Abstract
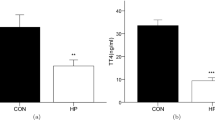
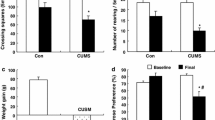
The purpose of this study was to investigate the depression-like behavior performances of subclinical hypothyroidism (SCH) rat. SCH rat model was induced by hemi-thyroid electrocauterization, and the behavior performances were measured by sucrose preference test, force swimming test (FST), and tail suspension test (TST). SCH rat model was established successfully by hemi-thyroid electrocauterization. In the behavior tasks, SCH rats displayed depression-like behavior were indicated as a significant elevation of immobility time in both the TST and FST, though the sucrose preference was not significantly decreased. The index of left adrenal cortex in both SCH and clinical hypothyroidism (CH) group significantly increased, and many large lipid vacuoles were observed in the zona fasciculata cells. The serum corticosterone concentration and hypothalamic corticotropin-releasing hormone mRNA expression 2 h after behavior test was markedly up-regulated in CH rats, but not SCH rats, indicated that SCH induced a less impairment of HPA axis than CH did. The important finding of this study was that the concentration of hippocampal T3 was lower in SCH group than that of the sham group. Furthermore, the results of Pearson correlation test showed that the immobility behaviors in TST and FST were both negatively correlated with hippocampal T3 concentration. Taking together, our results indicated that SCH could result in depression-like behavior, accompanied with subtle hyperactivity of HPA axis. The reduced hippocampal T3 prior to the reduction of thyroid hormone in serum might be taken as an early sign of hippocampus impairment in the progression from SCH to CH.


Similar content being viewed by others
References
J.M. de Guimaraes, C. Souza Lopes, J. Baima, R. Sichieri, Depression symptoms and hypothyroidism in a population-based study of middle-aged Brazilian women. J. Affect. Disord. 117(1–2), 120–123 (2009)
M.S. Gold, A.L. Pottash, I. Extein, Hypothyroidism and depression. Evidence from complete thyroid function evaluation. JAMA 245(19), 1919–1922 (1981)
D.F. Zhu, Z.X. Wang, D.R. Zhang, Z.L. Pan, S. He, X.P. Hu, X.C. Chen, J.N. Zhou, fMRI revealed neural substrate for reversible working memory dysfunction in subclinical hypothyroidism. Brain 129(Pt 11), 2923–2930 (2006)
E. Taskin, A.S. Artis, S. Bitiktas, N. Dolu, N. Liman, C. Suer, Experimentally induced hyperthyroidism disrupts hippocampal long-term potentiation in adult rats. Neuroendocrinology 94(3), 218–227 (2011)
M.H. Samuels, K.G. Schuff, N.E. Carlson, P. Carello, J.S. Janowsky, Health status, mood, and cognition in experimentally induced subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 92(7), 2545–2551 (2007)
X.S. He, N. Ma, Z.L. Pan, Z.X. Wang, N. Li, X.C. Zhang, J.N. Zhou, D.F. Zhu, D.R. Zhang, Functional magnetic resource imaging assessment of altered brain function in hypothyroidism during working memory processing. Eur. J. Endocrinol. 164(6), 951–959 (2011)
R. Cooper, B. Lerer, The use of thyroid hormones in the treatment of depression. Harefuah 149(8), 529–534 (2010)
C.U. Pae, L. Mandelli, C. Han, B.J. Ham, P.S. Masand, A.A. Patkar, D.C. Steffens, D. De Ronchi, A. Serretti, Thyroid hormones affect recovery from depression during antidepressant treatment. Psychiatry Clin. Neurosci. 63(3), 305–313 (2009)
C.E. Fardella, R.A. Artigas, S. Gloger, M. Jimenez, C.A. Carvajal, P.M. Krall, D. Quiroz, C. Campino, L.M. Mosso, Refractory depression in a patient with peripheral resistance to thyroid hormone (RTH) and the effect of triiodothyronine treatment. Endocrine 31(3), 272–278 (2007)
R.M. Pluta, A.E. Burke, R.M. Glass, JAMA patient page. Subclinical hypothyroidism. JAMA 304(12), 1402 (2010)
U. Feldt-Rasmussen, Is the treatment of subclinical hypothyroidism beneficial? Nat. Clin. Pract. Endocrinol. Metab. 5(2), 86–87 (2009)
O.P. Almeida, H. Alfonso, L. Flicker, G. Hankey, S.A. Chubb, B.B. Yeap, Thyroid hormones and depression: the health in men study. Am. J. Geriatr. Psychiatry 19(9), 763–770 (2011)
R.T. de Jongh, P. Lips, N.M. van Schoor, K.J. Rijs, D.J. Deeg, H.C. Comijs, M.H. Kramer, J.P. Vandenbroucke, O.M. Dekkers, Endogenous subclinical thyroid disorders, physical and cognitive function, depression, and mortality in older individuals. Eur. J. Endocrinol. 165(4), 545–554 (2011)
M. Holtmann, E. Duketis, K. Goth, L. Poustka, S. Boelte, Severe affective and behavioral dysregulation in youth is associated with increased serum TSH. J. Affect. Disord. 121(1–2), 184–188 (2010)
R.T. Joffe, E.N. Pearce, J.V. Hennessey, J.J. Ryan, R.A. Stern, Subclinical hypothyroidism, mood, and cognition in older adults: a review. Int. J. Geriatr. Psychiatry 28(2), 111–118 (2013)
J.D. Davis, R.A. Stern, L.A. Flashman, Cognitive and neuropsychiatric aspects of subclinical hypothyroidism: significance in the elderly. Curr. Psychiatry Rep. 5(5), 384–390 (2003)
V.B. Chueire, J.H. Romaldini, L.S. Ward, Subclinical hypothyroidism increases the risk for depression in the elderly. Arch. Gerontol. Geriatr. 44(1), 21–28 (2007)
D.F. Swaab, A.M. Bao, P.J. Lucassen, The stress system in the human brain in depression and neurodegeneration. Ageing Res. Rev. 4(2), 141–194 (2005)
D.L. Helmreich, D.B. Parfitt, X.Y. Lu, H. Akil, S.J. Watson, Relation between the hypothalamic–pituitary–thyroid (HPT) axis and the hypothalamic–pituitary–adrenal (HPA) axis during repeated stress. Neuroendocrinology 81(3), 183–192 (2005)
A. Montero-Pedrazuela, I. Fernandez-Lamo, M. Alieva, I. Pereda-Perez, C. Venero, A. Guadano-Ferraz, Adult-onset hypothyroidism enhances fear memory and upregulates mineralocorticoid and glucocorticoid receptors in the amygdala. PLoS ONE 6(10), e26582 (2011)
J.F. Ge, L. Peng, C.M. Hu, T.N. Wu, Impaired learning and memory performance in subclinical hypothyroidism rat model induced by hemi-thyroid electrocauterization. J. Neuroendocrinol. 24(6), 953–961 (2012)
L.M. Wu, H. Han, Q.N. Wang, H.L. Hou, H. Tong, X.B. Yan, J.N. Zhou, Mifepristone repairs region-dependent alteration of synapsin I in hippocampus in rat model of depression. Neuropsychopharmacology 32(12), 2500–2510 (2007)
Y. Yamawaki, M. Fuchikami, S. Morinobu, M. Segawa, T. Matsumoto, S. Yamawaki, Antidepressant-like effect of sodium butyrate (HDAC inhibitor) and its molecular mechanism of action in the rat hippocampus. World J. Biol. Psychiatry 13(6), 458–467 (2012)
H. Zhu, Q. Huang, H. Xu, L. Niu, J.N. Zhou, Antidepressant-like effects of sodium butyrate in combination with estrogen in rat forced swimming test: involvement of 5-HT(1A) receptors. Behav. Brain Res. 196(2), 200–206 (2009)
H. Gharib, R.M. Tuttle, H.J. Baskin, L.H. Fish, P.A. Singer, M.T. McDermott, Subclinical thyroid dysfunction: a joint statement on management from the American Association of Clinical Endocrinologists, the American Thyroid Association, and the Endocrine Society. J. Clin. Endocrinol. Metab. 90(1), 581–585 (2005). discussion 586–587
D.S. Cooper, B. Biondi, Subclinical thyroid disease. Lancet 379(9821), 1142–1154 (2012)
D.S. Cooper, Clinical practice. Subclinical hypothyroidism. N. Engl. J. Med. 345(4), 260–265 (2001)
M. Imaizumi, N. Sera, I. Ueki, I. Horie, T. Ando, T. Usa, S. Ichimaru, E. Nakashima, A. Hida, M. Soda, T. Tominaga, K. Ashizawa, R. Maeda, S. Nagataki, M. Akahoshi, Risk for progression to overt hypothyroidism in an elderly Japanese population with subclinical hypothyroidism. Thyroid 21(11), 1177–1182 (2011)
J.M. Kim, R. Stewart, S.Y. Kim, K.Y. Bae, S.J. Yang, S.W. Kim, I.S. Shin, J.S. Yoon, Thyroid stimulating hormone, cognitive impairment and depression in an older Korean population. Psychiatry Investig. 7(4), 264–269 (2010)
C. Almeida, M.A. Brasil, A.J. Costa, F.A. Reis, V. Reuters, P. Teixeira, M. Ferreira, A.M. Marques, B.A. Melo, L.B. Teixeira, A. Buescu, M. Vaisman, Subclinical hypothyroidism: psychiatric disorders and symptoms. Rev. Bras. Psiquiatr. 29(2), 157–159 (2007)
M.T. McDermott, E.C. Ridgway, Subclinical hypothyroidism is mild thyroid failure and should be treated. J. Clin. Endocrinol. Metab. 86(10), 4585–4590 (2001)
M. Bauer, T. Goetz, T. Glenn, P.C. Whybrow, The thyroid–brain interaction in thyroid disorders and mood disorders. J. Neuroendocrinol. 20(10), 1101–1114 (2008)
G. Bono, R. Fancellu, F. Blandini, G. Santoro, M. Mauri, Cognitive and affective status in mild hypothyroidism and interactions with l-thyroxine treatment. Acta Neurol. Scand. 110(1), 59–66 (2004)
L. Zhang, V.S. Hernandez, M. Medina-Pizarro, P. Valle-Leija, A. Vega-Gonzalez, T. Morales, Maternal hyperthyroidism in rats impairs stress coping of adult offspring. J. Neurosci. Res. 86(6), 1306–1315 (2008)
A. Kulikov, J. Torresani, R. Jeanningros, Experimental hypothyroidism increases immobility in rats in the forced swim paradigm. Neurosci. Lett. 234(2–3), 111–114 (1997)
A. Montero-Pedrazuela, C. Venero, R. Lavado-Autric, I. Fernandez-Lamo, J.M. Garcia-Verdugo, J. Bernal, A. Guadano-Ferraz, Modulation of adult hippocampal neurogenesis by thyroid hormones: implications in depressive-like behavior. Mol. Psychiatry 11(4), 361–371 (2006)
I. Berlin, C. Payan, E. Corruble, A.J. Puech, Serum thyroid-stimulating-hormone concentration as an index of severity of major depression. Int. J. Neuropsychopharmacol. 2(2), 105–110 (1999)
X.N. Chen, Q.Y. Meng, A.M. Bao, D.F. Swaab, G.H. Wang, J.N. Zhou, The involvement of retinoic acid receptor-alpha in corticotropin-releasing hormone gene expression and affective disorders. Biol. Psychiatry 66(9), 832–839 (2009)
D.F. Swaab, E. Fliers, W.J. Hoogendijk, D.J. Veltman, J.N. Zhou, Interaction of prefrontal cortical and hypothalamic systems in the pathogenesis of depression. Prog. Brain Res. 126, 369–396 (2000)
A. Tohei, G. Watanabe, K. Taya, Hypersecretion of corticotrophin-releasing hormone and arginine vasopressin in hypothyroid male rats as estimated with push-pull perfusion. J. Endocrinol. 156(2), 395–400 (1998)
K.B. Sanchez-Huerta, S. Montes, F. Perez-Severiano, C. Alva-Sanchez, C. Rios, J. Pacheco-Rosado, Hypothyroidism reduces glutamate-synaptic release by ouabain depolarization in rat CA3-hippocampal region. J. Neurosci. Res. 90(4), 905–912 (2012)
K.H. Alzoubi, A.M. Aleisa, K.A. Alkadhi, Molecular studies on the protective effect of nicotine in adult-onset hypothyroidism-induced impairment of long-term potentiation. Hippocampus 16(10), 861–874 (2006)
K.H. Alzoubi, N.Z. Gerges, K.A. Alkadhi, Levothyroxin restores hypothyroidism-induced impairment of LTP of hippocampal CA1: electrophysiological and molecular studies. Exp. Neurol. 195(2), 330–341 (2005)
N.Z. Gerges, K.H. Alzoubi, C.R. Park, D.M. Diamond, K.A. Alkadhi, Adverse effect of the combination of hypothyroidism and chronic psychosocial stress on hippocampus-dependent memory in rats. Behav. Brain Res. 155(1), 77–84 (2004)
P. Ambrogini, R. Cuppini, P. Ferri, C. Mancini, S. Ciaroni, A. Voci, E. Gerdoni, G. Gallo, Thyroid hormones affect neurogenesis in the dentate gyrus of adult rat. Neuroendocrinology 81(4), 244–253 (2005)
R. Eitan, G. Landshut, T. Lifschytz, O. Einstein, T. Ben-Hur, B. Lerer, The thyroid hormone, triiodothyronine, enhances fluoxetine-induced neurogenesis in rats: possible role in antidepressant-augmenting properties. Int. J. Neuropsychopharmacol. 13(5), 553–561 (2010)
N. Correia, S. Mullally, G. Cooke, T.K. Tun, N. Phelan, J. Feeney, M. Fitzgibbon, G. Boran, S. O’Mara, J. Gibney, Evidence for a specific defect in hippocampal memory in overt and subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 94(10), 3789–3797 (2009)
A.J. Cleare, A. McGregor, V. O’Keane, Neuroendocrine evidence for an association between hypothyroidism, reduced central 5-HT activity and depression. Clin. Endocrinol. (Oxf) 43(6), 713–719 (1995)
N. Firouzabadi, M. Shafiei, E. Bahramali, S.A. Ebrahimi, H. Bakhshandeh, N. Tajik, Association of angiotensin-converting enzyme (ACE) gene polymorphism with elevated serum ACE activity and major depression in an Iranian population. Psychiatry Res. 200(2–3), 336–342 (2012)
H. Murck, P. Schussler, A. Steiger, Renin-angiotensin-aldosterone system: the forgotten stress hormone system: relationship to depression and sleep. Pharmacopsychiatry 45(3), 83–95 (2012)
Y.B. Saab, P.R. Gard, M.S. Yeoman, B. Mfarrej, H. El-Moalem, M.J. Ingram, Renin-angiotensin-system gene polymorphisms and depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 31(5), 1113–1118 (2007)
J.W. Wright, J.W. Harding, Brain renin-angiotensin—a new look at an old system. Prog. Neurobiol. 95(1), 49–67 (2011)
E. Isingrini, T. Desmidt, C. Belzung, V. Camus, Endothelial dysfunction: a potential therapeutic target for geriatric depression and brain amyloid deposition in Alzheimer’s disease? Curr. Opin. Investig. Drugs 10(1), 46–55 (2009)
M.S. Carneiro-Ramos, G.P. Diniz, J. Almeida, R.L. Vieira, S.V. Pinheiro, R.A. Santos, M.L. Barreto-Chaves, Cardiac angiotensin II type I and type II receptors are increased in rats submitted to experimental hypothyroidism. J. Physiol. 583(Pt 1), 213–223 (2007)
Acknowledgments
This project was supported by the Natural Science Foundation of China (81030026), the Natural Science Foundation of Anhui Province of China (11040606Q19), and Scientific Research of BSKY from Anhui Medical University (KJ201210).
Ethical standards
All experimental procedures were approved by Animal Care and Use Committee at University of Science and Technology of China, which complies with the National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1985).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ge, JF., Peng, YY., Qi, CC. et al. Depression-like behavior in subclinical hypothyroidism rat induced by hemi-thyroid electrocauterization. Endocrine 45, 430–438 (2014). https://doi.org/10.1007/s12020-013-0001-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-013-0001-4