Abstract
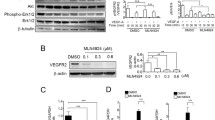
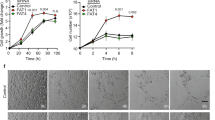
Accumulating evidence indicates that factor associated with suicide ligand (FasL) is a bidirectional regulator. At higher dosage, soluble FasL induced the apoptosis of Fas-expressing brain endothelial cells. Reduced concentration of soluble FasL (sFasL), however, promoted the proliferation and migration of brain endothelial cells. The effect of sFasL on endothelial cells proliferation was completely abolished by silencing Fas expression using siRNA. Treating brain endothelial cells with low-dose sFasL led to increased secretion of VEGF and up-regulated expression of FLIP, TRAF, and NF-κB that played a crucial role in the proliferation of endothelial cells. Our results further underscore the potential stimulating role of Fas/FasL interaction in angiogenesis.




Similar content being viewed by others
Abbreviations
- Fas:
-
Factor associated with suicide
- FasL:
-
Fas ligand
- TNF:
-
Tumor necrosis factor
- FADD:
-
Fas-associated death domain protein
- FLIP:
-
Flice-like inhibitory protein
- NF-κB:
-
Nuclear factor Κb
- VEGF:
-
Vascular endothelial growth factor
- EMSA:
-
Electrophoretic mobility shift assay
References
Fahmy, R. G., Dass, C. R., Sun, L. Q., Chesterman, C. N., & Khachigian, L. M. (2003). Transcription factor Egr-1 supports FGF-dependent angiogenesis during neovascularization and tumor growth. Nature Medicine, 9, 1026–1032.
Lamalice, L., Le Boeuf, F., & Huot, J. (2007). Endothelial cell migration during angiogenesis. Circulation Research, 100, 782–794.
Nagata, S., & Golstein, P. (1995). The Fas death factor. Science, 267, 1449–1456.
Clement, M. V., & Stamenkovic, I. (1994). Fas and tumor necrosis factor receptor-mediate cell death: Similarities and distinctions. Journal of Experimental Medicine, 180, 557–567.
Richardson, B. C., Lalwani, N. D., Johnson, K. J., & Marks, R. M. (1994). Fas ligation triggers apoptosis in macrophages but not endothelial cells. European Journal of Immunology, 24, 2640–2645.
Biancone, L., Martino, A. D., Orlandi, V., Conaldi, P. G., Toniolo, A., & Camussi, G. (1997). Development of inflammatory angiogenesis by local stimulation of Fas in vivo. Journal of Experimental Medicine, 186, 147–152.
Alderson, M. R., Armitage, R. J., Maraskovsky, E., Tough, T. W., Roux, E., Schooley, K., et al. (1993). Fas transduces activation signals in normal human T lymphocytes. Journal of Experimental Medicine, 178, 2231–2235.
Freiberg, R. A., Spencer, D. M., Choate, K. A., Duh, H. J., Schreiber, S. L., Crabtree, G. R., et al. (1997). Fas signal transduction triggers either proliferation or apoptosis in human fibroblasts. Journal of Investigative Dermatology, 108, 215–219.
Hyer, M. L., Samuel, T., & Reed, J. C. (2006). The FLIP-side of Fas signaling. Clinical Cancer Research, 12, 929–5931.
Zhou, L. F., Zhang, M. S., Yin, K. S., Ji, Y., Xie, W. P., Cui, X. F., et al. (2006). Effects of adenoviral gene transfer of mutated IkappaBalpha, a novel inhibitor of NF-kappaB, on human monocyte-derived dendritic cells. Acta Pharmacologica Sinica, 27, 609–616.
Gu, X. Y., Zhou, L. F., Zhang, M. S., Dai, W. J., Chen, S. Y., He, S. H., et al. (2009). Targeted NF-kappaB inhibition of asthmatic serum-mediated human monocyte-derived dendritic cell differentiation in a transendothelial trafficking model. Cellular Immunology, 260, 14–20.
Seghezzi, G., Patel, S., Ren, C. J., Gualandris, A., Pintucci, G., Robbins, E. S., et al. (1998). Fibroblast growth factor-2 (FGF-2) induces vascular endothelial growth factor (VEGF) expression in the endothelial cells of forming capillaries: An autocrine mechanism contributing to angiogenesis. Journal of Cell Biology, 141, 1659–1673.
Herzog, B., Pellet-Many, C., Britton, G., Hartzoulakis, B., & Zachary, I. C. (2011). VEGF binding to NRP1 is essential for VEGF stimulation of endothelial cell migration, complex formation between NRP1 and VEGFR2, and signaling via FAK Tyr407 phosphorylation. Molecular Biology of the Cell, 22, 2766–2776.
Rossi, B., Angiari, S., Zenaro, E., Budui, S. L., & Constantin, G. (2001). Vascular inflammation in central nervous system diseases: Adhesion receptors controlling leukocyte-endothelial interactions. Journal of Leukocyte Biology, 89, 539–556.
Folkman, J. (2002). Role of angiogenesis in tumor growth and metastasis. Seminars in Oncology, 29, 15–18.
Ryan, A. E., Shanahan, F., O’Connell, J., & Houston, A. M. (2006). Fas ligand promotes tumor immune evasion of colon cancer in vivo. Cell Cycle, 5, 246–249.
Gregory, M. S., Saff, R. R., Marshak-Rothstein, A., & Ksander, B. R. (2007). Control of ocular tumor growth and metastatic spread by soluble and membrane Fas ligand. Cancer Research, 67, 11951–11958.
Hohlbaum, A. M., Moe, S., & Marshak-Rothstein, A. (2000). Opposing effects of transmembrane and soluble Fas ligand expression on inflammation and tumor cell survival. Journal of Experimental Medicine, 191, 1209–1220.
Beck, H., & Plate, K. H. (2009). Angiogenesis after cerebral ischemia. Acta Neuropathologica, 117, 481–496.
Broughton, B. R., Reutens, D. C., & Sobey, C. G. (2009). Apoptotic mechanisms after cerebral ischemia. Stroke, 40, e331–e339.
Acknowledgments
This work was supported by Grants from the National Natural Science Foundation of China (81171659) and the Natural Science Foundation of Jiangsu Province (BE2010768) to C.B. Zhang.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C., Gao, F., Teng, F. et al. Fas/FasL Complex Promotes Proliferation and Migration of Brain Endothelial Cells Via FADD–FLIP–TRAF–NF-κB Pathway. Cell Biochem Biophys 71, 1319–1323 (2015). https://doi.org/10.1007/s12013-014-0351-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0351-4