Abstract
Background
Hyper- and hyponatremia are frequently observed in patients after subarachnoidal hemorrhage, and are potentially related to worse outcome. We hypothesized that the fluid regimen in these patients is associated with distinct changes in serum electrolytes, acid–base disturbances, and fluid balance.
Methods
Thirty-six consecutive patients with SAH were randomized double-blinded to either normal saline and hydroxyethyl starch dissolved in normal saline (Voluven®; saline) or balanced crystalloid and colloid solutions (Ringerfundin® and Tetraspan®; balanced, n = 18, each) for 48 h. Laboratory samples and fluid balance were evaluated at baseline and at 24 and 48 h.
Results
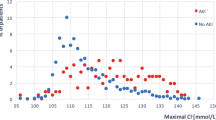
Age [57 ± 13 years (mean ± SD; saline) vs. 56 ± 12 years (balanced)], SAPS II (38 ± 16 vs. 34 ± 17), Hunt and Hess [3 (1–4) (median, range) vs. 2 (1–4)], and Fischer scores [3.5 (1–4) vs. 3.5 (1–4)] were similar. Serum sodium, chloride, and osmolality increased in saline only (p ≤ 0.010, time–group interaction). More patients in saline had Cl >108 mmol/L [16 (89 %) vs. 8 (44 %); p = 0.006], serum osmolality >300 mosmol/L [10 (56 %) vs. 2 (11 %); p = 0.012], a base excess <−2 [12 (67 %) vs. 2 (11 %); p = 0.001], and fluid balance >1,500 mL during the first 24 h [11 (61 %) vs. 5 (28 %); p = 0.046]. Hyponatremia and hypo-osmolality were not more frequent in the balanced group.
Conclusions
Treatment with saline-based fluids resulted in a greater number of patients with hyperchloremia, hyperosmolality, and positive fluid balance >1,500 mL early after SAH, while administration of balanced solutions did not cause more frequent hyponatremia or hypo-osmolality. These results should be confirmed in larger studies.





Similar content being viewed by others
References
Berendes E, Walter M, Cullen P, et al. Secretion of brain natriuretic peptide in patients with aneurysmal subarachnoid haemorrhage. Lancet. 1997;349:245–9.
Espiner EA, Leikis R, Ferch RD, et al. The neuro–cardio–endocrine response to acute subarachnoid haemorrhage. Clin Endocrinol (Oxf). 2002;56:629–35.
Wartenberg KE, Schmidt JM, Claassen J, et al. Impact of medical complications on outcome after subarachnoid hemorrhage. Crit Care Med. 2006;34:617–23.
Hasan D, Wijdicks EF, Vermeulen M. Hyponatremia is associated with cerebral ischemia in patients with aneurysmal subarachnoid hemorrhage. Ann Neurol. 1990;27:106–8.
Qureshi AI, Suri MF, Sung GY, et al. Prognostic significance of hypernatremia and hyponatremia among patients with aneurysmal subarachnoid hemorrhage. Neurosurgery. 2002;50:749–55.
Mandal AK, Saklayen MG, Hillman NM, Markert RJ. Predictive factors for high mortality in hypernatremic patients. Am J Emerg Med. 1997;15:130–2.
Bennani SL, Abouqal R, Zeggwagh AA, et al. Incidence, causes and prognostic factors of hyponatremia in intensive care. Rev Med Interne. 2003;24:224–9.
Lee CT, Guo HR, Chen JB. Hyponatremia in the emergency department. Am J Emerg Med. 2000;18:264–8.
Tommasino C, Moore S, Todd MM. Cerebral effects of isovolemic hemodilution with crystalloid or colloid solutions. Crit Care Med. 1988;16:862–8.
Zornow MH, McQuitty C, Prough DS. Perioperative fluid management of the neurosurgical patient. In: Albin MS, editor. Textbook of neuroanesthesia with neurosurgical and neuroscience perspectives. New York: McGraw-Hill; 1997. p. 117–48.
Williams EL, Hildebrand KL, McCormick SA, Bedel MJ. The effect of intravenous lactated Ringer’s solution versus 0.9 % sodium chloride solution on serum osmolality in human volunteers. Anesth Analg. 1999;88:999–1003.
O’Malley CM, Frumento RJ, Hardy MA, et al. A randomized, double-blind comparison of lactated Ringer’s solution and 0.9 % NaCl during renal transplantation. Anesth Analg. 2005;100:1518–24.
Wilkes NJ, Woolf R, Mutch M, et al. The effects of balanced versus saline-based hetastarch and crystalloid solutions on acid–base and electrolyte status and gastric mucosal perfusion in elderly surgical patients. Anesth Analg. 2001;93:811–6.
Van Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, van Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke. 1988;19:604–7.
Sayama T, Inamura T, Matsushima T, Inoha S, Inoue T, Fukui M. High incidence of hyponatremia in patients with ruptured anterior communicating artery aneurysms. Neurol Res. 2000;22:151–5.
Wijdicks EF, Vermeulen M, ten Haaf JA, Hijdra A, Bakker WH, van Gijn J. Volume depletion and natriuresis in patients with a ruptured intracranial aneurysm. Ann Neurol. 1985;18:211–6.
Diringer MN, Wu KC, Verbalis JG, Hanley DF. Hypervolemic therapy prevents volume contraction but not hyponatremia following subarachnoid hemorrhage. Ann Neurol. 1992;31:543–50.
Solomon RA, Post KD, McMurtry JG 3rd. Depression of circulating blood volume in patients after subarachnoid hemorrhage: implications for the management of symptomatic vasospasm. Neurosurgery. 1984;15:354–61.
Hasan D, Lindsay KW, Wijdicks EF, et al. Effect of fludrocortisone acetate in patients with subarachnoid hemorrhage. Stroke. 1989;20:1156–61.
Audibert G, Steinmann G, de Talancé N, et al. Endocrine response after severe subarachnoid hemorrhage related to sodium and blood volume regulation. Anesth Analg. 2009;108:1922–8.
Bederson JB, Connolly ES Jr, Batjer HH, American Heart Association, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke. 2009;40:994–1025.
Diringer M, Bleck T, Claude Hemphill J, et al. Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s multidisciplinary consensus conference. Neurocrit Care. 2011;15:211–40.
Isotani E, Suzuki R, Tomita K, et al. Alterations in plasma concentrations of natriuretic peptides and antidiuretic hormone after subarachnoid hemorrhage. Stroke. 1994;25:2198–203.
Kurokawa Y, Uede T, Ishiguro M, et al. Pathogenesis of hyponatremia following subarachnoid hemorrhage due to ruptured cerebral aneurysm. Surg Neurol. 1996;46:500–7.
Wijdicks EF, Schievink WI, Burnett JC Jr. Natriuretic peptide system and endothelin in aneurysmal subarachnoid hemorrhage. J Neurosurg. 1997;87:275–80.
Moro N, Katayama Y, Kojima J, Mori T, Kawamata T. Prophylactic management of excessive natriuresis with hydrocortisone for efficient hypervolemic therapy after subarachnoid hemorrhage. Stroke. 2003;34:2807–11.
Katayama Y, Haraoka J, Hirabayashi H, et al. A randomized controlled trial of hydrocortisone against hyponatremia in patients with aneurysmal subarachnoid hemorrhage. Stroke. 2007;38:2373–5.
Fukui S, Otani N, Katoh H, et al. Female gender as a risk factor for hypokalemia and QT prolongation after subarachnoid hemorrhage. Neurology. 2002;59:134–6.
Fukui S, Katoh H, Tsuzuki N, et al. Multivariate analysis of risk factors for QT prolongation following subarachnoid hemorrhage. Crit Care. 2003;7:R7–12.
Brown MJ, Brown DC, Murphy MB. Hypokalemia from beta2-receptor stimulation by circulating epinephrine. N Engl J Med. 1983;309:1414–9.
Mayer SA, Lin J, Homma S, et al. Myocardial injury and left ventricular performance after subarachnoid hemorrhage. Stroke. 1999;30:780–6.
Acknowledgments
We are grateful for skillful assistance by the research nurses Judith Erni-Kaufmann, Natalie Araya-Araya, Michael Lensch, Konrad Torsten, and for careful editing of the manuscript by Jeannie Wurz, medical writer. Supported by an unrestricted grant from BBraun Medical AG, Sempach, Switzerland.
Conflict of interest
The Department of Intensive Care Medicine has, or has had in the past, research contracts with Abbott Nutrition International, CSEM SA, Edwards Lifesciences Services GmbH, Kenta Biotech Ltd, Maquet Critical Care AB, Omnicare Clinical Research AG, and Orion Corporation; and research & development/consulting contracts with Edwards Lifesciences SA, Maquet Critical Care AB, and Néstle. The money is/was paid into a departmental fund; no author receives/received any personal financial gain. The department has received unrestricted educational grants from the following organizations for organizing a quarterly postgraduate educational symposium, the Berner Forum for Intensive Care: Fresenius Kabi; gsk; MSD; Lilly; Baxter; astellas; AstraZeneca; B|Braun; CSL Behring; Maquet; Novartis; Covidien; Mycomed; RobaPharma.
Author information
Authors and Affiliations
Corresponding author
Additional information
Registered with the Deutsches Register Klinischer Studien, trial number DRKS000034.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lehmann, L., Bendel, S., Uehlinger, D.E. et al. Randomized, Double-Blind Trial of the Effect of Fluid Composition on Electrolyte, Acid–Base, and Fluid Homeostasis in Patients Early After Subarachnoid Hemorrhage. Neurocrit Care 18, 5–12 (2013). https://doi.org/10.1007/s12028-012-9764-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-012-9764-3