Abstract
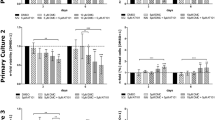
Temozolomide (TMZ) is widely used for treating glioblastoma (GBM), which can effectively inhibit the GBM growth for some months; however, it still cannot prevent the invariable recurrence of GBM. Improving the chemotherapeutic sensitization becomes an urgent agenda. In this study, we found low-dose demethoxycurcumin (DMC) could enhance the sensitivity of TMZ on glioma cells, and high-dose DMC has more significant effects on GBM cells compared with TMZ treatment alone both in vitro and in vivo. And co-administration of DMC and TMZ resulted in a significant increase in GBM apoptosis and a marked inhibition of cell growth pathogenesis of GBM. Mechanistically, DMC and TMZ synergistically increase intracellular level of reactive oxygen species (ROS) production, activate caspase-3-dependent apoptotic pathway, and inactivate of JAK/STAT3 signaling pathway in GBMs, which account for the cell apoptosis and proliferation inhibition. Together, these data implicate that low-dose DMC combined with TMZ represents an effective therapy regimen against GBMs by targeting multiple signaling pathways.





Similar content being viewed by others
References
Athanassiou, H., Synodinou, M., Maragoudakis, E., Paraskevaidis, M., Verigos, C., Misailidou, D., et al. (2005). Randomized phase II study of temozolomide and radiotherapy compared with radiotherapy alone in newly diagnosed glioblastoma multiforme. Journal of Clinical Oncology, 23, 2372–2377.
Barré, B., Avril, S., & Coqueret, O. (2003). Opposite regulation of Myc and p21waf1 transcription by STAT3 proteins. Journal of Biological Chemistry, 278(5), 2990–2996.
Bojes, H. K., Suresh, P. K., Mills, E. M., Spitz, D. R., Sim, J. E., & Kehrer, J. P. (1998). Bcl-2 and Bcl-xL in peroxide-resistant A549 and U87MG cells. Toxicological Sciences, 42, 109–116.
Chen, L., Zhang, J., Han, L., Zhang, A., Zhang, C., Zheng, Y., et al. (2012). Downregulation of miR-221/222 sensitizes glioma cells to temozolomide by regulating apoptosis independently of p53 status. Oncology Reports, 27(3), 854–860.
Dauer, D. J., Ferraro, B., Song, L., Yu, B., Mora, L., Buettner, R., et al. (2005). Stat3 regulates genes common to both wound healing and cancer. Oncogene, 24(21), 3397–3408.
Garrido, C., Galluzzi, L., Brunet, M., Puig, P. E., Didelot, C., & Kroemer, G. (2006). Mechanisms of cytochrome c release from mitochondria. Cell Death and Differentiation, 13(9), 1423–1433.
Hsu, Y. C., Weng, H. C., Lin, S., & Chien, Y. W. (2007). Curcuminoids-cellular uptake by human primary colon cancer cells as quantitated by a sensitive HPLC assay and its relation with the inhibition of proliferation and apoptosis. Journal of Agriculture and Food Chemistry, 55(20), 8213–8222.
Huang, T. Y., Hsu, C. W., Chang, W. C., Wang, M. Y., Wu, J. F., & Hsu, Y. C. (2012). Demethoxycurcumin retards cell growth and induces apoptosis in human brain malignant glioma GBM 8401 cells. Evidence-Based Complementary and Alternative Medicine, 2012, 396573.
Ji, J. D., Kim, H. J., Rho, Y. H., Choi, S. J., Lee, Y. H., Cheon, H. J., et al. (2005). Inhibition of IL-10-induced STAT3 activation by 15-deoxy-Delta 12,14-prostaglandin J2. Rheumatology (Oxford), 44(8), 983–988.
Johnson, D. R., & O’Neill, B. P. (2012). Glioblastoma survival in the United States before and during the temozolomide era. Journal of Neuro-Oncology, 107(2), 359–364.
Ko, Y. C., Lien, J. C., Liu, H. C., Hsu, S. C., Ji, B. C., Yang, M. D., et al. (2015). Demethoxycurcumin induces the apoptosis of human lung cancer NCI-H460 cells through the mitochondrial-dependent pathway. Oncology Reports, 33(5), 2429–2437.
Lee, J. S., Ahn, J. H., Cho, Y. J., Kim, H. Y., Yang, Y. I., Lee, K. T., et al. (2015). α-Terthienylmethanol, isolated from Eclipta prostrata, induces apoptosis by generating reactive oxygen species via NADPH oxidase in human endometrial cancer cells. Journal of Ethnopharmacology, 169, 426–434.
Lee, J. W., Hong, H. M., Kwon, D. D., Pae, H. O., & Jeong, H. J. (2010). Dimethoxycurcumin, a structural analogue of curcumin, induces apoptosis in human renal carcinoma caki cells through the production of reactive oxygen species, the release of cytochrome C, and the activation of caspase-3. Korean Journal of Urology, 51(12), 870–878.
Luthra, P. M., Kumar, R., & Prakash, A. (2009). Demethoxycurcumin induces Bcl-2 mediated G2/M arrest and apoptosis in human glioma U87 cells. Biochemical and Biophysical Research Communications, 384(4), 420–425.
Narayan, P., Mentzer, R. M, Jr, & Lasley, R. D. (2001). Annexin V staining during reperfusion detects cardiomyocytes with unique properties. American Journal of Physiology Heart and Circulatory Physiology, 281, H1931–H1937.
Ni, X., Zhang, A., Zhao, Z., Shen, Y., & Wang, S. (2012). Demethoxycurcumin inhibits cell proliferation, migration and invasion in prostate cancer cells. Oncology Reports, 28(1), 85–90.
Oliva, C. R., Moellering, D. R., Gillespie, G. Y., & Griguer, C. E. (2011). Acquisition of chemoresistance in gliomas is associated with increased mitochondrial coupling and decreased ROS production. PLoS ONE, 6(9), e24665.
Rahaman, S. O., Harbor, P. C., Chernova, O., Barnett, G. H., Vogelbaum, M. A., & Haque, S. J. (2002). Inhibition of constitutively active Stat3 suppresses proliferation and induces apoptosis in glioblastoma multiforme cells. Oncogene, 21(55), 8404–8413.
Ravindran, J., Prasad, S., & Aggarwal, B. B. (2009). Curcumin and cancer cells: How many ways can curry kill tumor cells selectively? AAPS Journal, 11(3), 495–510.
Sandur, S. K., Pandey, M. K., Sung, B., Ahn, K. S., Murakami, A., Sethi, G., et al. (2007). Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis, 28(8), 1765–1773.
Sang, D. P., Li, R. J., & Lan, Q. (2014). Quercetin sensitizes human glioblastoma cells to temozolomide in vitro via inhibition of Hsp27. Acta Pharmacologica Sinica, 35(6), 832–838.
Selvendiran, K., Tong, L., Bratasz, A., Kuppusamy, M. L., Ahmed, S., Ravi, Y., et al. (2010). Anticancer efficacy of a difluorodiarylidenylpiperidone (HO-3867) in human ovarian cancer cells and tumor xenografts. Molecular Cancer Therapeutics, 9(5), 1169–1179.
Senft, C., Polacin, M., Priester, M., Seifert, V., Kögel, D., & Weissenberger, J. (2010). The nontoxic natural compound Curcumin exerts anti-proliferative, anti-migratory, and anti-invasive properties against malignant gliomas. BMC Cancer, 10, 491.
Shi, L., Chen, J., Wang, Y. Y., Sun, G., Liu, J. N., Zhang, J. X., et al. (2012a). Gossypin induces G2/M arrest in human malignant glioma U251 cells by the activation of Chk1/Cdc25C pathway. Cellular and Molecular Neurobiology, 32(2), 289–296.
Shi, L., Fei, X., & Wang, Z. (2015). Demethoxycurcumin was prior to temozolomide on inhibiting proliferation and induced apoptosis of glioblastoma stem cells. Tumour Biology [Epub ahead of print].
Shi, L., Zhang, S., Feng, K., Wu, F., Wan, Y., Wang, Z., et al. (2012b). MicroRNA-125b-2 confers human glioblastoma stem cells resistance to temozolomide through the mitochondrial pathway of apoptosis. International Journal of Oncology, 40(1), 119–129.
Shi, L., Zhang, J., Pan, T., Zhou, J., Gong, W., Liu, N., et al. (2010). MiR-125b is critical for the suppression of human U251 glioma stem cell proliferation. Brain Research, 1312, 120–126.
Shieh, J. M., Chen, Y. C., Lin, Y. C., Lin, J. N., Chen, W. C., Chen, Y. Y., et al. (2013). Demethoxycurcumin inhibits energy metabolic and oncogenic signaling pathways through AMPK activation in triple-negative breast cancer cells. Journal of Agriculture and Food Chemistry, 61(26), 6366–6375.
Stupp, R., Tonn, J. C., Brada, M., Pentheroudakis, G., & ESMO Guidelines Working Group. (2010). High-grade malignant glioma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Annals of Oncology, 21, v190–v193.
Taha, H., Looi, C. Y., Arya, A., Wong, W. F., Yap, L. F., Hasanpourghadi, M., et al. (2015). (6E,10E) isopolycerasoidol and (6E,10E) isopolycerasoidol methyl ester, prenylated benzopyran derivatives from pseuduvaria monticola induce mitochondrial-mediated apoptosis in human breast adenocarcinoma cells. PLoS ONE, 10(5), e0126126.
Teng, Y. N., Hsieh, Y. W., Hung, C. C., & Lin, H. Y. (2015). Demethoxycurcumin modulates human P-glycoprotein function via uncompetitive inhibition of ATPase hydrolysis activity. Journal of Agriculture and Food Chemistry, 63(3), 847–855.
Terrano, D. T., Upreti, M., & Chambers, T. C. (2010). Cyclin-dependent kinase 1-mediated Bcl-xL/Bcl-2 phosphorylation acts as a functional link coupling mitotic arrest and apoptosis. Molecular and Cellular Biology, 30, 640–656.
Wen, X., Huang, A., Liu, Z., Liu, Y., Hu, J., Liu, J., et al. (2014). Downregulation of ROCK2 through nanocomplex sensitizes the cytotoxic effect of temozolomide in U251 glioma cells. PLoS ONE, 9(3), e92050.
Xu, Z. Y., Wang, K., Li, X. Q., Chen, S., Deng, J. M., Cheng, Y., et al. (2013). The ABCG2 transporter is a key molecular determinant of the efficacy of sonodynamic therapy with Photofrin in glioma stem-like cells. Ultrasonics, 53(1), 232–238.
Yang, M. C., Loh, J. K., Li, Y. Y., Huang, W. S., Chou, C. H., Cheng, J. T., et al. (2015). Bcl2L12 with a BH3-like domain in regulating apoptosis and TMZ-induced autophagy: A prospective combination of ABT-737 and TMZ for treating glioma. International Journal of Oncology, 46(3), 1304–1316.
Zhang, S., Wan, Y., Pan, T., Gu, X., Qian, C., Sun, G., et al. (2012). MicroRNA-21 inhibitor sensitizes human glioblastoma U251 stem cells to chemotherapeutic drug temozolomide. Journal of Molecular Neuroscience, 47(2), 346–356.
Acknowledgments
This work was supported by the China Natural Science Foundation (81000963, 81370062 and 81302196), Jiangsu Province’s 333 Talent Program (BRA2011046), Jiangsu Province “six personnel peak” funded projects (2013-WSN-145/028), Jiangsu Province’s Natural Science Foundation (BK2012670), Medical Research Foundation by Jiangsu Province Health Department (YG201301 and Z201318), the Clinical Technology Development of Jiangsu University (JLY20120053), the Kunshan Social Development Foundation (KS1006, KS1009), and the Suzhou Social Development Foundation (SYS201063). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have declared the sources of research funding for this manuscript and have no financial or other contractual agreements that might cause (or be perceived as causes of) conflicts of interest.
Additional information
Lei Shi and Guan Sun contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shi, L., Sun, G. Low-Dose DMC Significantly Enhances the Effect of TMZ on Glioma Cells by Targeting Multiple Signaling Pathways Both In Vivo and In Vitro. Neuromol Med 17, 431–442 (2015). https://doi.org/10.1007/s12017-015-8372-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-015-8372-8