Abstract
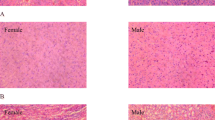
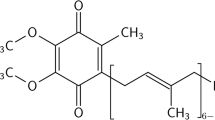
Our previous study showed that chromium malate improved the regulation of blood glucose in mice with alloxan-induced diabetes. The present study was designed to evaluate the 90-day oral toxicity of chromium malate in Sprague-Dawley rats. The present study inspected the effect of chromium malate on glycometabolism, glycometabolism-related enzymes, lipid metabolism, and learning and memory ability in metabolically healthy Sprague-Dawley rats. The results showed that all rats survived and pathological, toxic, feces, and urine changes were not observed. Chromium malate did not cause measurable damage on liver, brain, and kidney. The fasting blood glucose, serum insulin, insulin resistance index, C-peptide, hepatic glycogen, glucose-6-phosphate dehydrogenase, glucokinase, total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and triglyceride levels of normal rats in chromium malate groups had no significant change when compared with control group and chromium picolinate group under physiologically relevant conditions. The serum and organ content of Cr in chromium malate groups had no significant change compared with control group. No significant changes were found in morris water maze test and superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and true choline esterase (TChE) activity. The results indicated that supplementation with chromium malate did not cause measurable toxicity and has no obvious effect on glycometabolism and related enzymes, learning and memory ability, and related enzymes and lipid metabolism of female and male rats. The results of this study suggest that chromium malate is safe for human consumption.







Similar content being viewed by others
References
Economou-Eliopoulos M, Frei R, Atsarou C (2014) Application of chromium stable isotopes to the evaluation of Cr(VI) contamination in groundwater and rock leachates from central Euboea and the Assopos basin (Greece). Catena 122:216–228
Parksa JL, McNeill L, Edwards M (2014) NOM and alkalinity interference in trace-level hexavalent chromium analysis. Talanta 130:226–232
Li Y, Li P, Yu SF, Zhang J, Wang TC, Jia G (2014) miR-3940-5p associated with genetic damage in workers exposed to hexavalent chromium. Toxicol Lett 229:319–326
Hend MAEA, Amal AEB, Afaf MA, Ahmed L, Shery SK, Ayman ZE, Hussein AA (2014) DNA fragmentation, caspase 3 and prostate-specific antigen genes expression induced by arsenic, cadmium, and chromium on nontumorigenic human prostate cells. Biol Trace Elem Res 162:95–105
Suh M, Thompson CM, Kirman CR, Carakostas MC, Hawse LC, Harris MA, Proctora DM (2014) High concentrations of hexavalent chromium in drinking water alter iron homeostasis in F344 rats and B6C3F1 mice. Food Chem Toxicol 65:381–388
Stanley JA, Sivakumar KK, Arosh JA, Burghardt RC, Banu SK (2014) Edaravone mitigates hexavalent chromium-induced oxidative stress and depletion of antioxidant enzymes while estrogen restores antioxidant enzymes in the rat ovary in F1 offspring. Biol Reprod 91:12. doi: 10.1095/biolreprod.113.113332
Deb DD, Parimala G, Devi SS, Chakrabarti T (2012) Role of Carum copticum seeds in modulating chromium-induced toxicity on human bronchial epithelial cells and human peripheral blood lymphocytes. Exp Toxicol Pathol 64:889–897
Di Bona KR, Love S, Rhodes NR, McAdory D, Sinha SH, Kern N, Kent J, Strickland J, Wilson A, Beaird J (2011) Chromium is not an essential trace element for mammals: effects of a “low-chromium” diet. J Biol Inorg Chem 16:381–390
Rudolf E, Cervinka M (2009) Trivalent chromium activates Rac-1 and Src and induces switch in the cell death mode in human dermal fibroblasts. Toxicol Lett 188:236–242
Lewicki S, Zdanowski R, Krzyzowska M, Lewicka A, Debski B, Niemcewicz M, Goniewicz M (2014) The role of Chromium III in the organism and its possible use in diabetes and obesity treatment. Ann Agric Environ Med 21:331–335
Yoshida M, Hatakeyama E, Hosomi R, Kanda S, Nishiyama T, Fukunaga K (2010) Tissue accumulation and urinary excretion of chromium in rats fed diets containing graded levels of chromium chloride or chromium picolinate. J Toxicol Sci 35:485–491
De Lucca PC, Dutrey PL, Villarino ME, Ubios AM (2009) Effect of different doses of hexavalent chromium on mandibular growth and tooth eruption in juvenile Wistar rats. Exp Toxicol Pathol 61:347–352
Farshchi A, Nikfar S, Seyedifar M, Abdollahi M (2013) Effect of chromium on glucose and lipid profiles in patients with type 2 diabetes; a meta-analysis review of randomized trials. J Pharm Pharm Sci 16:99–114
Li F, Wu XY, Zou YM, Zhao T, Zhang M, Feng WW, Yang LQ (2012) Comparing anti-hyperglycemic activity and acute oral toxicity of three different trivalent chromium complexes in mice. Food Chem Toxicol 50:1623–1631
Wu XY, Li F, Xu WD, Zhao JL, Zhao T, Liang LH, Yang LQ (2011) Anti-hyperglycemic activity of chromium (III) malate complex in alloxan-induced diabetic rats. Biol Trace Elem Res 143:1031–1043
Zhang Q, Xiao XH, Li M, Li WH, Yu M, Zhang HB, Ping F, Wang ZX, Zheng J, Xiang HD (2014) miR-375 and miR-30d in the rffect of chromium-containing Chinese medicine moderating glucose metabolism. J Diabetes Res 2014:1–6
Leiva T, Cooke RF, Aboin AC, Drago FL, Gennari R, Vasconcelos JLM (2014) Effects of excessive energy intake and supplementation with chromium propionate on insulin resistance parameters in nonlactating dairy cows. J Anim Sci 92:775–782
Preuss HG, Echard B, Bagchi D, Perricone NV (2013) Comparing effects of carbohydrate (CHO) blockers and trivalent chromium on CHO-induced insulin resistance and elevated blood pressure in rats. J Am Coll Nutr 32:58–65
Hoffman NJ, Penque BA, Habegger KM, Sealls W, Tackett L, Elmendorf JS (2014) Chromium enhances insulin responsiveness via AMPK. J Nutr Biochem 25:565–572
Suksomboon N, Poolsup N, Yuwanakorn A (2014) Systematic review and meta-analysis of the efficacy and safety of chromium supplementation in diabetes. J Clin Pharm Ther 39:292–306
Sharma S, Agrawal RP, Choudhary M, Jain S, Goyal S, Agarwal V (2011) Beneficial effect of chromium supplementation on glucose, HbA1C and lipid variables in individuals with newly onset type-2 diabetes. J Trace Elem Med Biol 25:149–153
Liu L, Jin W, Lv JP (2010) Oral administration of the high-chromium yeast improve blood plasma variables and pancreatic islet tissue in diabetic mice. Biol Trace Elem Res 138:250–264
Morya K, Vachhrajani KD (2014) Impairment of renal structure and function following heterogeneous chemical mixture exposure in rats. Indian J Exp Biol 52:332–343
Castro MP, de Moraes FR, Fujimoto RY, da Cruz C, Belo MAD, de Moraes JRE (2014) Acute toxicity by water containing hexavalent or trivalent chromium in native Brazilian fish, Piaractus mesopotamicus: anatomopathological alterations and mortality. B Environ Contam Toxicol 92:213–219
Mozaffari MS, Baban B, Abdelsayed R, Liu JY, Wimborne H, Rodriguez N, Abebe W (2012) Renal and glycemic effects of high-dose chromium picolinate in db/db mice: assessment of DNA damage. J Nutr Biochem 23:977–985
Tan GY, Zheng SS, Zhang MH, Feng JH, Xie P, Bi JM (2008) Study of oxidative damage in growing-finishing pigs with continuous excess dietary chromium picolinate intake. Biol Trace Elem Res 126:129–140
Andersson MA, Grawe KVP, Karlsson OM, Abramsson-Zetterberg LAG, Hellman BE (2007) Evaluation of the potential genotoxicity of chromium picolinate in mammalian cells in vivo and in vitro. Food Chem Toxicol 45:1097–1106
Bailey MM, Boohaker JG, Sawyer RD, Behling JE, Rasco JF, Jernigan JJ, Hood RD, Vincent JB (2006) Exposure of pregnant mice to chromium picolinate results in skeletal defects in their offspring. Birth Defects Res B 77:244–249
Mahmoud AA, Karam SH, Abdel-Wahhab MA (2006) Chromium-picolinate induced ocular changes: protective role of ascorbic acid. Toxicology 226:143–151
China’s Ministry of Health (2003) Procedures for toxicological assessment of food: Procedure and methods of food safety toxicological assessment. GB15193–2003
Yoshikawa Y, Kishimoto Y, Tagami H, Kanahori S (2013) Assessment of the safety of hydrogenated resistant maltodextrin: reverse mutation assay, acute and 90-day subchronic repeated oral toxicity in rats, and acute no-effect level for diarrhea in humans. J Toxicol Sci 38:459–470
Feist B, Mikula B, Pytlakowska K, Puzio B, Buhl F (2008) Determination of heavy metals by icp-oes and f-aas after preconcentration with 2, 2′-bipyridyl and erythrosine. J Hazard Mater 152:1122–1129
Enomoto T, Ishibashi T, Tokuda K, Ishiyama T, Toma S, Ito A (2008) Lurasidone reverses MK-801-induced impairment of learning and memory in the Morris water maze and radial-arm maze tests in rats. Behav Brain Res 186:197–207
Huang S, Peng WF, Jiang XH, Shao K, Xia LL, Tang YB, Qiu JY (2014) The effect of chromium picolinate supplementation on the pancreas and macroangiopathy in type II diabetes mellitus rats. J Diabetes Res 2014:1–8
Waylan AT, Quinn PRO, Goodband RD, Unruh JA, Nelssen JL, Woodworth JC, Tokach MD (2003) Effects of dietary additions of modified tall oil, chromium nicotinate, and L-carnitine on growth performance, carcass characteristics, and bacon characteristics of growing-finishing pigs. Can J Anim Sci 83:459–467
Hepburn DDD, Xiao J, Bindom S, Vincent JB, O’Donnell J (2003) Nutritional supplement chromium picolinate causes sterility and lethal mutations in Drosophila melanogaster. Abstr Pap Am Chem Soc 225:U31–U31
Stallings DM, Hepburn DDD, Hannah M, Vincent JB, O’Donnell J (2006) Nutritional supplement chromium picolinate generates chromosomal aberrations and impedes progeny development in Drosophila melanogaster. Mutat Res Genet Tox Environ Mutagen 610:101–113
Acknowledgments
This work was supported financially by Specialized Research Fund for the Natural Science Foundation of China (31271850), Graduate innovative projects in Jiangsu University (CXLX13_685). We also appreciate Dr. Samuel Jerry Cobbina for language polishing.
Conflict of Interest
The authors declare that there are no competing interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Weiwei Feng and Huiyu Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Feng, W., Wu, H., Li, Q. et al. Evaluation of 90-day Repeated Dose Oral Toxicity, Glycometabolism, Learning and Memory Ability, and Related Enzyme of Chromium Malate Supplementation in Sprague-Dawley Rats. Biol Trace Elem Res 168, 181–195 (2015). https://doi.org/10.1007/s12011-015-0341-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0341-4