Abstract
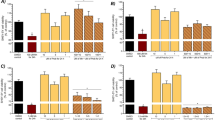
Manganese (Mn) is an essential metal for biological systems; however, occupational or clinical exposure to high levels of Mn can produce a neurological disorder called manganism. Oxidative stress and neuroinflammation play major roles in the Mn-induced neurodegeneration leading to dysfunction of the basal ganglia. We investigated the toxic effects of MnCl2 in an immortalized rat brain endothelial cell line (RBE4) and the protective effects of the radical scavenging aminosalicylic acids, 5-aminosalicylic acid (5-ASA) and 4-aminosalicylic acid (4-PAS). Mn cytotoxicity was determined with 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) reduction and lactate dehydrogenase (LDH) activity. A significant decrease in MTT reduction concomitant with increased LDH release was noted in RBE4 cells exposed for 24 h to MnCl2 (600 and 800 μM; p < 0.0001). Our results establish that compared to 4-PAS, 5-ASA has greater efficacy in protecting RBE4 cells from Mn-induced neurotoxicity after preexposure to MnCl2 800 μM (p < 0.0001).


Similar content being viewed by others
References
Barnes PJ, Karin M (1997) Nuclear factor-kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 336:1066–1071
Blanc P (1990) Poisoning and drug overdose. Appleton & Lange, Norwalk
Bressler JP, Olivi L, Cheong JH, Kim Y, Bannona D (2004) Divalent metal transporter 1 in lead and cadmium transport. Ann N Y Acad Sci 1012:142–152
Cotzias GC, Horiuchi K, Fuenzalida S, Mena I (1968) Chronic manganese poisoning. Clearance of tissue manganese concentrations with persistance of the neurological picture. Neurology 18:376–382
Demeuse P, Kerkhofs A, Struys-Ponsar C, Knoops B, Remacle C, van den Bosch de Aguilar P (2002) Compartmentalized coculture of rat brain endothelial cells and astrocytes: a syngenic model to study the blood–brain barrier. J Neurosci Methods 121:21–31
dos Santos AP, Milatovic D, Au C, Yin Z, Batoreu MC, Aschner M (2010) Rat brain endothelial cells are a target of manganese toxicity. Brain Res 1326:152–161
Durieu-Trautmann O, Federici C, Creminon C, Foignant-Chaverot N, Roux F, Claire M, Strosberg AD, Couraud PO (1993) Nitric oxide and endothelin secretion by brain microvessel endothelial cells: regulation by cyclic nucleotides. J Cell Physiol 155:104–111
Fitzwater SP, Sechler GA, Jave O, Coronel J, Mendoza A, Gilman RH, Friedland JS, Moore DA (2013) Second-line anti-TB drug concentrations for susceptibility testing in the MODS assay. Eur Respir J. doi:10.1183/09031936.00059812
Goncalves E, Almeida LM, Dinis TC (1998) Antioxidant activity of 5-aminosalicylic acid against peroxidation of phosphatidylcholine liposomes in the presence of alpha-tocopherol: a synergistic interaction? Free Radic Res 29:53–66
Herrero Hernandez E, Discalzi G, Valentini C, Venturi F, Chio A, Carmellino C, Rossi L, Sacchetti A, Pira E (2006) Follow-up of patients affected by manganese-induced Parkinsonism after treatment with CaNa2EDTA. Neurotoxicology 27:333–339
Kanski J, Lauderback C, Butterfield DA (2001) 5-Aminosalicylic acid protection against oxidative damage to synaptosomal membranes by alkoxyl radicals in vitro. Neurochem Res 26:23–29
Koller WC, Lyons KE, Truly W (2004) Effect of levodopa treatment for parkinsonism in welders: a double-blind study. Neurology 62:730–733
Legrand C, Bour JM, Jacob C, Capiaumont J, Martial A, Marc A, Wudtke M, Kretzmer G, Demangel C, Duval D et al (1992) Lactate dehydrogenase (LDH) activity of the cultured eukaryotic cells as marker of the number of dead cells in the medium [corrected]. J Biotechnol 25:231–243
Marreilha dos Santos AP, Santos D, Au C, Milatovic D, Aschner M, Batoreu MC (2008) Antioxidants prevent the cytotoxicity of manganese in RBE4 cells. Brain Res 1236:200–205
Mergler D, Baldwin M, Belanger S, Larribe F, Beuter A, Bowler R, Panisset M, Edwards R, de Geoffroy A, Sassine MP, Hudnell K (1999) Manganese neurotoxicity, a continuum of dysfunction: results from a community based study. Neurotoxicology 20:327–342
Milatovic D, Aschner M (2009) Measurement of isoprostanes as markers of oxidative stress in neuronal tissue. Curr Protoc Toxicol, Chapter 12 Unit 12 14
Milatovic D, Zaja-Milatovic S, Gupta RC, Yu Y, Aschner M (2009) Oxidative damage and neurodegeneration in manganese-induced neurotoxicity. Toxicol Appl Pharmacol 240:219–225
Miles AM, Grisham MB (1994) Antioxidant properties of aminosalicylates. Methods Enzymol 234:555–572
Pearson DC, Jourd’heuil D, Meddings JB (1996) The anti-oxidant properties of 5-aminosalicylic acid. Free Radic Biol Med 21:367–373
Prabhakaran K, Ghosh D, Chapman GD, Gunasekar PG (2008) Molecular mechanism of manganese exposure-induced dopaminergic toxicity. Brain Res Bull 76:361–367
Ramesh GT, Ghosh D, Gunasekar PG (2002) Activation of early signaling transcription factor, NF-kappaB following low-level manganese exposure. Toxicol Lett 136:151–158
Roels H, Sarhan MJ, Hanotiau I, de Fays M, Genet P, Bernard A, Buchet JP, Lauwerys R (1985) Preclinical toxic effects of manganese in workers from a Mn salts and oxides producing plant. Sci Total Environ 42:201–206
Rosenstock L, Cullen M (1994) Metals and related compounds. In: Rosenstock L, Cullen M (eds) Textbook of clinical and occupational and environmental medicine. Saunders, Philadelphia
Roux F, Durieu-Trautmann O, Chaverot N, Claire M, Mailly P, Bourre JM, Strosberg AD, Couraud PO (1994) Regulation of gamma-glutamyl transpeptidase and alkaline phosphatase activities in immortalized rat brain microvessel endothelial cells. J Cell Physiol 159:101–113
Sadek AH, Rauch R, Schulz PE (2003) Parkinsonism due to manganism in a welder. Int J Toxicol 22:393–401
Santos AP, Lucas RL, Andrade V, Mateus ML, Milatovic D, Aschner M, Batoreu MC (2012) Protective effects of ebselen (Ebs) and para-aminosalicylic acid (PAS) against manganese (Mn)-induced neurotoxicity. Toxicol Appl Pharmacol 258:394–402
Santos D, Milatovic D, Andrade V, Batoreu MC, Aschner M, Marreilha dos Santos AP (2012) The inhibitory effect of manganese on acetylcholinesterase activity enhances oxidative stress and neuroinflammation in the rat brain. Toxicology 292:90–98
Svartz N (1988) Sulfasalazine: II. Some notes on the discovery and development of salazopyrin. Am J Gastroenterol 83:497–503
UKPAR (2009) Pentasa slow release tablets 1 g. ed. M.a. H.P. R. Agency, 19. United Kingdom: Ferring Pharmaceuticals Limited
Valentine JF (2001) Mesalamine induces manganese superoxide dismutase in rat intestinal epithelial cell lines and in vivo. Am J Physiol Gastrointest Liver Physiol 281:G1044–G1050
Yppolito R, Pappano N, Debattista N, Miskoski S, Bertolotti SG, Garcia NA (2002) On the antioxidant properties of therapeutic drugs: quenching of singlet molecular oxygen by aminosalicylic acids. Redox Rep 7:229–233
Zhang D, He X, Huang S, Li Y (2001) Effect of manganese exposure on brain development in postnatal mice. Wei Sheng Yan Jiu 30:260–262
Zhao F, Cai T, Liu M, Zheng G, Luo W, Chen J (2009) Manganese induces dopaminergic neurodegeneration via microglial activation in a rat model of manganism. Toxicol Sci 107:156–164
Zheng W, Jiang YM, Zhang Y, Jiang W, Wang X, Cowan DM (2009) Chelation therapy of manganese intoxication with para-aminosalicylic acid (PAS) in Sprague–Dawley rats. Neurotoxicology 30:240–248
Acknowledgments
I am thankful to Prof. Eunsook Lee from Meharry Medical College and Dr. Yingchun Yu from Vanderbilt University. The study was supported by FCT (Foundation for Science and Technology of Portugal; SFRH/BD/64128/2009) and the National Institute of Environmental Health Sciences R01 ES 10563 to MA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santos, D., Batoreu, M.C., Aschner, M. et al. Comparison Between 5-Aminosalicylic Acid (5-ASA) and Para-Aminosalicylic Acid (4-PAS) as Potential Protectors Against Mn-Induced Neurotoxicity. Biol Trace Elem Res 152, 113–116 (2013). https://doi.org/10.1007/s12011-012-9597-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-012-9597-0