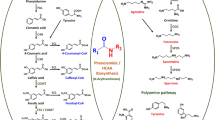
Abstract
Phenylalanine ammonia-lyase (PAL) is one of the most extensively studied enzymes with its crucial role in secondary phenylpropanoid metabolism of plants. Recently, its demand has been increased for aromatic chemical production, but its applications in trans-cinnamic acid production were not much explored. In the present study, a putative PAL gene from Zea mays designated as ZmPAL2 was expressed and characterized in Escherichia coli BL21 (DE3). The recombinant ZmPAL2 exhibited a high PAL activity (7.14 U/mg) and a weak tyrosine ammonia-lyase activity. The optimal temperature of ZmPAL2 was 55 °C, and the thermal stability results showed that about 50 % of enzyme activity remained after a treatment at 60 °C for 6 h. The recombinant ZmPAL2 is a good candidate for the production of trans-cinnamic acid. The vitro conversion indicated that the recombinant ZmPAL2 could effectively catalyze the l-phenylalanine to trans-cinnamic acid, and the trans-cinnamic acid concentration can reach up to 5 g/l.








Similar content being viewed by others
References
Cui, J. D., Qiu, J. Q., Fan, X. W., Jia, S. R., & Tan, Z. L. (2014). Biotechnological production and applications of microbial phenylalanine ammonia lyase: a recent review. Critical Reviews in Biotechnology, 34, 258–268.
Fritz, R. R., Hodgns, D. S., & Abell, C. W. (1976). Phenylalanine ammonia-lyase. Induction and purification from yeast and clearance in mammals. Journal of Biological Chemistry, 251, 4646–4650.
Tanaka, Y., Matsuoka, M., Yamanoto, N., Ohashi, Y., Kano-Murakami, Y., & Ozeki, Y. (1989). Structure and characterization of a cDNA clone for phenylalanine ammonia-lyase from cut-injured roots of sweet potato. Plant Physiology, 90, 1403–1407.
Hahlbrock, K., & Scheel, D. (1989). Physiology and molecular biology of phenylpropanoid metabolism. Annual Review of Plant Biology, 40, 347–369.
Dixon, R. A., & Palva, N. L. (1995). Stress-induced phenylpropanoid metabolism. The Plant Cell, 7, 1085–1097.
Marusich, W. C., Jensen, R. A., & Zamir, L. O. (1981). Induction of L-phenylalanine ammonia-lyase during utilization of phenylalanine as a carbon or nitrogen source in Rhodotorula glutinis. Journal of Bacteriology, 146, 1013–1019.
Koukol, J., & Conn, E. E. (1961). The metabolism of aromatic compounds in higher plants. Journal of Biological Chemistry, 236, 2692–2698.
Aydas, S. B., Ozturk, S., & Aslim, B. (2013). Phenylalanine ammonia lyase (PAL) enzyme activity and antioxidant properties of some cyanobacteria isolates. Food Chemistry, 136, 164–169.
Moffitt, M. C., Louie, G. V., Bowman, M. E., Pence, J., Noel, J. P., & Moore, B. S. (2007). Discovery of two cyanobacterial phenylalanine ammonia lyases: kinetic and structural characterization. Biochemistry, 46, 1004–1012.
Cochrane, F. C., Davin, L. B., & Lewis, N. G. (2004). The Arabidopsis phenylalanine ammonia lyase gene family: kinetic characterization of the four PAL isoforms. Phytochemistry, 65, 1557–1564.
Wang, L., Gamez, A., Archer, H., Abola, E. E., Sarkissian, C. N., Fitzpatrick, P., Wendt, D., Zhang, Y., Vellard, M., Bliesath, J., Bell, S. M., Lemontt, J. F., Scriver, C. R., & Stevens, R. C. (2008). Structural and biochemical characterization of the therapeutic Anabaena variabilis phenylalanine ammonia lyase. Journal of Molecular Biology, 380, 623–635.
Jaliani, H. Z., Farajnia, S., Mohammadi, S. A., Barzegar, A., & Talebi, S. (2013). Engineering and kinetic stabilization of the therapeutic enzyme Anabeana variabilis phenylalanine ammonia lyase. Applied Biochemistry and Biotechnology, 171, 1805–1818.
Louie, G. V., Bowman, M. E., Moffitt, M. C., Baiga, T. J., Moore, B. S., & Noel, J. P. (2006). Structural determinants and modulation of substrate specificity in phenylalanine-tyrosine ammonia-lyases. Chemistry & Biology, 13, 1327–1338.
Logemann, E., Parniske, M., & Hahlbrock, K. (1995). Modes of expression and common structural features of the complete phenylalanine ammonia-lyase gene family in parsley. Proceedings of the National Academy of Sciences, 92, 5905–5909.
Song, J., & Wang, Z. (2009). Molecular cloning, expression and characterization of a phenylalanine ammonia-lyase gene (SmPAL1) from Salvia miltiorrhiza. Molecular Biology Reports, 36, 939–952.
Xu, F., Deng, G., Cheng, S., Zhang, W., Huang, X., Li, L., Cheng, H., Rong, X., & Li, J. (2012). Molecular cloning, characterization and expression of the phenylalanine ammonia-lyase gene from Juglans regia. Molecules, 17, 7810–7823.
Xiang, L. K., & Moore, B. S. (2005). Biochemical characterization of a prokaryotic phenylalanine ammonia lyase. Journal of Bacteriology, 187, 4286–4289.
Ma, W., Wu, M., Wu, Y., Ren, Z., & Zhong, Y. (2013). Cloning and characterisation of a phenylalanine ammonia-lyase gene from Rhus chinensis. Plant Cell Reports, 32, 1179–1190.
Hsieh, L. S., Hsieh, Y. L., Yeh, C. S., Cheng, C. Y., Yang, C. C., & Lee, P. D. (2011). Molecular characterization of a phenylalanine ammonia-lyase gene (BoPAL1) from Bambusa oldhamii. Molecular Biology Reports, 38, 283–290.
Hsieh, L. S., Ma, G. J., Yang, C. C., & Lee, P. D. (2010). Cloning, expression, site-directed mutagenesis and immunolocalization of phenylalanine ammonia-lyase in Bambusa oldhamii. Phytochemistry, 71, 1999–2009.
Cui, J. D., Zhang, S., & Sun, L. M. (2012). Cross-linked enzyme aggregates of phenylalanine ammonia lyase: novel biocatalysts for synthesis of L-phenylalanine. Applied Biochemistry and Biotechnology, 167, 835–844.
Jia, S. R., Cui, J. D., Li, Y., & Sun, A. Y. (2008). Production of L-phenylalanine from trans-cinnamic acids by high-level expression of phenylalanine ammonia lyase gene from Rhodosporidium toruloides in Escherichia coli. Biochemical Engineering Journal, 42, 193–197.
Zhu, L. B., Cui, W. J., Fang, Y. Q., Liu, Y., Gao, X. X., & Zhou, Z. M. (2013). Cloning, expression and characterization of phenylalanine ammonia-lyase from Rhodotorula glutinis. Biotechnology Letters, 35, 751–756.
Alejandra, V. T., Martinez, L. M., Hernandez-Chavez, G., Rocha, M., Martinez, A., Bolivar, F., & Gosset, G. (2015). Production of cinnamic and p-hydroxycinnamic acid from sugar mixtures with engineered Escherichia coli. Microbial Cell Factories, 14, 6.
Gao, Z. M., Wang, X. C., Peng, Z. H., Zheng, B., & Liu, Q. (2012). Characterization and primary functional analysis of phenylalanine ammonia-lyase gene from Phyllostachys edulis. Plant Cell Reports, 31, 1345–1356.
Babich, O. O., Pokrovsky, V. S., Anisimova, N. Y., Sokolov, N. N., & Prosekov, A. Y. (2013). Recombinant L-phenylalanine ammonia lyase from Rhodosporidium toruloides as a potential anticancer agent. Biotechnology and Applied Biochemistry, 60, 316–322.
Hyun, M. W., Yun, Y. H., Kim, J. Y., & Kim, S. H. (2011). Fungal and plant phenylalanine ammonia-lyase. Microbiology, 39, 257–265.
Li, C. L., Bai, Y. C., Chen, H., Zhao, H. X., Shao, J. R., & Wu, Q. (2012). Cloning, characterization and functional analysis of a phenylalanine ammonia-lyase gene (FtPAL) from Fagopyrum tataricum Gaertn. Plant Molecular Biology Reporter, 30, 1172–1182.
Hu, G. S., Jia, J. M., Hur, Y. J., Chung, Y. S., Lee, J. H., Yun, D. J., Chung, W. S., Yi, G. H., Kim, T. H., & Kim, D. H. (2011). Molecular characterization of phenylalanine ammonia lyase gene from Cistanche deserticola. Molecular Biology Reports, 38, 3741–3750.
Rosler, J., Krekel, F., Amrhein, N., & Schmid, J. (1997). Maize phenylalanine ammonia-lyase has tyrosine ammonia-lyase activity. Plant Physiology, 113, 175–179.
Burt, S. (2004). Essential oils: their antibacterial properties and potential applications in foods—a review. International Journal of Food Microbiology, 94, 223–253.
Hoskins, J. A. (1984). The occurrence, metabolism and toxicity of cinnamic acid and related compounds. Journal of Applied Toxicology, 4, 283–292.
Miyamoto, K., Sasaki, M., Minamisawa, Y., Kurahashi, Y., Kano, H., & Ishikawa, S. (2004). Evaluation of in vivo biocompatibility and biodegradation of photocrosslinked hyaluronate hydrogels (HADgels). Journal of Biomedical Materials Research. Part A, 70, 550–559.
Edwards, M., Rourk, P. M., Riby, P. G., & Mendham, A. P. (2014). Not quite the last word on the Perkin reaction. Tetrahedron, 70, 7245–7252.
Wall, V. M., Eisenstadt, A., Ager, D. J., & Laneman, S. A. (1999). The Heck reaction and cinnamic acid synthesis by heterogeneous catalysis. Platinum Metals Review, 43, 138–145.
Mitra, A. K., De, A., & Karchaudhuri, N. (1999). Application of microwave irradiation techniques for the syntheses of cinnamic acids by Doebner condensation. Synthetic Communications, 29, 573–581.
Nijkamp, K., Van Luijk, N., DeBont, J. A., & Wery, J. (2005). The solvent-tolerant Pseudomonas putida S12 as host for the production of cinnamic acid from glucose. Applied Microbiology and Biotechnology, 69, 170–177.
Noda, S., Miyazaki, T., Miyoshi, T., Miyake, M., Okai, N., Tanaka, T., Ogino, C., & Kondo, A. (2011). Cinnamic acid production using Streptomyces lividans expressing phenylalanine ammonia lyase. Journal of Industrial Microbiology & Biotechnology, 38, 643–648.
Hsieh, L. S., Yeh, C. S., Pan, H. C., Cheng, C. Y., Yang, C. C., & Lee, P. D. (2010). Cloning and expression of a phenylalanine ammonia-lyase gene (BoPAL2) from Bambusa oldhamii in Escherichia coli and Pichia pastoris. Protein Expression and Purification, 71, 224–230.
Kyndt, J. A., Meyer, T. E., Cusanovich, M. A., & Van Beeumen, J. J. (2002). Characterization of a bacterial tyrosine ammonia lyase, a biosynthetic enzyme for the photoactive yellow protein. FEBS Letters, 512, 240–244.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
MacDonald, M. J., & D'Cunha, G. B. (2007). A modern view of phenylalanine ammonia lyase. Biochemistry and Cell Biology, 85, 273–282.
Xue, Z., McCluskey, M., Cantera, K., Sariaslani, F. S., & Huang, L. (2007). Identification, characterization and functional expression of a tyrosine ammonia-lyase and its mutants from the photosynthetic bacterium Rhodobacter sphaeroides. Journal of Industrial Microbiology & Biotechnology, 34, 599–604.
Bartsch, S., & Bornscheuer, U. T. (2010). Mutational analysis of phenylalanine ammonia lyase to improve reactions rates for various substrates. Protein Engineering Design and Selection, 23, 929–933.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (31200443), Program for New Century Excellent Talents in University (NCET-11-0988), and Excellent Youth Foundation of Jiangsu Province of China (BK2012038). The authors are also grateful to National Science and Technology Support Program (2012BAD32B06) and PAPD for partial funding of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zang, Y., Jiang, T., Cong, Y. et al. Molecular Characterization of a Recombinant Zea mays Phenylalanine Ammonia-Lyase (ZmPAL2) and Its Application in trans-Cinnamic Acid Production from l-Phenylalanine. Appl Biochem Biotechnol 176, 924–937 (2015). https://doi.org/10.1007/s12010-015-1620-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1620-4