Abstract
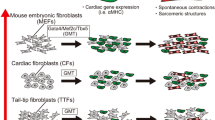
Fibroblasts can be reprogrammed by ectopic expression of reprogramming factors to yield induced pluripotent stem (iPS) cells that are capable of transdifferentiating into diverse types of somatic cell lines. In this study, we examined if functional cardiomyocytes (CMs) can be produced from mouse cardiac fibroblasts (CFs), using iPS cell factor-based reprogramming. CFs were isolated from Oct4-GFP-C57 mice and infected with a retrovirus expressing the Yamanaka reprogramming factors, Oct4, Sox2, Klf4, and c-Myc to reprogram the CFs into a CF-iPS cell line. Primary mouse embryonic fibroblast cells (MEFs) were used as a control. We found that the dedifferentiated CF-iPS cells showed similar biological characteristics (morphology, pluripotent factor expression, and methylation level) as embryonic stem cells (ESs) and MEF-iPS cells. We used the classical embryoid bodies (EBs)-based method and a transwell CM co-culture system to simulate the myocardial paracrine microenvironment for performing CF-iPS cell cardiogenic differentiation. Under this simulated myocardial microenvironment, CF-iPS cells formed spontaneously beating EBs. The transdifferentiated self-beating cells expressed cardiac-specific transcription and structural factors and also displayed typical myocardial morphology and electrophysiological characteristics. CFs can be dedifferentiated into iPS cells and further transdifferentiated into CMs. CFs hold great promise for CM regeneration as an autologous cell source for functional CM in situ without the need for exogenous cell transplantation in ischemic heart disease.




Similar content being viewed by others
References
Segers, V. F., & Lee, R. T. (2008). Stem-cell therapy for cardiac disease. Nature, 451, 937–942.
Reinecke, H., Minami, E., Zhu, W. Z., & Laflamme, M. A. (2008). Cardiogenic differentiation and transdifferentiation of progenitor cells. Circulation Research, 103, 1058–1071.
Dong, H. Y., Xu, Z. W., Zhang, Z. M., et al. (2009). Mobilization of bone marrow-derived Nk2-5+ cardiac progenitor cells under acute myocardial ischemia. Acta Physiologica Sinica, 61, 189–193.
Nussbaum, J., Minami, E., Laflamme, M. A., Virag, J. A., Ware, C. B., Masino, A., et al. (2007). Transplantation of undifferentiated murine embryonic stem cells in the heart: Teratoma formation and immune response. FASEB Journal, 21, 1345–1357.
Kuzmenkin, A., Liang, H., Xu, G., et al. (2009). Functional characterization of cardiomyocytes derived from murine induced pluripotent stem cells in vitro. FASEB Journal, 23, 4168–4180.
Zhang, J., Wilson, G. F., Soerens, A. G., Koonce, C. H., Yu, J., Palecek, S. P., et al. (2009). Functional cardiomyocytes derived from human induced pluripotent stem cells. Circulation Research, 104, e30–e41.
Nelson, T. J., Martinez-Fernandez, A., Yamada, S., et al. (2009). Repair of acute myocardial infarction by human stemness factors induced pluripotent stem cells. Circulation, 120, 408–416.
Camelliti, P., Borg, T. K., & Kohl, Peter. (2005). Structural and functional characterisation of cardiac fibroblasts. Cardiovascular Research, 65, 40–51.
Anversa, P., & Nadal-Ginard, B. (2002). Myocyte renewal and ventricular remodeling. Nature, 415, 240–243.
Etzion, S., Barbash, I. M., Feinberg, M. S., et al. (2002). Cellular cardiomyoplasty of cardiac fibroblasts by adenoviral delivery of MyoD ex vivo: An unlimited source of cells for myocardial repair. Circulation, 106, 125–130.
Ieda, M., Fu, J. D., Delgado-Olguin, P., Vedantham, V., Hayashi, Y., Bruneau, B. G., et al. (2010). Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell, 142, 375–386.
Hochedlinger, K., & Jaenisch, R. (2006). Nuclear reprogramming and pluripotency. Nature, 441, 1061–1067.
Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126, 663–676.
Okita, K., Ichisaka, T., & Yamanaka, S. (2007). Generation of germline-competent induced pluripotent stem cells. Nature, 448, 313–317.
Ieda, M., Tsuchihashi, T., Ive, K. N., et al. (2009). Cardiac fibroblasts regulate myocardial proliferation through beta1 integrin signaling. Developmental Cell, 16, 233–244.
Jiang, B., Dong, H., Zhang, Z., et al. (2007). Hypoxic response elements control expression of human vascular endothelial growth factor165 genes transferred to ischemia myocardium in vivo and in vitro. Journal of Gene Medicine, 9, 788–796.
Pucéat, M. (2008). Protocols for cardiac differentiation of embryonic stem cells. Methods, 45, 168–171.
Aasen, T., Raya, A., Barrero, M. J., Garreta, E., Consiglio, A., Gonzalez, F., et al. (2008). Efficient and rapid generation of induced pluripotent stem cells from human keratinocytes. Nature Biotechnology, 26, 1276–1284.
Nakagawa, M., Koyanagi, M., Tanabe, K., et al. (2007). Generation of high quality iPS cells. Neuroscience Research, 58(Suppl 1), S19.
Carey, B. W., Markoulaki, S., Hanna, J., et al. (2009). Reprogramming of murine and human somatic cells using a single polycistronic vector. Proceedings of the National Academy of Sciences, 106(1), 157–162.
Hanna, J., Wernig, M., Markoulaki, S., et al. (2007). Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science, 318, 1920–1923.
Park, I. H., Arora, N., Huo, H., et al. (2008). Disease-specific induced pluripotent stem cells. Cell, 134, 877–886.
Chen, H. S., Kim, C., & Mercola, M. (2009). Electrophysiological challenges of cell-based myocardial repair. Circulation, 120, 2496–2508.
Mummery, C., Ward-van Oostwaard, D., Doevendans, P., et al. (2003). Differentiation of human embryonic stem cells to cardiomyocytes: Role of coculture with visceral endoderm-like cells. Circulation, 107, 2733–2740.
Zhang, Y. M., Hartzell, C., Narlow, M., & Dudley, S. C., Jr. (2002). Stem cell-derived cardiomyocytes demonstrate arrhythmic potential. Circulation, 106, 1294–1299.
Sun, Y., Kiani, M. F., Postlethwaite, A. E., & Weber, K. T. (2002). Infarct scar as living tissue. Basic Research in Cardiology, 97, 343–347.
Zannad, F., Dousset, B., & Alla, F. (2001). Treatment of congestive heart failure: interfering the aldosterone-cardiac extracellular matrix relationship. Hypertension, 38, 1227–1232.
Cho, H. J., Lee, C. S., Kwon, Y. W., et al. (2010). Induction of pluripotent stem cells from adult somatic cells by protein-based reprogramming without genetic manipulation. Blood, 116, 386–395.
Maherali, N., Sridharan, R., Xie, W., et al. (2007). Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell, 1, 55–70.
Maherali, N., Sridharan, R., Xie, W., et al. (2002). Stem cell differentiation requires a paracrine pathway in the heart. FASEB Journal, 16, 1558–1566.
Kofidis, T., de Bruin, J. L., Yamane, T., et al. (2005). Stimulation of paracrine pathways with growth factors enhances embryonic stem cell engraftment and host-specific differentiation in the heart after ischemic myocardial injury. Circulation, 111, 2486–2493.
Sun, Y. (2009). Myocardial repair/remodelling following infarction: Roles of local factors. Cardiovascular Research, 81, 482–490.
Snider, P., Standley, K. N., Wang, J., et al. (2009). Origin of cardiac fibroblasts and the role of periostin. Circulation Research, 105, 934–947.
Yoshida, Y., Takahashi, K., Okita, K., et al. (2009). Hypoxia enhances the generation of induced pluripotent stem cells. Cell Stem Cell, 5, 237–241.
Polo, J. M., Liu, S., Figueroa, M. E., Kulalert, W., Eminli, S., Tan, K. Y., et al. (2010). Cell type of origin influences the molecular and functional properties of mouse induced pluripotent stem cells. Nature Biotechnology, 28, 848–855.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (31071307) to Zhongming Zhang, Ministry of Science and Technology (2009CB941100) and Chinese Academy of Sciences (XDA01010401) to Gang Wang. We thank Dr. Jingwen Yin, Xiaona Chen, Zhen Liu and Yu Fu for their excellent technical assistance and all the members in Prof. Gang Wang’s lab for their helpful discussion in this study. We are grateful to Prof. Anning Lin for his kind help.
Conflict of interest
The authors confirm that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Bo Jiang and Hongyan Dong contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 5 (AVI 611 kb)
Rights and permissions
About this article
Cite this article
Jiang, B., Dong, H., Li, Q. et al. Differentiation of Reprogrammed Mouse Cardiac Fibroblasts into Functional Cardiomyocytes. Cell Biochem Biophys 66, 309–318 (2013). https://doi.org/10.1007/s12013-012-9487-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9487-2