Abstract
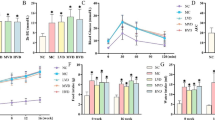
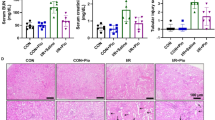
We investigated whether 1,25-dihydroxy-vitamin D3 (1,25(OH)2D3) could improve early diabetic nephropathy through the DNA-damage-inducible transcript 4/tuberous sclerosis 2/mammalian target of rapamycin pathway. Rat mesangial cells were cultured in media containing normal glucose or high glucose and were treated with or without 1,25(OH)2D3. Mesangial cells proliferation was measured. Streptozotocin-induced diabetic rats were injected intravenously with a recombinant lentivirus against the rat vitamin D receptor gene. Urinary and serum albumin, fasting plasma glucose, serum triglyceride, total cholesterol, calcium, parathyroid hormone and serum 25-dihydroxy-vitamin D (25(OH)D) levels, mean glomerular volume, glomerular basement membrane thickness and total kidney volume were determined. The expressions of vitamin D receptor, DNA-damage-inducible transcript 4, and mammalian target of rapamycin were measured. 1,25(OH)2D3 inhibited the proliferation of mesangial cells induced by hyperglycemia. 1,25(OH)2D3 also significantly reduced albumin excretion, mean glomerular volume, glomerular basement membrane, and total kidney volume in rats with diabetic nephropathy. The expression of DNA-damage-inducible transcript 4 was elevated by 1,25(OH)2D3 treatment. The phosphorylation of mammalian target of rapamycin was reduced by 1,25(OH)2D3 treatment. Vitamin D receptor gene silencing blocked all of the above results. The current study demonstrates that 1,25(OH)2D3 can effectively inhibit mesangial cells proliferation induced by hyperglycemia, thus suppressing the development of diabetic nephropathy. This study also shows that the nephron-protective effect of 1,25(OH)2D3 occurs partly through the DDIT4/TSC2/mTOR pathway.





Similar content being viewed by others
References
U. Krairittichai, S. Potisat, A. Jongsareejit, C. Sattaputh, Prevalence and risk factors of diabetic nephropathy among Thai patients with type 2 diabetes mellitus. J. Med. Assoc. Thai. 94 (2011)
F.P. Schena, L. Gesualdo, Pathogenetic mechanisms of diabetic nephropathy. J. Am. Soc. Nephrol. 16 (2005)
M.W. Steffes, R. Østerby, B. Chavers, S.M. Mauer, Mesangial expansion as a central mechanism for loss of kidney function in diabetic patients. Diabetes 38, 1077–1081 (1989)
S.Y. Goh, M.E. Cooper, Clinical review: The role of advanced glycation end products in progression and complications of diabetes. J. Clin. Endocrinol. Metab. 93(4), 1143 (2008)
K. Fukami, S. Ueda, S. Yamagishi, S. Kato, Y. Inagaki, M. Takeuchi, Y. Motomiya, R. Bucala, S. Iida, K. Tamaki, T. Imaizumi, M.E. Cooper, S. Okuda, AGEs activate mesangial TGF-beta-Smad signaling via an angiotensin II type I receptor interaction. Kidney Int. 66(6), 2137–2147 (2004)
R. Yacoub, K.N. Campbell, Inhibition of RAS in diabetic nephropathy. Int. J. Nephrol. Renovasc. Dis. 8, 29–40 (2015)
B. Fernandez-Fernandez, A. Ortiz, C. Gomez-Guerrero, J. Egido, Therapeutic approaches to diabetic nephropathy–beyond the RAS. Nat. Rev. Nephrol. 10(6), 325–346 (2014)
K. Inoki, M. Haneda, S. Maeda, D. Koya, R. Kikkawa, TGF-β1 stimulates glucose uptake by enhancing GLUT1 expression in mesangial cells. Kidney Int. 55(5), 1704–1712 (1999)
S. Di Paolo, L. Gesualdo, E. Ranieri, G. Grandaliano, F.P. Schena, High glucose concentration induces the overexpression of transforming growth factor-beta through the activation of a platelet-derived growth factor loop in human mesangial cells. Am. J. Pathol. 149(6), 2095–2106 (1996)
S.B. Connolly, D. Sadlier, N.E. Kieran, P. Doran, H.R. Brady, Transcriptome profiling and the pathogenesis of diabetic complications. J. Am. Soc. Nephrol. 14, S279–S283 (2003)
J.M. Forbes, M.T. Coughlan, M.E. Cooper, Oxidative stress as a major culprit in kidney disease in diabetes. Diabetes 57(6), 1446–1454 (2008)
Z. Wang, T. Jiang, J. Li, G. Proctor, J.L. McManaman, S. Lucia, S. Chua, M. Levi, Regulation of renal lipid metabolism, lipid accumulation, and glomerulosclerosis in FVBdb/db mice with type 2 diabetes. Diabetes 54(8), 2328–2335 (2005)
D. Dubois, P. Chanson, J. Timsit, D. Chauveau, D. Nochy, P.J. Guillausseau, J. Lubetzki, Remission of proteinuria following correction of hyperlipidemia in NIDDM patients with nondiabetic glomerulopathy. Diabetes Care 17(8), 906–908 (1994)
M.K. Lu, X.G. Gong, K.L. Guan, mTOR in podocyte function: is rapamycin good for diabetic nephropathy?. Cell Cycle 10(20), 3415–3416 (2011)
K. Nagai, T. Matsubara, A. Mima, E. Sumi, H. Kanamori, N. Iehara, A. Fukatsu, M. Yanagita, T. Nakano, Y. Ishimoto, T. Kita, T. Doi, H. Arai, Gas6 induces Akt/mTOR-mediated mesangial hypertrophy in diabetic nephropathy. Kidney Int. 68(2), 552–561 (2005)
R. Zoncu, A. Efeyan, D.M. Sabatini, mTOR: from growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 12(1), 21–35 (2011)
H. Mori, K. Inoki, K. Masutani, Y. Wakabayashi, K. Komai, R. Nakagawa, K.L. Guan, A. Yoshimura, The mTOR pathway is highly activated in diabetic nephropathy and rapamycin has a strong therapeutic potential. Biochem. Biophys. Res. Commun. 384(4), 471–475 (2009)
A. Eisenreich, U. Rauch, PI3K inhibitors in cardiovascular disease. Cardiovasc. Ther. 29(1), 29–36 (2011)
W. Lieberthal, J.S. Levine, The role of the mammalian target of rapamycin (mTOR) in renal disease. J. Am. Soc. Nephrol. 20(12), 2493–2502 (2009)
Y. Yang, J. Wang, L. Qin, Z. Shou, J. Zhao, H. Wang, Y. Chen, J. Chen, Rapamycin prevents early steps of the development of diabetic nephropathy in rats. Am. J. Nephrol. 27(5), 495–502 (2007)
S. Langer, R. Kreutz, A. Eisenreich, Metformin modulates apoptosis and cell signaling of human podocytes under high glucose conditions. J. Nephrol. (2016) [Epub ahead of print 5 Jan]
M. Fraenkel, M. Ketzinel-Gilad, Y. Ariav, O. Pappo, M. Karaca, J. Castel, M.F. Berthault, C. Magnan, E. Cerasi, N. Kaiser, G. Leibowitz, mTOR inhibition by rapamycin prevents beta-cell adaptation to hyperglycemia and exacerbates the metabolicstate in type 2 diabetes. Diabetes 57(4), 945–957 (2008)
M.C. Borges, L.A. Martini, M.M. Rogero, Current perspectives on vitamin D, immune system, and chronic diseases. Nutrition 27(4), 399–404 (2011)
T.S. Lisse, T. Liu, M. Irmler, J. Beckers, H. Chen, J.S. Adams, M. Hewison, Gene targeting by the vitamin D response element binding protein reveals a role for vitamin D in osteoblast mTORsignaling. FASEB J. 25(3), 937–947 (2011)
T.S. Lisse, M. Hewison, Vitamin D: a new player in the world of mTOR signaling. Cell Cycle 10(12), 1888–1889 (2011)
V.A. Diaz, A.G. Mainous 3rd, P.J. Carek, A.M. Wessell, C.J. Everett, The association of vitamin D deficiency and insufficiency with diabetic nephropathy: implications for healthdisparities. J. Am. Board Fam. Med. 22(5), 521–527 (2009)
A. Levin, M. Le Barbier, L. Er, D. Andress, M.K. Sigrist, O. Djurdjev, Incident isolated 1,25(OH)(2)D(3) deficiency is more common than 25(OH)D deficiency in CKD. J. Nephrol. 25(2), 204–210 (2012)
Nirmalya Dey, Nandini Ghosh-Choudhury, Falguni Das, Xiaonan Li, Balachandar Venkatesan, Jeffrey L. Barnes, Balakuntalam S. Kasinath, Goutam Ghosh Choudhury, PRAS40 acts as a nodal regulator of high glucose-induced TORC1 activation in glomerular mesangial cell hypertrophy. J. Cell Physiol. 225(1), 27–41 (2010)
S.I. Jeong, D.H. Kwak, S. Lee, Y.K. Choo, W.H. Woo, K.S. Keum, B.K. Choi, K.Y. Jung, Inhibitory effects of Cnidium officinale Makino and Tabanus fulvus Meigan on the high glucose-induced proliferation of glomerular mesangial cells. Phytomedicine 12(9), 648–655 (2005)
Kojiro Nagai, Takeshi Matsubara, Akira Mima, Eriko Sumi, Hiroshi Kanamori et al. Gas6 induces Akt/mTOR-mediated mesangial hypertrophy in diabetic nephropathy. Kidney Int 68(2), 552–561 (2005)
T.W. Lee, Y.H. Kao, T.I. Lee, C.J. Chang, G.S. Lien, Y.J. Chen, Calcitriol modulates receptor for advanced glycation end products (RAGE) in diabetic hearts. Int. J. Cardiol. 173(2), 236–241 (2014)
G.Y. Oudit, G.C. Liu, J. Zhong, R. Basu, F.L. Chow, J. Zhou, H. Loibner, E. Janzek, M. Schuster, J.M. Penninger, A.M. Herzenberg, Z. Kassiri, J.W. Scholey, Human recombinant ACE2 reduces the progression of diabetic nephropathy. Diabetes 59(2), 529–538 (2010)
S. Okada, S. Inaga, Y. Kawaba, T. Hanada, A. Hayashi, H. Nakane, T. Naguro, T. Kaidoh, S. Kanzaki, A novel approach to the histological diagnosis of pediatric nephrotic syndrome by low vacuum scanning electronmicroscopy. Biomed. Res. 35(4), 227–236 (2014)
D. Sankaran, N. Bankovic-Calic, C.Y. Peng, M.R. Ogborn, H.M. Aukema, Dietary flax oil during pregnancy and lactation retards disease progression in rat offspring with inherited kidneydisease. Pediatr. Res. 60(6), 729–733 (2006)
R.R. Singh, L.A. Cullen-McEwen, M.M. Kett, W.M. Boon, J. Dowling, J.F. Bertram, K.M. Moritz, Prenatal corticosterone exposure results in altered AT1/AT2, nephron deficit and hypertension in the rat offspring. J. Physiol. 579, 503–513 (2007)
Z. Yang, F. Liu, H. Qu, H. Wang, X. Xiao, H. Deng, 1, 25(OH)2D3 protects β cell against high glucose-induced apoptosis through mTOR suppressing. Mol. Cell Endocrinol. 414, 111–119 (2015)
H. Derakhshanian, S. Shab-Bidar, J.R. Speakman, H. Nadimi, K. Djafarian, Vitamin D and diabetic nephropathy: A systematic review and meta-analysis. Nutrition 31(10), 1189–1194 (2015)
D.K. Deb, Y. Chen, Z. Zhang, Y. Zhang, F.L. Szeto, K.E. Wong, J. Kong, Y.C. Li, 1,25-Dihydroxyvitamin D3 suppresses high glucose-induced angiotensinogen expression in kidney cells byblocking the NF-{kappa}B pathway. Am. J. Physiol. Renal. Physiol. 296(5), F1212–F1218 (2009)
Z. Zhang, W. Yuan, L. Sun, F.L. Szeto, K.E. Wong, X. Li, J. Kong, Y.C. Li, 1,25-Dihydroxyvitamin D3 targeting of NF-kappaB suppresses high glucose-induced MCP-1 expression in mesangial cells. Kidney Int. 72(2), 193–201 (2007)
A.K. Bhalla, E.P. Amento, B. Serog, L.H. Glimcher, 1,25-Dihydroxyvitamin D3 inhibits antigen-induced T cell activation. J. Immunol. 133(4), 1748–1754 (1984)
M. Kajikawa, H. Ishida, S. Fujimoto, E. Mukai, M. Nishimura, J. Fujita, Y. Tsuura, Y. Okamoto, A.W. Norman, Y. Seino, An insulinotropic effect of vitamin D analog with increasing intracellular Ca2+ concentration in pancreatic beta-cells through nongenomic signal transduction. Endocrinology 140(10), 4706–4712 (1999)
A. Cardus, S. Panizo, M. Encinas, X. Dolcet, C. Gallego, M. Aldea, E. Fernandez, J.M. Valdivielso, 1,25-dihydroxyvitamin D3 regulates VEGF production through a vitamin D response element in the VEGFpromoter. Atherosclerosis 204(1), 85–89 (2009)
M. Garsen, R. Sonneveld, A.L. Rops, S. Huntink, T.H. van Kuppevelt, T.J. Rabelink, J.G. Hoenderop, J.H. Berden, T. Nijenhuis, J. van der Vlag, Vitamin D attenuates proteinuria by inhibition of heparanase expression in the podocyte. J. Pathol 237(4), 472–481 (2015)
X.X. Wang, T. Jiang, Y. Shen, H. Santamaria, N. Solis, C. Arbeeny, M. Levi, Vitamin D receptor agonist doxercalciferol modulates dietary fat-induced renal disease and renal lipid metabolism. Am. J. Physiol. Renal Physiol. 300(3), F801–F810 (2011)
A. Kuhlmann, C.S. Haas, M.L. Gross, U. Reulbach, M. Holzinger, U. Schwarz, E. Ritz, K. Amann, 1,25-Dihydroxyvitamin D3 decreases podocyte loss and podocyte hypertrophy in the subtotally nephrectomizedrat. Am. J. Physiol. Renal Physiol. 286(3), F526–F533 (2004)
F. Marampon, G.L. Gravina, C. Festuccia, V.M. Popov, E.A. Colapietro, P. Sanità, D. Musio, F. De Felice, A. Lenzi, E.A. Jannini, E. Di Cesare, V. Tombolini, Vitamin D protects endothelial cells from irradiation-induced senescence and apoptosis by modulating MAPK/SirT1 axis. J. Endocrinol Invest 39(4), 411–422 (2015)
Y. Zhang, D.K. Deb, J. Kong, G. Ning, Y. Wang, G. Li, Y. Chen, Z. Zhang, S. Strugnell, Y. Sabbagh, C. Arbeeny, Y.C. Li, Long-term therapeutic effect of vitamin D analog doxercalciferol on diabetic nephropathy: strong synergism with AT1 receptor antagonist. Am. J. Physiol. Renal Physiol. 297(3), F791–F801 (2009)
M. Fraenkel, M. Ketzinel-Gilad, Y. Ariav, O. Pappo, M. Karaca, J. Castel, M.F. Berthault, C. Magnan, E. Cerasi, N. Kaiser, G. Leibowitz, mTOR inhibition by rapamycin prevents beta-cell adaptation to hyperglycemia and exacerbates the metabolic state in type 2 diabetes. Diabetes 57(4), 945–957 (2008)
O.A. Adegoke, H.E. Bates, M.A. Kiraly, M. Vranic, M.C. Riddell, E.B. Marliss, Exercise in ZDF rats does not attenuate weight gain, but prevents hyperglycemia concurrent with modulation of amino acid metabolism and AKT/mTOR activation in skeletal muscle. Eur. J. Nutr. 54(5), 751–759 (2015)
B.A. Heller, M. Ghidinelli, J. Voelkl, S. Einheber, R. Smith, E. Grund, G. Morahan, D. Chandler, L. Kalaydjieva, F. Giancotti, R.H. King, A.N. Fejes-Toth, G. Fejes-Toth, M.L. Feltri, F. Lang, J.L. Salzer, Functionally distinct PI 3-kinase pathways regulate myelination in the peripheral nervous system. J. Cell Biol 204(7), 1219–1236 (2014)
Y. Weisman, A. Harell, S. Edelstein, M. David, Z. Spirer, A. Golander, 1 alpha, 25-Dihydroxyvitamin D3 and 24,25-dihydroxyvitamin D3 in vitro synthesis by human decidua andplacenta. Nature 281(5729), 317–319 (1979)
J.L. Omdahl, H.A. Morris, B.K. May, Hydroxylase enzymes of the vitamin D pathway: expression, function, and regulation. Annu. Rev. Nutr. 22, 139–166 (2002)
Acknowledgments
This study was supported by research grants from the National Nature Science Foundation of China (81070639, 81270911, 30771038, and 30570744) and National Key Clinical Specialties Construction Program of China. We specially thank the technical support from Laboratory of Lipid and Glucose Metabolism, the First Affiliated Hospital of Chongqing Medical University, Chongqing, P.R. China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, H., Wang, J., Qu, H. et al. In vitro and in vivo inhibition of mTOR by 1,25-dihydroxyvitamin D3 to improve early diabetic nephropathy via the DDIT4/TSC2/mTOR pathway. Endocrine 54, 348–359 (2016). https://doi.org/10.1007/s12020-016-0999-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-0999-1