Abstract
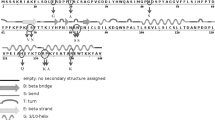
Ubiquitin is a small, globular protein, structure of which has been perfected and conserved through evolution to manage diverse functions in the macromolecular metabolism of eukaryotic cells. Several non-homologous proteins interact with ubiquitin through entirely different motifs. Though the roles of lysines in the multifaceted functions of ubiquitin are well documented, very little is known about the contribution of other residues. In the present study, the importance of two invariant residues, Gln2 and Ser65, have been examined by substituting them with Asn and Asp, respectively, generating single residue variants of ubiquitin UbQ2N and UbS65D. Gln2 and Ser65 form part of parallel G1 β-bulge adjacent to Lys63, a residue involved in DNA repair, cell-cycle regulated protein synthesis and imparting resistance to protein synthesis inhibitors. The secondary structure of variants is similar to that of UbF45W, a structural homologue of wild-type ubiquitin (UbWt). However, there are certain functional differences observed in terms of resistance to cycloheximide, while there are no major differences pertaining to growth under normal conditions, adherence to N-end rule and survival under heat stress. Further, expression of UbQ2N impedes protein degradation by ubiquitin fusion degradation (UFD) pathway. Such differential responses with respect to functions of ubiquitin produced by mutations may be due to interference in the interactions of ubiquitin with selected partner proteins, hint at biomedical implications.







Similar content being viewed by others
References
Pagano, M. (1997). Cell cycle regulation by ubiquitin pathway. FASEB Journal, 11, 1066–1075.
Levinger, L., & Varshavsky, A. (1982). Selective arrangement of ubiquitinated and D1 protein- containing nucleosomes within the drosophila genome. Cell, 28, 375–385.
Jentsch, S., McGrath, J. P., & Varshavsky, A. (1987). The yeast DNA repair gene RAD6 encodes a ubiquitin-conjugating enzyme. Nature, 329, 131–134.
Hochstrasser, M., & Varshavsky, A. (1990). In vivo degradation of a transcriptional regulator: The yeast Matα2 repressor. Cell, 61, 697–708.
Varshavsky, A. (1997). The ubiquitin system. Trends in Biochemical Sciences, 22, 383–387.
Weissmann, A. M. (1997). Regulating protein degradation by ubiquitination. Immunology Today, 18, 189–196.
Galan, J. M., Moreau, V., Andre, B., Volland, C., & Haguenauer-Tsapis, R. (1996). Ubiquitination mediated by the Npi1p/Rsp5p ubiquitin protein ligase is required for endocytosis of the yeast uracil permease. Journal of Biological Chemistry, 271, 10946–10952.
Hershko, A., & Ciechanover, A. (1998). The ubiquitin system. Annual Review of Biochemistry, 67, 425–479.
Pickart, C., & Fushman, D. (2004). Polyubiquitin chains: Polymeric protein signals. Current Opinion in Chemical Biology, 8, 610–616.
Schwartz, A. L., & Ciechanover, A. (2009). Targeting proteins for destruction by the ubiquitin system: Implications for human pathobiology. Annual Review of Pharmacology and Toxicology, 49, 73–96.
Reinstein, E., & Ciechanover, A. (2006). Protein degradation and human diseases: The ubiquitin connection. Annals of Internal Medicine, 145, 676–684.
Gavilanes, J. G., de Buitrago, G. G., Castells, R. P., & Rodrigues, R. (1982). Isolation, characterization, and amino acid sequence of a ubiquitin like protein from insect eggs. Journal of Biological Chemistry, 257, 10267–10270.
Watson, D. C., Leavy, W. B., & Dixon, G. H. (1978). Free ubiquitin is a non-histone protein of trout testis chromatin. Nature, 276, 196–198.
Schlesinger, D. H., Goldstein, G., & Nail, H. D. (1975). Complete amino acid sequence of ubiquitin, an adenylate cyclase stimulating polypeptide probably universal in living cells. Biochemistry, 14, 2214–2218.
Schlesinger, D. H., & Goldstein, G. (1975). Molecular conservation of 74 amino acid sequence of ubiquitin between cattle and man. Nature, 255, 423–424.
Wilkinson, K. D., Cox, M. J., O’Cornnor, B. B., & Shapira, R. (1986). Structure and activities of a variant ubiquitin sequence from bakers’ yeast. 1986. Biochemistry, 25, 4999–5004.
Vierstra, R. D., Langan, S. M., & Schaller, G. E. (1986). Complete amino acid sequence of ubiquitin from the higher plant Avena sativa. Biochemistry, 25, 3105–3108.
Jentsch, S., & Pyrowolakis, G. (2000). Ubiquitin and its kin: How close are the family ties? Trends in Cell Biology, 10, 335–342.
Vijay-kumar, S., Bugg, C. E., & Cook, W. J. (1987). Structure of ubiquitin refined at 1.8 Å resolutions. Journal of Molecular Biology, 194, 513–544.
Briggs, M. S., & Roder, H. (1992). Early hydrogen-bonding events in the folding reaction of ubiquitin. Proceedings of the National Academy of Sciences of the United States of America, 89, 2017–2021.
Loladze, V. V., Ermolenko, D. N., & Makhatadze, G. I. (2001). Heat capacity changes upon burial of polar and nonpolar groups in proteins. Protein Science, 10, 1343–1352.
Loladze, V. V., & Makhatadze, G. I. (2002). Removal of surface charge–charge interactions from ubiquitin leaves the protein folded and very stable. Protein Science, 11, 174–177.
Burch, T. J., & Hass, A. L. (1994). Site-directed mutagenesis of ubiquitin differential roles for arginine in the interaction with ubiquitin-activating enzyme. Biochemistry, 33, 7300–7308.
Mishra, P., Volety, S., Rao, C. M., & Prabha, C. R. (2009). Glutamate64 to glycine substitution in G1 β-bulge of ubiquitin impairs function and stabilizes structure of the protein. Journal of Biochemistry, 146, 563–569.
Khorasnizadadeh, S., Peters, L. D., Butt, T. R., & Roder, H. (1993). Folding and stability of a tryptophan containing mutant of ubiquitin. Biochemistry, 32, 7054–7063.
Cox, J. P. L., Evans, P. A., Packman, L. C., Williams, D. H., & Wolfson, D. N. (1993). Dissecting the structure of a partially folded protein. Circular dichroism and nuclear magnetic resonance studies of peptides from ubiquitin. Journal of Molecular Biology, 234, 483–492.
Platt, G. W., Simpson, S. A., Layfield, R., & Searle, M. S. (2003). Stability and folding kinetics of a ubiquitin mutant with a strong propensity for non-native β-hairpin conformation in the unfolded state. Biochemistry, 42, 13762–13771.
Spence, J., Sadis, S., Haas, A. L., & Finley, D. (1995). Ubiquitin mutant with specific defects in DNA repair and multiubiquitination. Molecular and Cellular Biology, 15, 1265–1273.
Arnason, T., & Ellison, M. J. (1994). Stress resistance in Saccharomyces cerevisiae is strongly correlated with assembly of a novel type of multiubiquitin chain. Molecular and Cellular Biology, 14, 7876–7883.
Chan, A. W. E., Hutchinson, E. G., Harris, D., & Thornton, J. M. (1993). Identification, classification, and analysis of β-bulges in proteins. Protein Science, 2, 1574–1590.
Spence, J., Gali, R., Dittmar, G., Sherman, F., Karin, M., & Finley, D. (2000). Cell cycle–regulated modification of the ribosome by a variant multiubiquitin chain. Cell, 102, 67–76.
Finley, D., Ozkaynak, E., & Varshavsky, A. (1987). The yeast polyubiquitin gene is essential for resistance to high temperatures, starvation, and other stresses. Cell, 48, 1035–1046.
Bachmair, A., Finley, D., & Varshavsky, A. (1986). In vivo half-life of a protein is a function of its amino-terminal residue. Science, 223, 179–186.
Finley, D., Sadis, S., Monia, B. P., Boucher, P., Ecker, D. J., Cooke, S. T., et al. (1994). Inhibition of proteolysis and cell cycle progression in a multiubiquitination-deficient yeast mutant. Molecular and Cellular Biology, 14, 5501–5509.
Ecker, D. J., Butt, T. R., Marsh, J., Sternberg, E. J., Margolis, N., Monia, B. P., et al. (1987). Gene synthesis, expression, structures and functional activities of site-specific mutants of ubiquitin. Journal of Biological Chemistry, 262, 14213–14221.
Chen, P., Johnson, P., Sommer, T., Jentsch, S., & Hochstrasser, M. (1993). Multiple ubiquitin- conjugating enzymes participate in the in vivo degradation of the yeast MATα2 repressor. Cell, 74, 357–369.
Wilkinson, K. D., Urban, M. K., & Haas, A. L. (1980). Ubiquitin is the ATP-dependent proteolysis factor I of rabbit reticulocytes. Journal of Biological Chemistry, 255, 7529–7532.
Hershko, A., Ciechanover, A., & Rose, I. A. (1979). Resolution of the ATP-dependent proteolytic system from reticulocytes: A component that interacts with ATP. Proceedings of the National Academy of Sciences of the United States of America, 76, 3107–3110.
Ciechanover, A., Hod, Y., & Hershko, A. (1978). A heat-stable polypeptide component of an ATP-dependent proteolytic system from reticulocytes. Biochemical and Biophysical Research Communications, 81, 1100–1105.
Haas, A. L., & Wilkinson, K. D. (1985). The large scale purification of ubiquitin from human erythrocytes. Preparative Biochemistry, 15, 49–60.
Johnson, E. S., Bartel, B., Seufert, W., & Varshavsky, A. (1992). Ubiquitin as a degradation signal. The EMBO Journal, 11, 497–505.
Varshavsky, A. (1996). The N-end rule: Functions, mysteries, uses. Proceedings of the National Academy of Sciences of the United States of America, 93, 12142–12149.
Johnson, E. S., Ma, P. C., Ota, I. M., & Varshavsky, A. (1995). A proteolytic pathway that recognizes ubiquitin as a degradation signal. The Journal of Biological Chemistry, 270, 17442–17456.
Hanna, J., Leggett, D. S., & Finley, D. (2003). Ubiquitin depletion as a key mediator of toxicity by translational inhibitors. Molecular and Cellular Biology, 23, 9251–9261.
Kuwajima, K. (1989). The molten globule state as a clue for understanding the folding and cooperativity of globularprotein structure. Proteins, 6, 87–103.
Buck, M., Radford, S. E., & Dobson, C. M. (1993). A partially folded state of hen egg-white lysozyme in trifluoroethanol: structural characterization and implications for protein folding. Biochemistry, 32, 669–678.
Ratnaprabha, C., & Sasidhar, Y. U. (1998). Conformational features of disulfide intact and reduced forms of hen egg white lysozyme in aqueous solution in the presence of trifluoroethanol (TFE): implications for protein folding intermediates. Journal of the Chemical Society Faraday Transactions, 94, 3631–3637.
Hershko, A., & Ciechanover, A. (1992). The ubiquitin system for protein degradation. Annual Review of Biochemistry, 61, 761–807.
Pickart, C. M. (1997). Targeting of substrates to the 26S proteasome. FASEB Journal, 11, 1055–1066.
Ciechanover, A. (1998). The ubiquitin–proteasome pathway: on protein death and cell life. The EMBO Journal, 17, 7151–7160.
Peng, J., Schwartz, D., Elias, J. E., Thoreen, C. C., Cheng, D., Marsischky, G., et al. (2003). A proteomics approach to understanding protein ubiquitination. Nature Biotechnology, 21, 921–926.
Saeki, Y., Kudo, T., Sone, T., Kikuchi, Y., Yokosawa, H., Toh-e, A., et al. (2009). Lysine 63-linked polyubiquitin chain may serve as a targeting signal for the 26S proteasome. EMBO Journal, 28, 359–371.
Baboshina, O. V., & Haas, A. L. (1996). Novel multiubiquitin chain linkages catalyzed by the conjugating enzymes E2EPF and RAD6 are recognized by 26 S proteasome subunit 5*. Journal of Biological Chemistry, 271, 2823–2831.
Koegl, M., Hoppe, T., Schlenker, S., Ulrich, H. D., Mayer, T. U., & Jentsch, S. (1999). A novel ubiquitination factor, E4, is involved in multiubiquitin chain assembly. Cell, 96, 635–644.
Jin, L., Williamson, A., Banerjee, S., Philipp, I., & Rape, M. (2008). Mechanism of ubiquitin-chain formation by the human anaphase-promoting complex. Cell, 133, 653–665.
Wang, L., Mao, X., Ju, D., & Xie, Y. (2004). Rpn4 is a physiological substrate of the Ubr2 ubiquitin ligase. Journal of Biological Chemistry, 279, 55218–55223.
Mulder, L. C. F., & Muesing, M. A. (2000). Degradation of HIV-1 integrase by the N-end rule pathway. Journal of Biological Chemistry, 275, 29749–29753.
Aviel, S., Winberg, G., Massucci, M., & Ciechanover, A. (2000). Degradation of the Epstein-Barr virus latent membrane protein 1 (LMP1) by the ubiquitin-proteasome pathway. Targeting via ubiquitination of the N-terminal residue. Journal of Biological Chemistry, 275, 23491–23499.
Breitschopf, K., Bengal, E., Ziv, T., Admon, A., & Ciechanover, A. (1998). A novel site for ubiquitination: The N-terminal residue, and not internal lysines of MyoD, is essential for conjugation and degradation of the protein. EMBO Journal, 17, 5964–5973.
Acknowledgments
C.R.P. thanks the University Grants Commission, India, for the research grant. The said author is grateful to Prof. Mark Searle and Prof. Daniel Finley for providing plasmids and strains necessary for the study. The author acknowledges the help received from her students Brinda Panchamia and Mrinal Sharma in the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishra, P., Ratna Prabha, C., Rao, C.M. et al. Q2N and S65D Substitutions of Ubiquitin Unravel Functional Significance of the Invariant Residues Gln2 and Ser65. Cell Biochem Biophys 61, 619–628 (2011). https://doi.org/10.1007/s12013-011-9247-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-011-9247-8