Abstract
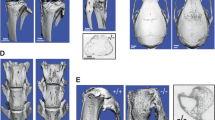
SHARPIN is a novel protein thought to interact with SHANK family and is widely expressed in multiple tissues/cells, including osteoblasts and osteoclasts. Loss-of-function of Sharpin develops the chronic proliferative dermatitis mutation (CPDM) in mice as well as a severe inflammation in other organs. The actual function of SHARPIN is poorly understood. Our aim was to determine the functional roles of SHARPIN in bone metabolism by using CPDM mice. The skeletal phenotypes were determined by peripheral quantitative computed tomography, micro-computed tomography, and quantitative real-time RT-PCR, the cellular functions of osteoblasts and osteoclasts were investigated by ex vivo cell culture. Compared to wild-type controls, CPDM mice demonstrated significantly lower total and cortical bone mineral content and bone mineral density, trabecular and cortical bone volume, and trabecular number. The mRNA expression of Runx2, osterix, type I collagen, and osteocalcin was significantly lower in the bone from CPDM mice. Osteoclasts and osteoblasts from CPDM mice were functionally defective. Our result suggests that SHARPIN plays important regulating roles in bone metabolism. These functional roles may either come from systemic chronic inflammatory or directly signaling pathway within bone cells.




Similar content being viewed by others
References
Y. Daigo, I. Takayama, S.M. Ward, K.M. Sanders, M.A. Fujino, Novel human and mouse genes encoding a shank-interacting protein and its upregulation in gastric fundus of W/WV mouse. J. Gastroenterol. Hepatol. 18, 712–718 (2003)
A. Marchler-Bauer, J.B. Anderson, M.K. Derbyshire, C. DeWeese-Scott, N.R. Gonzales, M. Gwadz, L. Hao, S. He, D.I. Hurwitz, J.D. Jackson, Z. Ke, D. Krylov, C.J. Lanczycki, C.A. Liebert, C. Liu, F. Lu, S. Lu, G.H. Marchler, M. Mullokandov, J.S. Song, N. Thanki, R.A. Yamashita, J.J. Yin, D. Zhang, S.H. Bryant, CDD: a conserved domain database for interactive domain family analysis. Nucleic Acids Res. 35, D237–D240 (2007)
S. Lim, C. Sala, J. Yoon, S. Park, S. Kuroda, M. Sheng, E. Kim, Sharpin, a novel postsynaptic density protein that directly interacts with the shank family of proteins. Mol. Cell Neurosci. 17, 385–397 (2001)
J. Jung, J.M. Kim, B. Park, Y. Cheon, B. Lee, S.H. Choo, S.S. Koh, S. Lee, Newly identified tumor-associated role of human Sharpin. Mol. Cell. Biochem. 340, 161–167 (2010)
R.E. Seymour, M.G. Hasham, G.A. Cox, L.D. Shultz, H. Hogenesch, D.C. Roopenian, J.P. Sundberg, Spontaneous mutations in the mouse Sharpin gene result in multiorgan inflammation, immune system dysregulation and dermatitis. Genes Immun. 8, 416–421 (2007)
H. HogenEsch, M.J. Gijbels, E. Offerman, J. van Hooft, D.W. van Bekkum, C. Zurcher, A spontaneous mutation characterized by chronic proliferative dermatitis in C57BL mice. Am. J. Pathol. 143, 972–982 (1993)
H.I. Gallardo Torres, M.J. Gijbels, H. HegnEsch, G. Kraal, Chronic proliferative dermatitis in mice: neutrophil-endothelium interactions and the role of adhesion molecules. Pathobiology 63, 341–347 (1995)
M.J. Gijbels, H. HogenEsch, B. Blauw, P. Roholl, C. Zurcher, Ultrastructure of epidermis of mice with chronic proliferative dermatitis. Ultrastruct. Pathol. 19, 107–111 (1995)
M.J. Gijbels, C. Zurcher, G. Kraal, G.R. Elliott, H. HogenEsch, G. Schijff, H.F. Savelkoul, P.L. Bruijnzeel, Pathogenesis of skin lesions in mice with chronic proliferative dermatitis (cpdm/cpdm). Am. J. Pathol. 148, 941–950 (1996)
H. HogenEsch, D. Boggess, J.P. Sundberg, Changes in keratin and filaggrin expression in the skin of CPDM mutant mice. Pathobiology 67, 45–50 (1999)
H. HogenEsch, S. Janke, D. Boggess, J.P. Sundberg, Absence of Peyer’s patches and abnormal lymphoid architecture in chronic proliferative dermatitis (cpdm/cpdm) mice. J. Immunol. 162, 3890–3896 (1999)
R. Hardy, M.S. Cooper, Bone loss in inflammatory disorders. J. Endocrinol. 201, 309–320 (2009)
T.M. Schroeder, E.D. Jensen, J.J. Westendorf, Runx2: a master organizer of gene transcription in developing and maturing osteoblasts. Birth Defects Res. C Embryo Today 75, 213–225 (2005)
K. Nakashima, X. Zhou, G. Kunkel, Z. Zhang, J.M. Deng, R.R. Behringer, B. de Crombrugghe, The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108, 17–29 (2002)
C. Desbois, G. Karsenty, Osteocalcin cluster: implications for functional studies. J. Cell Biochem. 57, 379–383 (1995)
J. Lorenzo, M. Horowitz, Y. Choi, Osteoimmunology: interactions of the bone and immune system. Endocr. Rev. 29, 403–440 (2008)
L. Gilbert, X. He, P. Farmer, S. Boden, M. Kozlowski, J. Rubin, M.S. Nanes, Inhibition of osteoblast differentiation by tumor necrosis factor-alpha. Endocrinology 141, 3956–3964 (2000)
M.L. Renninger, R. Seymour, J.W. Lillard Jr., J.P. Sundberg, H. HogenEsch, Increased expression of chemokines in the skin of chronic proliferative dermatitis mutant mice. Exp. Dermatol. 14, 906–913 (2005)
I.M. Haeck, N.A. Hamdy, L. Timmer-de Mik, E.G. Lentjes, H.J. Verhaar, M.J. Knol, M.S. de Bruin-Weller, C.A. Bruijnzeel-Koomen, Low bone mineral density in adult patients with moderate to severe atopic dermatitis. Br. J. Dermatol. 161, 1248–1254 (2009)
A. Meixner, R. Zenz, H.B. Schonthaler, L. Kenner, H. Scheuch, J.M. Penninger, E.F. Wagner, Epidermal JunB represses G-CSF transcription and affects haematopoiesis and bone formation. Nat. Cell Biol. 10, 1003–1011 (2008)
J.E. Onyia, B. Miller, J. Hulman, J. Liang, R. Galvin, C. Frolik, S. Chandrasekhar, A.K. Harvey, J. Bidwell, J. Herring, J.M. Hock, Proliferating cells in the primary spongiosa express osteoblastic phenotype in vitro. Bone 20, 93–100 (1997)
K.J. Livak, T.D. Schmittgen, Analysis of relative gene expression data using real-time qPCR and the 2(-Delta Delta C(T)) method. Methods 25, 402–408 (2001)
Acknowledgment
This work was support by a grant to Dr. Xijie Yu from National Natural Science Foundation of China (No. 30872632).
Author information
Authors and Affiliations
Corresponding author
Additional information
Tian Xia and Yanhua Liang contribute equally to this work.
Rights and permissions
About this article
Cite this article
Xia, T., Liang, Y., Ma, J. et al. Loss-of-function of SHARPIN causes an osteopenic phenotype in mice. Endocr 39, 104–112 (2011). https://doi.org/10.1007/s12020-010-9418-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-010-9418-1