I, Waguri S. Measurement of autophagy in cells and tissues. Methods Mol Biol. 2010;648:193–214.
Fujii S, Mitsunaga S, Yamazaki M, et al. Autophagy is activated in pancreatic cancer cells and correlates with poor patient outcome. Cancer Sci. 2008;99:1813–9.
Sato K, Tsuchihara K, Fujii S, et al. Autophagy is activated in colorectal cancer cells and contributes to the tolerance to nutrient deprivation. Cancer Res. 2007;67:9677–84.
Tanida I. Autophagy basics. Microbiol Immunol. 2011;55:1–11.
Othman Ekhlas Qaid Gazem, Kaur Gurjeet, Mutee Ahmad Faisal, et al. Immunohistochemical Expression of MAP1LC3A and MAP1LC3B protein in breast carcinoma tissues. J Clin Lab Anal. 2009;23:249–58.
Goldhirsch A, Wood WC, Coates AS, et al. Strategies for subtypes-dealing with the diversity of breast cancer: highlights of the St Gallen international expert consensus on the primary therapy of early breast cancer, 2011. Ann Oncol. 2011;22(8):1736–47.
Lazova Rossitza, Camp Robert L, Klump Vincent, et al. Punctate LC3B expression is a common feature of solid tumors and associated with proliferation, metastasis, and poor outcome. Clin Cancer Res. 2012;18:370–9.
Klionsky DJ, Abeliovich H, Agostinis P. Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy. 2008;4:151–75.
He H, Dang Y, Dai F, et al. Post-translational modifications of three members of the human MAP1LC3 family and detection of a novel type of modification for MAP1LC3B. J Biol Chem. 2003;278:29278–87.
Czaja MJ. Functions of autophagy in hepatic and pancreatic physiology and disease. Gastroenterology. 2011;140:1895–908.
Liu M, Lawson G, Delos M, et al. Predictive value of the fraction of cancer cells immunolabeled for proliferating cell nuclear antigen or Ki-67 in biopsies of head and neck carcinomas to identify lymph node metastasis: comparison with clinical and radiologic examinations. Head Neck. 2003;25(4):280–8.
Yoshioka A, Miyata H, Doki Y, et al. LC3, an autophagosome marker, is highly expressed in gastrointestinal cancers. Int J Oncol. 2008;33:461–8.
Sivridis E, Giatromanolaki A, Liberis V, et al. Autophagy in endometrial carcinomas and prognostic relevance of “stone-like” structures (SLS). What is destined for the atypical endo metrial hyperplasia? Autophagy. 2011;7(1):74–82.
Giatromanolaki A, Koukourakis MI, Harris AL, et al. Prognostic relevance of light chain 3 (LC3A) autophagy patterns in colorectal adenocarcinomas. J Clin Pathol. 2010;63(10):867–72.
Sivridis E, Giatromanolaki A, Karpathiou G, et al. LC3A-positive “stone-like” structures in cutaneous squamous cell carcinomas. Am J Dermatopathol. 2011;33(3):285–90.
Aoki H, Kondo Y, Aldape K, et al. Monitoring autophagy in glioblastoma with antibody against isoform B of human microtubule-associated protein 1 light chain 3. Autophagy. 2008;4(4):467–75.
Acknowledgments
This experiment was finished in the Oncobiology Key Laboratory of the Heilongjiang Province Common Institution of Higher Learning. This work is supported by a grant from the start-up foundation for scientific research of the Third Affiliated Hospital of Harbin Medical University (JJZ2011-13).
Conflict of interest
All authors declared that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hong Zhao and Maopeng Yang contributed equally to the work and should be considered co-first authors.
Rights and permissions
About this article
Cite this article
Zhao, H., Yang, M., Zhao, J. et al. High expression of LC3B is associated with progression and poor outcome in triple-negative breast cancer. Med Oncol 30, 475 (2013). https://doi.org/10.1007/s12032-013-0475-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0475-1