Abstract
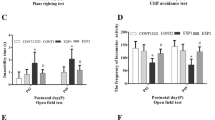
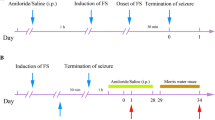
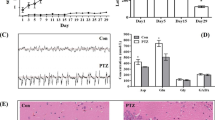
The divalent cation zinc is associated with cortical plasticity. However, the mechanism of zinc in the pathophysiology of cortical injury-associated neurobehavioral damage following neonatal seizures is uncertain. We have previously shown upregulated expression of ZnT-3; MT-3 in hippocampus of neonatal rats submitted to flurothyl-induced recurrent seizures, which was restored by pretreatment with ketogenic diet (KD). In this study, utilizing a novel “twist” seizure model by coupling early-life flurothyl-induced seizures with later exposure to penicillin, we further investigated the long-term effects of KD on cortical expression of zinc homeostasis-related genes in a systemic scale. Ten Sprague-Dawley rats were assigned each averagely into the non-seizure plus normal diet (NS + ND), non-seizure plus KD (NS + KD), recurrent seizures plus normal diet (RS + ND) and recurrent seizures plus KD (RS + KD) group. Recurrent seizures were induced by volatile flurothyl during P9-P21. During P23-P53, rats in NS + KD and RS + KD groups were dieted with KD. Neurological behavioral parameters of brain damage (plane righting reflex, cliff avoidance reflex, and open field test) were observed at P43. At P63, we examined seizure threshold using penicillin, then the cerebral cortex were evaluated for real-time RT-PCR and western blot study. The RS + ND group showed worse performances in neurological reflex tests and reduced latencies to myoclonic seizures induced by penicillin compared with the control, which was concomitant with altered expressions of ZnT-7, MT-1, MT-2, and ZIP7. Specifically, there was long-term elevated expression of ZIP7 in RS + ND group compared with that in NS + ND that was restored by chronic ketogenic diet (KD) treatment in RS + KD group, which was quite in parallel with the above neurobehavioral changes. Taken together, these findings indicate that the long-term altered expression of the metal transporter ZIP7 in adult cerebral cortex might correlate with neurobehavioral damage and reduced seizure threshold following recurrent neonate seizures and further highlights ZIP7 as a candidate for therapeutic target of KD for the treatment of neonatal seizure-induced long-term brain damage.




Similar content being viewed by others
References
Fraga CG, Oteiza PI, Golub MS, et al. (1990) Effects of aluminum on brain lipid peroxidation. Toxicol Lett 51:213–219
Cornett CR, Markesbery WR, Ehmann WD (1998) Imbalances of trace elements related to oxidative damage in Alzheimer’s disease brain. Neurotoxicology 19:339–345
Zaleska MM, Floyd RA (1985) Regional lipid peroxidation in rat brain in vitro: possible role of endogenous iron. Neurochem Res 10:397–410
Prakash A, Bharti K, Majeed AB (2015) Zinc: indications in brain disorders. Fundam Clin Pharmacol 29:131–149
Kim EY, Koh JY, Kim YH, et al. (1999) Zn2+ entry produces oxidative neuronal necrosis in cortical cell cultures. Eur J Neurosci 11:327–334
Szewczyk B (2013) Zinc homeostasis and neurodegenerative disorders. Front Aging Neurosci 5:33
Chimienti F, Aouffen M, Favier A, et al. (2003) Zinc homeostasis-regulating proteins: new drug targets for triggering cell fate. Curr Drug Targets 4:323–338
Berger MM, Shenkin A (1998) Trace elements in trauma and burns. Curr Opin Clin Nutr Metab Care 1:513–517
Yeiser EC, Vanlandingham JW, Levenson CW (2002) Moderate zinc deficiency increases cell death after brain injury in the rat. Nutr Neurosci 5:345–352
Chimienti F, Aouffen M, Favier A (2003) Zinc homeostasis-regulating proteins: new drug targets for triggering cell fate. Curr Drug Targets 4:323–338
Tian T, Ni H, Sun BL (2015) Neurobehavioral deficits in a rat model of recurrent neonatal seizures are prevented by a ketogenic diet and correlate with hippocampal zinc/lipid transporter signals. Biol Trace Elem Res 167:251–258
Yeiser EC, Lerant AA, Casto RM (1999) Free zinc increases at the site of injury after cortical stab wounds in mature but not immature rat brain. Neurosci Lett 277:75–78
Ni H, Yan JZ, Zhang LL, et al. (2012) Long-term effects of recurrent neonatal seizures on neurobehavioral function and related Gene expression and its intervention by inhibitor of cathepsin B. Neurochem Res 37:31–39
Moser VC, Phillips PM, Hedge JM, et al. (2015) Neurotoxicological and thyroid evaluations of rats developmentally exposed to tris(1,3-dichloro-2-propyl)phosphate (TDCIPP) and tris(2-chloro-2-ethyl)phosphate (TCEP). Neurotoxicol Teratol 52:236–247
Souza AC, Souza A, Medeiros LF, et al. (2015) Maternal caffeine exposure alters neuromotor development and hippocampus acetylcholinesterase activity in rat offspring. Brain Res 1595:10–18
Kaizaki A, Tanaka S, Yoshida T, et al. (2014) Maternal MDMA administration in mice leads to neonatal growth delay. J Toxicol Sci 39:33–39
Ni H, Jiang YW, Bo T, et al. (2005) C-Fos, N-methyl-d-aspartate receptor 2C, GABA-A-alpha1 immonoreactivity, seizure latency and neuronal injury following single or recurrent neonatal seizures in hippocampus of Wistar rat. Neurosci Lett 380(1–2):149–154
Johnson MR, Wang K, Smith JB, et al. (2000) Quantitation of dihydropyrimidine dehydrogenase expression by real-time reverse transcription polymerase chain reaction. Anal Biochem 278:175–184
Lugo JN, Swann JW, Anderson AE (2014) Early-life seizures result in deficits in social behavior and learning. Exp Neurol 256:74–80
Holmes GL, Tian C, Hernan AE (2015) Alterations in sociability and functional brain connectivity caused by early-life seizures are prevented by bumetanide. Neurobiol Dis 77:204–219
Torolira D, Suchomelova L, Wasterlain CG (2016) Widespread neuronal injury in a model of cholinergic status epilepticus in postnatal day 7 rat pups. Epilepsy Res 120:47–54
Scholl EA, Dudek FE, Ekstrand JJ (2013) Neuronal degeneration is observed in multiple regions outside the hippocampus after lithium pilocarpine-induced status epilepticus in the immature rat. Neuroscience 252:45–59
Ni H, Feng X, Xiao ZJ, et al. (2011) Dynamic pattern of gene expression of ZnT-4, caspase-3, LC3, and PRG-3 in rat cerebral cortex following flurothyl-induced recurrent neonatal seizures. Biol Trace Elem Res 143:1607–1615
Yan G, Zhang Y, Yu J, et al. (2012) Slc39a7/zip7 plays a critical role in development and zinc homeostasis in zebrafish. PLoS One 7:e42939
Grubman A, Lidgerwood GE, Duncan C, et al. (2014) Deregulation of subcellular biometal homeostasis through loss of the metal transporter, Zip7, in a childhood neurodegenerative disorder. Acta Neuropathol Commun 2:25
Huang L, Kirschke CP, Zhang Y, et al. (2005) The ZIP7 gene (Slc39a7) encodes a zinc transporter involved in zinc homeostasis of the Golgi apparatus. J Biol Chem 280:15456–15463
Liu Y, Batchuluun B, Ho L, et al. (2015) Characterization of zinc influx transporters (ZIPs) in pancreatic β cells: roles in regulating cytosolic zinc homeostasis and insulin secretion. J Biol Chem 290:18757–18769
Myers SA, Nield A, Chew GS, et al. (2013) The zinc transporter, Slc39a7 (Zip7) is implicated in glycaemic control in skeletal muscle cells. PLoS One 8:e79316
Shacka JJ, Lu J, Xie ZL, et al. (2007) Kainic acid induces early and transient autophagic stress in mouse hippocampus. Neurosci Lett 414:57–60
Ni H, Zhao DJ, Tian T (2016) Ketogenic diet change cPLA2/clusterin and autophagy related gene expression and correlate with cognitive deficits and hippocampal MFs sprouting following neonatal seizures. Epilepsy Res 120:13–18
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81271458, 81471337), Jiangsu Province’s Key Provincial Talents Program (RC2011113).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments procedures were in agreement with the principles approved by the National Institutes of Health Guide for the Humane Treatment of Laboratory animals.
Rights and permissions
About this article
Cite this article
Tian, T., Li, LL., Zhang, SQ. et al. Long-Term Effects of Ketogenic Diet on Subsequent Seizure-Induced Brain Injury During Early Adulthood: Relationship of Seizure Thresholds to Zinc Transporter-Related Gene Expressions. Biol Trace Elem Res 174, 369–376 (2016). https://doi.org/10.1007/s12011-016-0730-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0730-3