Abstract
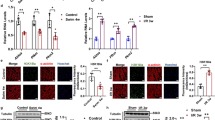
Dehydroepiandrosterone (DHEA) has been proposed to regulate muscle dystrophy, while the underlying mechanisms for its protection against muscle atrophy are unknown. The present study was carried out to improve our understanding of the mechanism of DHEA’s protective effect on muscle cells. We observed that DHEA significantly decreased the loss of cell death associated with H2O2-induced toxicity. Pretreating the muscle cells with DHEA led to a reduction of the intracellular accumulation of reactive oxygen species (ROS) in response to H2O2. In addition, DHEA reduced the H2O2-induced phosphorylation of ERK and p38 in a dose-dependent manner. Moreover, DHEA stimulated the activation of Nrf2, which led to the expression of an antioxidant response gene, HO-1. These results suggest that both antioxidants and anti-inflammatory properties mediate DHEA’s effects for protection against muscle atrophy.





Similar content being viewed by others
References
Bodine, S. C., et al. (2001). Identification of ubiquitin ligases required for skeletal muscle atrophy. Science, 294, 1704–1708. doi:10.1126/science.1065874.
Costelli, P., & Baccino, F. M. (2003). Mechanisms of skeletal muscle depletion in wasting syndromes: role of ATP‐ubiquitin‐dependent proteolysis. Current Opinion in Clinical Nutrition and Metabolic Care, 6, 407–412. doi:10.1097/01.mco.0000078984.18774.02.
Reid, M. B., & Li, Y. P. (2001). Cytokines and oxidative signalling in skeletal muscle. Acta Physiologica Scandinavica, 171, 225–232. doi:10.1046/j.1365-201x.2001.00824.x.
Moylan, J. S., & Reid, M. B. (2007). Oxidative stress, chronic disease, and muscle wasting. Muscle & Nerve, 35, 411–429. doi:10.1002/mus.20743.
Mantovani, G., et al. (2002). Quantitative evaluation of oxidative stress, chronic inflammatory indices and leptin in cancer patients: correlation with stage and performance status. International Journal of Cancer, 98, 84–91. doi:10.1002/ijc.10143.
Kang, E. M., et al. (2005). Hematopoietic stem cell transplantation prevents diabetes in NOD mice but does not contribute to significant islet cell regeneration once disease is established. Experimental Hematology, 33, 699–705. doi:10.1016/j.exphem.2005.03.008.
Sullivan-Gunn, M., & Lewandowski, P. (2013). Elevated hydrogen peroxide and decreased catalase and glutathione peroxidase protection are associated with aging sarcopenia. BMC Geriatrics, 13, 104.
Gomes-Marcondes, M. C. C., Smith, H. J., Cooper, J. C., & Tisdale, M. J. (2002). Development of an in-vitro model system to investigate the mechanism of muscle protein catabolism induced by proteolysis-inducing factor. British Journal of Cancer, 86, 1628–1633.
Li, P., Oparil, S., Sun, J.-Z., Thompson, J. A. & Chen, Y.-F. (2003). Fibroblast growth factor mediates hypoxia-induced endothelin-A receptor expression in lung artery smooth muscle cells. Journal of Applied Physiology, 95(2), 643–651. doi:10.1152/japplphysiol.00652.2002
Labrie, F. (2010). DHEA, important source of sex steroids in men and even more in women. Progress in brain research 182:97–148. doi:10.1016/S0079-6123(10)82004-7
Tchernof, A., Calles-Escandon, J., Sites, C. K., & Poehlman, E. T. (1998). Menopause, central body fatness, and insulin resistance: effects of hormone-replacement therapy. Coronary Artery Disease, 9, 503–511
Goldfarb, A. H., McIntosh, M. K., & Boyer, B. T. (1996). Vitamin E attenuates myocardial oxidative stress induced by DHEA in rested and exercised rats. Journal of Applied Physiology, 80(2), 486–490
Mastrocola, R., et al. (2003). Pro-oxidant effect of dehydroepiandrosterone in rats is mediated by PPAR activation. Life Sciences, 73, 289–299. doi:10.1016/S0024-3205(03)00287-X.
Iwasaki, T., et al. (2005). Marked attenuation of production of collagen type I from cardiac fibroblasts by dehydroepiandrosterone. American Journal of Physiology, Endocrinology and Metabolism, 288(6), E1222–1228. doi:10.1152/ajpendo.00370.2004
Yorek, M. A., et al. (2002). Effect of treatment of diabetic rats with dehydroepiandrosterone on vascular and neural function. American Journal of Physiology, Endocrinology and Metabolism, 283(5), E1067–1075. doi:10.1152/ajpendo.00173.2002
Jang, Y. C., et al. (2010). Increased superoxide in vivo accelerates age-associated muscle atrophy through mitochondrial dysfunction and neuromuscular junction degeneration. The FASEB Journal, 24, 1376–1390. doi:10.1096/fj.09-146308.
Park, S. Y., Kim, J.-H., Lee, S. J., & Kim, Y. (2013). Involvement of PKA and HO-1 signaling in anti-inflammatory effects of surfactin in BV-2 microglial cells. Toxicology and Applied Pharmacology, 268, 68–78. doi:10.1016/j.taap.2013.01.017.
Nakaso, K., Ito, S., & Nakashima, K. (2008). Caffeine activates the PI3K/Akt pathway and prevents apoptotic cell death in a Parkinson’s disease model of SH-SY5Y cells. Neuroscience Letters, 432, 146–150. doi:10.1016/j.neulet.2007.12.034.
Weilbacher, A., Gutekunst, M., Oren, M., Aulitzky, W. E., & van der Kuip, H. (2014). RITA can induce cell death in p53-defective cells independently of p53 function via activation of JNK/SAPK and p38. Cell Death and Disease, 5, e1318. doi:10.1038/cddis.2014.284.
Bogoyevitch, M. A., et al. (1996). Stimulation of the stress-activated mitogen-activated protein kinase subfamilies in perfused heart: p38/RK mitogen-activated protein kinases and c-Jun N-terminal kinases are activated by ischemia/reperfusion. Circulation Research, 79, 162–173. doi:10.1161/01.res.79.2.162.
Bowie, A. G., & O’Neill, L. A. J. (2000). Vitamin C inhibits NF-κB activation by TNF via the activation of p38 mitogen-activated protein kinase. The Journal of Immunology, 165, 7180–7188. doi:10.4049/jimmunol.165.12.7180.
Campbell, C. S. G., et al. (2004). The phosphatidylinositol/AKT/atypical PKC pathway is involved in the improved insulin sensitivity by DHEA in muscle and liver of rats in vivo. Life Sciences, 76, 57–70. doi:10.1016/j.lfs.2004.06.017.
Chua, C. K. Z., Henderson, V. W., Dennerstein, L., Ames, D., & Szoeke, C. (2014). Dehydroepiandrosterone sulfate and cognition in midlife, post-menopausal women. Neurobiology of Aging, 35, 1654–1655. doi:10.1016/j.neurobiolaging.2014.01.140.
Yoshida, S., et al. (2010). Dehydroepiandrosterone sulfate is inversely associated with sex-dependent diverse carotid atherosclerosis regardless of endothelial function. Atherosclerosis, 212, 310–315. doi:10.1016/j.atherosclerosis.2010.05.011.
Sinha, M., Saha, A., Basu, S., Pal, K., & Chakrabarti, S. (2010). Aging and antioxidants modulate rat brain levels of homocysteine and dehydroepiandrosterone sulphate (DHEA-S): implications in the pathogenesis of Alzheimer’s disease. Neuroscience Letters, 483, 123–126. doi:10.1016/j.neulet.2010.07.075.
Traish, A. M., Kang, H. P., Saad, F., & Guay, A. T. (2011). Dehydroepiandrosterone (DHEA)—a precursor steroid or an active hormone in human physiology (CME). The Journal of Sexual Medicine, 8, 2960–2982. doi:10.1111/j.1743-6109.2011.02523.x.
Park, E. J., et al. (2013). Induction of HO-1 through p38 MAPK/Nrf2 signaling pathway by ethanol extract of Inula helenium L. reduces inflammation in LPS-activated RAW 264.7 cells and CLP-induced septic mice. Food and Chemical Toxicology, 55, 386–395. doi:10.1016/j.fct.2012.12.027.
Saha, R. N., Jana, M., & Pahan, K. (2007). MAPK p38 regulates transcriptional activity of NF-κB in primary human astrocytes via acetylation of p65. The Journal of Immunology, 179, 7101–7109. doi:10.4049/jimmunol.179.10.7101.
Patel, D. N., et al. (2007). Interleukin-17 Stimulates C-reactive protein expression in hepatocytes and smooth muscle cells via p38 MAPK and ERK1/2-dependent NF-κB and C/EBPβ activation. Journal of Biological Chemistry, 282, 27229–27238. doi:10.1074/jbc.M703250200.
Bowie, A., & O’Neill, L. A. J. (2000). Oxidative stress and nuclear factor-κB activation: a reassessment of the evidence in the light of recent discoveries. Biochemical Pharmacology, 59, 13–23. doi:10.1016/S0006-2952(99)00296-8.
Schmidt, K. N., Amstad, P., Cerutti, P., & Baeuerle, P. A. (1995). The roles of hydrogen peroxide and superoxide as messengers in the activation of transcription factor NF-κB. Chemistry & Biology, 2, 13–22. doi:10.1016/1074-5521(95)90076-4.
Chen, D.-J., Xu, Y.-M., Du, J.-Y., Huang, D.-Y., & Lau, A. T. Y. (2014). Cadmium induces cytotoxicity in human bronchial epithelial cells through upregulation of eIF5A1 and NF-kappaB. Biochemical and Biophysical Research Communications, 445, 95–99. doi:10.1016/j.bbrc.2014.01.146.
Acknowledgments
This work was supported by the National Research Foundation of Korea funded by the Ministry of Education, Science, and Technology (NRF-2013R1A1A1058835), (NRF-2013M3A9B4076485), (NRF-2013M3A9B4044387), and the Next-Generation BioGreen 21 Program, Rural Development Administration (Project Name: Development of animal models for human neurological and immune disorders).
Conflict of Interest
There is no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeon, S., Hur, J. & Kim, J. DHEA Alleviates Oxidative Stress of Muscle Cells via Activation of Nrf2 Pathway. Appl Biochem Biotechnol 176, 22–32 (2015). https://doi.org/10.1007/s12010-015-1500-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1500-y