Abstract
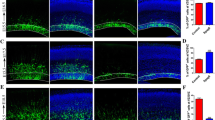
Sperm-associated antigen 6 (Spag6) is a Chlamydomonas reinhardtii PF16 homologous gene detected in the human testis and is crucial for sperm motility. Neuronal migration is a dynamic process requiring coordinated cytoskeletal remodeling, and Spag6 is co-localized with microtubules in Chinese hamster ovary cells and COS-1 cells. However, the role of Spag6 in neuronal migration remains unclear. Here, we demonstrated that Spag6 was continuously expressed in the developing cerebral cortex. Using in utero electroporation (IUE), we found that overexpression of Spag6 delayed the rate of neuronal migration, rather than affecting the ultimate fate of cortical neurons. Furthermore, overexpression of Spag6 caused a significant decrease in neurite number and length of cortical neurons. Our results indicated that Spag6 controlled neuronal migration as well as neurite branching and elongation.





Similar content being viewed by others
References
Audesirk G, Cabell L, Kern M (1997) Modulation of neurite branching by protein phosphorylation in cultured rat hippocampal neurons. Brain Res Dev Brain Res 102:247–260
Chai X, Fan L, Shao H et al. (2014) Reelin induces branching of neurons and radial glial cells during corticogenesis. Cereb Cortex
Frotscher M, Chai X, Bock HH, Haas CA, Forster EZhao S (2009) Role of Reelin in the development and maintenance of cortical lamination. J Neural Transm 116:1451–1455
Hamada T, Teraoka M, Imaki J, Ui-Tei K, Ladher R, KAsahara T (2010) Gene expression of Spag6 in chick central nervous system. Anat Histol Embryol 39:227–232
Inglis-Broadgate SL, Thomson RE, Pellicano F et al (2005) FGFR3 regulates brain size by controlling progenitor cell proliferation and apoptosis during embryonic development. Dev Biol 279:73–85
Kwan KY, Sestan N, Anton ES (2012) Transcriptional co-regulation of neuronal migration and laminar identity in the neocortex. Development 139:1535–1546
Liu JS (2011) Molecular genetics of neuronal migration disorders. Curr Neurol Neurosci Rep 11:171–178
Liu Z, Steward R, Luo L (2000) Drosophila Lis1 is required for neuroblast proliferation, dendritic elaboration and axonal transport. Nat Cell Biol 2:776–783
Moon HM, Wynshaw-Boris A (2013) Cytoskeleton in action: lissencephaly, a neuronal migration disorder. Wiley Interdiscip Rev Dev Biol 2:229–245
Neilson LI, Schneider PA, Van Deerlin PG et al (1999) cDNA cloning and characterization of a human sperm antigen (SPAG6) with homology to the product of the Chlamydomonas PF16 locus. Genomics 60:272–280
Reiner O, Carrozzo R, Shen Y et al (1993) Isolation of a Miller-Dieker lissencephaly gene containing G protein beta-subunit-like repeats. Nature 364:717–721
Sapir T, Elbaum M, Reiner O (1997) Reduction of microtubule catastrophe events by LIS1, platelet-activating factor acetylhydrolase subunit. EMBO J 16:6977–6984
Sapir T, Shmueli A, Levy T et al (2008) Antagonistic effects of doublecortin and MARK2/Par-1 in the developing cerebral cortex. J Neurosci 28:13008–13013
Sapiro R, Tarantino LM, Velazquez F et al (2000) Sperm antigen 6 is the murine homologue of the Chlamydomonas reinhardtii central apparatus protein encoded by the PF16 locus. Biol Reprod 62:511–518
Sapiro R, Kostetskii I, Olds-Clarke P, Gerton GL, Radice G, LStrauss IJ (2002) Male infertility, impaired sperm motility, and hydrocephalus in mice deficient in sperm-associated antigen 6. Mol Cell Biol 22:6298–6305
Sasaki S, Shionoya A, Ishida M et al (2000) A LIS1/NUDEL/cytoplasmic dynein heavy chain complex in the developing and adult nervous system. Neuron 28:681–696
Slepak TI, Salay LD, Lemmon V, PBixby JL (2012) Dyrk kinases regulate phosphorylation of doublecortin, cytoskeletal organization, and neuronal morphology. Cytoskeleton (Hoboken) 69:514–527
Smith EF, Lefebvre PA (1996) PF16 encodes a protein with armadillo repeats and localizes to a single microtubule of the central apparatus in Chlamydomonas flagella. J Cell Biol 132:359–370
Smith EF, Lefebvre PA (1997) The role of central apparatus components in flagellar motility and microtubule assembly. Cell Motil Cytoskeleton 38:1–8
Smith EF, Lefebvre PA (2000) Defining functional domains within PF16: a central apparatus component required for flagellar motility. Cell Motil Cytoskeleton 46:157–165
Tabata H, Nakajima K (2001) Efficient in utero gene transfer system to the developing mouse brain using electroporation: visualization of neuronal migration in the developing cortex. Neuroscience 103:865–872
Tanaka T, Serneo FF, Higgins C, Gambello MJ, Wynshaw-Boris AGleeson JG (2004) Lis1 and doublecortin function with dynein to mediate coupling of the nucleus to the centrosome in neuronal migration. J Cell Biol 165:709–721
van Ooyen A, Carnell A, de Ridder S et al (2014) Independently outgrowing neurons and geometry-based synapse formation produce networks with realistic synaptic connectivity. PLoS One 9, e85858
Weimer JM, Yokota Y, Stanco A, Stumpo DJ, Blackshear P, JAnton ES (2009) MARCKS modulates radial progenitor placement, proliferation and organization in the developing cerebral cortex. Development 136:2965–2975
Wu Q, Liu J, Fang A et al (2014) The dynamics of neuronal migration. Adv Exp Med Biol 800:25–36
Zhang Z, Sapiro R, Kapfhamer D et al (2002) A sperm-associated WD repeat protein orthologous to Chlamydomonas PF20 associates with Spag6, the mammalian orthologue of Chlamydomonas PF16. Mol Cell Biol 22:7993–8004
Zhang Z, Jones BH, Tang W et al (2005) Dissecting the axoneme interactome: the mammalian orthologue of Chlamydomonas PF6 interacts with sperm-associated antigen 6, the mammalian orthologue of Chlamydomonas PF16. Mol Cell Proteomics 4:914–923
Acknowledgments
This work was supported by the Foundation for Special Talent of Nwsuaf (No. Z111021101) and the Foundation for Basal Research of Nwsuaf (2014YB015). We thank Parks Martin for critical proofreading of the manuscript.
Conflict of Interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Runchuan Yan and Xinde Hu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yan, R., Hu, X., Zhang, Q. et al. Spag6 Negatively Regulates Neuronal Migration During Mouse Brain Development. J Mol Neurosci 57, 463–469 (2015). https://doi.org/10.1007/s12031-015-0608-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0608-4