Abstract
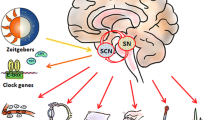
Parkinson’s disease (PD) is a common neurodegenerative disorder that is characterized by the degeneration of dopaminergic neurons in the substantia nigra and dopamine depletion in the striatum. Although the motor symptoms are still regarded as the main problem, non-motor symptoms in PD also markedly impair the quality of life. Several non-motor symptoms, such as sleep disturbances and depression, are suggested to be implicated in the alteration in circadian clock function. In this study, we investigated circadian disruption and the mechanism in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mouse model of PD. MPTP-treated mice exhibited altered 24-h rhythms in body temperature and locomotor activity. In addition, MPTP treatment also affected the circadian clock system at the genetic level. The exposure of human neuroblastoma cells (SH-SY5Y) to 1-metyl-4-phenylpyridinium (MPP+) increased or decreased the mRNA levels of several clock genes in a dose-dependent manner. MPP+-induced changes in clock genes expression were reversed by Compound C, an inhibitor of AMP-activated protein kinase (AMPK). Most importantly, addition of ATP to the drinking water of MPTP-treated mice attenuated neurodegeneration in dopaminergic neurons, suppressed AMPK activation and prevented circadian disruption. The present findings suggest that the activation of AMPK caused circadian dysfunction, and ATP may be a novel therapeutic strategy based on the molecular clock in PD.





Similar content being viewed by others
References
Alfinito, P. D., Wang, S. P., Manzino, L., Rijhsinghani, S., Zeevalk, G. D., & Sonsalla, P. K. (2003). Adenosinergic protection of dopaminergic and GABAergic neurons against mitochondrial inhibition through receptors located in the substantia nigra and striatum, respectively. Journal of Neuroscience, 23(34), 10982–10987.
Almirall, H., Bautista, V., Sanchez-Bahillo, A., & Trinidad-Herrero, M. (2001). Ultradian and circadian body temperature and activity rhythms in chronic MPTP treated monkeys. Neurophysiologie Clinique, 31(3), 161–170.
Barcia, C., De Pablos, V., Bautista-Hernandez, V., Sanchez-Bahillo, A., Fernandez-Barreiro, A., Poza, M., et al. (2004). Measurement of motor disability in MPTP-treated macaques using a telemetry system for estimating circadian motor activity. Journal of Neuroscience Methods, 134(1), 59–64.
Brooks, W. J., Jarvis, M. F., & Wagner, G. C. (1989). Astrocytes as a primary locus for the conversion MPTP into MPP+. Journal of Neural Transmission, 76(1), 1–12.
Bruguerolle, B. (2008). Clinical chronopharmacology in the elderly. Chronobiology International, 25(1), 1–15.
Bruguerolle, B., & Simon, N. (2002). Biologic rhythms and Parkinson’s disease: A chronopharmacologic approach to considering fluctuations in function. Clinical Neuropharmacology, 25(4), 194–201.
Cai, Y., Liu, S., Sothern, R. B., Xu, S., & Chan, P. (2010). Expression of clock genes Per1 and Bmal1 in total leukocytes in health and Parkinson’s disease. European Journal of Neurology, 17(4), 550–554.
Cheng, M. Y., Bullock, C. M., Li, C., Lee, A. G., Bermak, J. C., Belluzzi, J., et al. (2002). Prokineticin 2 transmits the behavioural circadian rhythm of the suprachiasmatic nucleus. Nature, 417(6887), 405–410.
Chikahisa, S., Fujiki, N., Kitaoka, K., Shimizu, N., & Sei, H. (2009). Central AMPK contributes to sleep homeostasis in mice. Neuropharmacology, 57(4), 369–374.
Choi, J. S., Park, C., & Jeong, J. W. (2010). AMP-activated protein kinase is activated in Parkinson’s disease models mediated by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Biochemical and Biophysical Research Communications, 391(1), 147–151.
Ciruela, F., Gomez-Soler, M., Guidolin, D., Borroto-Escuela, D. O., Agnati, L. F., Fuxe, K., et al. (2011). Adenosine receptor containing oligomers: Their role in the control of dopamine and glutamate neurotransmission in the brain. Biochimica et Biophysica Acta, 1808(5), 1245–1255.
Cunha, R. A. (2005). Neuroprotection by adenosine in the brain: From A(1) receptor activation to A (2A) receptor blockade. Purinergic Signaling, 1(2), 111–134.
Curtis, A. M., Cheng, Y., Kapoor, S., Reilly, D., Price, T. S., & Fitzgerald, G. A. (2007). Circadian variation of blood pressure and the vascular response to asynchronous stress. Proceedings of National Academy of Sciences USA, 104(9), 3450–3455.
Dauer, W., & Przedborski, S. (2003). Parkinson’s disease: Mechanisms and models. Neuron, 39(6), 889–909.
de Mendonca, A., Sebastiao, A. M., & Ribeiro, J. A. (2000). Adenosine: Does it have a neuroprotective role after all? Brain Research Reviews, 33(2–3), 258–274.
Delle Donne, K. T., & Sonsalla, P. K. (1994). Protection against methamphetamine-induced neurotoxicity to neostriatal dopaminergic neurons by adenosine receptor activation. Journal of Pharmacology and Experimental Therapeutics, 271(3), 1320–1326.
Eastman, C. I., Mistlberger, R. E., & Rechtschaffen, A. (1984). Suprachiasmatic nuclei lesions eliminate circadian temperature and sleep rhythms in the rat. Physiology & Behavior, 32(3), 357–368.
Etchegaray, J. P., Lee, C., Wade, P. A., & Reppert, S. M. (2003). Rhythmic histone acetylation underlies transcription in the mammalian circadian clock. Nature, 421(6919), 177–182.
Fastbom, J., Pazos, A., & Palacios, J. M. (1987). The distribution of adenosine A1 receptors and 5′-nucleotidase in the brain of some commonly used experimental animals. Neuroscience, 22(3), 813–826.
Fredholm, B. B., Abbracchio, M. P., Burnstock, G., Daly, J. W., Harden, T. K., Jacobson, K. A., et al. (1994). Nomenclature and classification of purinoceptors. Pharmacological Reviews, 46(2), 143–156.
Fredholm, B. B., Irenius, E., Kull, B., & Schulte, G. (2001). Comparison of the potency of adenosine as an agonist at human adenosine receptors expressed in Chinese hamster ovary cells. Biochemical Pharmacology, 61(4), 443–448.
Gekakis, N., Staknis, D., Nguyen, H. B., Davis, F. C., Wilsbacher, L. D., King, D. P., et al. (1998). Role of the CLOCK protein in the mammalian circadian mechanism. Science, 280(5369), 1564–1569.
Goldberg, N. R., Haack, A. K., Lim, N. S., Janson, O. K., & Meshul, C. K. (2011). Dopaminergic and behavioral correlates of progressive lesioning of the nigrostriatal pathway with 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine. Neuroscience, 180, 256–271.
Gordon, J. L. (1986). Extracellular ATP: Effects, sources and fate. Biochemical Journal, 233(2), 309–319.
Hartmann, A., Veldhuis, J. D., Deuschle, M., Standhardt, H., & Heuser, I. (1997). Twenty-four hour cortisol release profiles in patients with Alzheimer’s and Parkinson’s disease compared to normal controls: Ultradian secretory pulsatility and diurnal variation. Neurobiology of Aging, 18(3), 285–289.
Hastings, M. H. (1997). Central clocking. Trends in Neurosciences, 20(10), 459–464.
Hayashida, M., Fukuda, K., & Fukunaga, A. (2005). Clinical application of adenosine and ATP for pain control. Journal of Anesthesia, 19(3), 225–235.
Isojima, Y., Okumura, N., & Nagai, K. (2003). Molecular mechanism of mammalian circadian clock. Journal of Biochemistry, 134(6), 777–784.
Jackson-Lewis, V., & Przedborski, S. (2007). Protocol for the MPTP mouse model of Parkinson’s disease. Nature Protocols, 2(1), 141–151.
Jin, X., Shearman, L. P., Weaver, D. R., Zylka, M. J., de Vries, G. J., & Reppert, S. M. (1999). A molecular mechanism regulating rhythmic output from the suprachiasmatic circadian clock. Cell, 96(1), 57–68.
Knutsson, A. (2003). Health disorders of shift workers. Occupational Medicine, 53(2), 103–108.
Koyanagi, S., Kuramoto, Y., Nakagawa, H., Aramaki, H., Ohdo, S., Soeda, S., et al. (2003). A molecular mechanism regulating circadian expression of vascular endothelial growth factor in tumor cells. Cancer Research, 63(21), 7277–7283.
Kudo, T., Loh, D. H., Truong, D., Wu, Y., & Colwell, C. S. (2011). Circadian dysfunction in a mouse model of Parkinson’s disease. Experimental Neurology, 232(1), 66–75.
Kuhn, K., Wellen, J., Link, N., Maskri, L., Lubbert, H., & Stichel, C. C. (2003). The mouse MPTP model: Gene expression changes in dopaminergic neurons. European Journal of Neuroscience, 17(1), 1–12.
Kume, K., Zylka, M. J., Sriram, S., Shearman, L. P., Weaver, D. R., Jin, X., et al. (1999). mCRY1 and mCRY2 are essential components of the negative limb of the circadian clock feedback loop. Cell, 98(2), 193–205.
Lamia, K. A., Sachdeva, U. M., DiTacchio, L., Williams, E. C., Alvarez, J. G., Egan, D. F., et al. (2009). AMPK regulates the circadian clock by cryptochrome phosphorylation and degradation. Science, 326(5951), 437–440.
Lau, Y. S., & Mouradian, M. M. (1993). Protection against acute MPTP-induced dopamine depletion in mice by adenosine A1 agonist. Journal of Neurochemistry, 60(2), 768–771.
Liu, S., Chen, X. M., Yoda, T., Nagashima, K., Fukuda, Y., & Kanosue, K. (2002). Involvement of the suprachiasmatic nucleus in body temperature modulation by food deprivation in rats. Brain Research, 929(1), 26–36.
Maruyama, A., & Fujio, T. (2001). ATP production from adenine by a self-coupling enzymatic process: high-level accumulation under ammonium-limited conditions. Bioscience, Biotechnology, and Biochemistry, 65(3), 644–650.
Matz, H., & Hertz, L. (1989). Adenosine metabolism in neurons and astrocytes in primary cultures. Journal of Neuroscience Research, 24(2), 260–267.
Menza, M., Dobkin, R. D., Marin, H., & Bienfait, K. (2010). Sleep disturbances in Parkinson’s disease. Movement Disorders, 25(Suppl 1), S117–S122.
Michel, P. P., Marien, M., Ruberg, M., Colpaert, F., & Agid, Y. (1999). Adenosine prevents the death of mesencephalic dopaminergic neurons by a mechanism that involves astrocytes. Journal of Neurochemistry, 72(5), 2074–2082.
Mihaylova, M. M., & Shaw, R. J. (2011). The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nature Cell Biology, 13(9), 1016–1023.
Miyasaki, J. M., Martin, W., Suchowersky, O., Weiner, W. J., & Lang, A. E. (2002). Practice parameter: initiation of treatment for Parkinson’s disease: An evidence-based review: Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology, 58(1), 11–17.
Monge, A., Viselli, F., Stocchi, F., Barbato, L., Bolner, A., Modugno, N., et al. (2004). Variation in the dopaminergic response during the day in Parkinson disease. Clinical Neuropharmacology, 27(3), 116–118.
Monteleone, P., & Maj, M. (2008). The circadian basis of mood disorders: Recent developments and treatment implications. European Neuropsychopharmacology, 18(10), 701–711.
Moore, R. Y., & Silver, R. (1998). Suprachiasmatic nucleus organization. Chronobiology International, 15(5), 475–487.
Nagashima, K., Matsue, K., Konishi, M., Iidaka, C., Miyazaki, K., Ishida, N., et al. (2005). The involvement of Cry1 and Cry2 genes in the regulation of the circadian body temperature rhythm in mice. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology, 288(1), R329–R335.
Ng, C. H., Guan, M. S., Koh, C., Ouyang, X., Yu, F., Tan, E. K., et al. (2012). AMP kinase activation mitigates dopaminergic dysfunction and mitochondrial abnormalities in drosophila models of Parkinson’s disease. The Journal of Neuroscience, 32(41), 14311–14317.
Ohdo, S., Koyanagi, S., Matsunaga, N., & Hamdan, A. (2011). Molecular basis of chronopharmaceutics. Journal of Pharmaceutical Sciences, 100(9), 3560–3576.
Ohdo, S., Koyanagi, S., Suyama, H., Higuchi, S., & Aramaki, H. (2001). Changing the dosing schedule minimizes the disruptive effects of interferon on clock function. Nature Medicine, 7(3), 356–360.
Ongini, E., & Schubert, P. (1998). Neuroprotection induced by stimulating A1 or blocking A2A adenosine receptors: An apparent paradox. Drug Development Research, 45, 387–393.
Pahwa, R., Factor, S. A., Lyons, K. E., Ondo, W. G., Gronseth, G., Bronte-Stewart, H., et al. (2006). Practice Parameter: treatment of Parkinson disease with motor fluctuations and dyskinesia (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology, 66(7), 983–995.
Ponsen, M. M., Stoffers, D., Booij, J., van Eck-Smit, B. L., Wolters, E. C., & Berendse, H. W. (2004). Idiopathic hyposmia as a preclinical sign of Parkinson’s disease. Annals of Neurology, 56(2), 173–181.
Preitner, N., Damiola, F., Lopez-Molina, L., Zakany, J., Duboule, D., Albrecht, U., et al. (2002). The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell, 110(2), 251–260.
Ransom, B. R., Kunis, D. M., Irwin, I., & Langston, J. W. (1987). Astrocytes convert the Parkinsonism inducing neurotoxin, MPTP, to its active metabolite, MPP+. Neuroscience Letters, 75(3), 323–328.
Refinetti, R., Kaufman, C. M., & Menaker, M. (1994). Complete suprachiasmatic lesions eliminate circadian rhythmicity of body temperature and locomotor activity in golden hamsters. Journal of Comparative Physiology A, 175(2), 223–232.
Reichmann, H., Schneider, C., & Lohle, M. (2009). Non-motor features of Parkinson’s disease: Depression and dementia. Parkinsonism & Related Disorders, 15(Suppl 3), S87–S92.
Reppert, S. M., & Weaver, D. R. (2002). Coordination of circadian timing in mammals. Nature, 418(6901), 935–941.
Ripperger, J. A., Shearman, L. P., Reppert, S. M., & Schibler, U. (2000). CLOCK, an essential pacemaker component, controls expression of the circadian transcription factor DBP. Genes & Development, 14(6), 679–689.
Ronnett, G. V., Ramamurthy, S., Kleman, A. M., Landree, L. E., & Aja, S. (2009). AMPK in the brain: Its roles in energy balance and neuroprotection. Journal of Neurochemistry, 109(Suppl 1), 17–23.
Rozas, G., Lopez-Martin, E., Guerra, M. J., & Labandeira-Garcia, J. L. (1998). The overall rod performance test in the MPTP-treated-mouse model of Parkinsonism. Journal of Neuroscience Methods, 83(2), 165–175.
Ruby, N. F., Dark, J., Burns, D. E., Heller, H. C., & Zucker, I. (2002). The suprachiasmatic nucleus is essential for circadian body temperature rhythms in hibernating ground squirrels. Journal of Neuroscience, 22(1), 357–364.
Rudolphi, K. A., Schubert, P., Parkinson, F. E., & Fredholm, B. B. (1992). Neuroprotective role of adenosine in cerebral ischaemia. Trends in Pharmacological Sciences, 13(12), 439–445.
Sakamoto, K., Nagase, T., Fukui, H., Horikawa, K., Okada, T., Tanaka, H., et al. (1998). Multitissue circadian expression of rat period homolog (rPer2) mRNA is governed by the mammalian circadian clock, the suprachiasmatic nucleus in the brain. Journal of Biological Chemistry, 273(42), 27039–27042.
Sato, T. K., Yamada, R. G., Ukai, H., Baggs, J. E., Miraglia, L. J., Kobayashi, T. J., et al. (2006). Feedback repression is required for mammalian circadian clock function. Nature Genetics, 38(3), 312–319.
Shinohara, A., Koyanagi, S., Hamdan, A. M., Matsunaga, N., Aramaki, H., & Ohdo, S. (2008). Dosing schedule-dependent change in the disruptive effects of interferon-alpha on the circadian clock function. Life Sciences, 83(15–16), 574–580.
Smeyne, R. J., & Jackson-Lewis, V. (2005). The MPTP model of Parkinson’s disease. Molecular Brain Research, 134(1), 57–66.
Stiasny-Kolster, K., Doerr, Y., Moller, J. C., Hoffken, H., Behr, T. M., Oertel, W. H., et al. (2005). Combination of ‘idiopathic’ REM sleep behaviour disorder and olfactory dysfunction as possible indicator for alpha-synucleinopathy demonstrated by dopamine transporter FP-CIT-SPECT. Brain, 128, 126–137.
Stiasny-Kolster, K., Mayer, G., Schafer, S., Moller, J. C., Heinzel-Gutenbrunner, M., & Oertel, W. H. (2007). The REM sleep behavior disorder screening questionnaire—A new diagnostic instrument. Movement Disorders, 22(16), 2386–2393.
Takahashi, J. S., Hong, H. K., Ko, C. H., & McDearmon, E. L. (2008). The genetics of mammalian circadian order and disorder: Implications for physiology and disease. Nature Reviews Genetics, 9(10), 764–775.
Terazono, H., Mutoh, T., Yamaguchi, S., Kobayashi, M., Akiyama, M., Udo, R., et al. (2003). Adrenergic regulation of clock gene expression in mouse liver. Proceedings of National Academy of Sciences USA, 100(11), 6795–6800.
Torres-Farfan, C., Abarzua-Catalan, L., Valenzuela, F. J., Mendez, N., Richter, H. G., Valenzuela, G. J., et al. (2009). Cryptochrome 2 expression level is critical for adrenocorticotropin stimulation of cortisol production in the capuchin monkey adrenal. Endocrinology, 150(6), 2717–2722.
Ueda, H. R., Hayashi, S., Chen, W., Sano, M., Machida, M., Shigeyoshi, Y., et al. (2005). System-level identification of transcriptional circuits underlying mammalian circadian clocks. Nature Genetics, 37(2), 187–192.
Ukai-Tadenuma, M., Yamada, R. G., Xu, H., Ripperger, J. A., Liu, A. C., & Ueda, H. R. (2011). Delay in feedback repression by cryptochrome 1 is required for circadian clock function. Cell, 144(2), 268–281.
Um, J. H., Pendergast, J. S., Springer, D. A., Foretz, M., Viollet, B., Brown, A., et al. (2011). AMPK regulates circadian rhythms in a tissue- and isoform-specific manner. PLoS One, 6(3), e18450.
Um, J. H., Yang, S., Yamazaki, S., Kang, H., Viollet, B., Foretz, M., et al. (2007). Activation of 5′-AMP-activated kinase with diabetes drug metformin induces casein kinase Iepsilon (CKIepsilon)-dependent degradation of clock protein mPer2. Journal of Biological Chemistry, 282(29), 20794–20798.
Vieira, E., Nilsson, E. C., Nerstedt, A., Ormestad, M., Long, Y. C., Garcia-Roves, P. M., et al. (2008). Relationship between AMPK and the transcriptional balance of clock-related genes in skeletal muscle. American Journal of Physiology: Endocrinology and Metabolism, 295(5), E1032–E1037.
Vingtdeux, V., Davies, P., Dickson, D. W., & Marambaud, P. (2011). AMPK is abnormally activated in tangle- and pre-tangle-bearing neurons in Alzheimer’s disease and other tauopathies. Acta Neuropathologica, 121(3), 337–349.
Wardas, J. (2002). Neuroprotective role of adenosine in the CNS. Polish Journal of Pharmacology, 54(4), 313–326.
Wu, Y., Li, X., Zhu, J. X., Xie, W., Le, W., Fan, Z., et al. (2011). Resveratrol-activated AMPK/SIRT1/autophagy in cellular models of Parkinson’s disease. Neurosignals, 19(3), 163–174.
Acknowledgments
This study was supported partially by Grants-in-Aid for Scientific Research on Priority Areas “Cancer” (S.O. 20014016) from the Ministry of Education, Culture, Sport, Science and Technology of Japan; for Scientific Research (B) (S.O. 21390047); for Challenging Exploratory Research (S.O. 21659041); and for the Encouragement of Young Scientists (N.M. 20790137) from the Japan Society for the Promotion of Science.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Akane Hayashi and Naoya Matsunaga contributed equally to this study.
Rights and permissions
About this article
Cite this article
Hayashi, A., Matsunaga, N., Okazaki, H. et al. A Disruption Mechanism of the Molecular Clock in a MPTP Mouse Model of Parkinson’s Disease. Neuromol Med 15, 238–251 (2013). https://doi.org/10.1007/s12017-012-8214-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-012-8214-x