Abstract
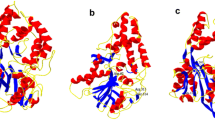
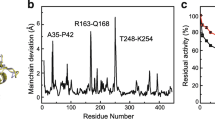
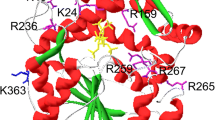
Microbial phytase, a widely used animal feed enzyme, needs to be active and stable in the acidic milieu for better performance in the monogastric gut. Aspergillus niger phytases exhibit an activity dip in the pH range from 3.0 to 3.5. Replacement of amino acids, which changed the pKa of catalytic residues H82 and D362, resulted in alteration of the pH profile of a thermostable phytase from A. niger NII 08121. Substitution P212H in the protein loop at 14 Å distance to the active site amended the pH optimum from 2.5 to pH 3.2 nevertheless with a decrease in thermostability than the wild enzyme. This study described the utility of amino acid replacements based on pKa shifts of catalytic acid/base to modulate the pH profile of phytases.





Similar content being viewed by others
References
Pandey, A., Szakacs, G., Soccol, C. R., Rodriguez-Leon, J. A., & Soccol, V. T. (2001). Bioresource Technology, 77, 203–214.
Vats, P., & Banerjee, U. C. (2004). Enzyme and Microbial Technology, 35, 3–14.
Yi, Z., & Kornegay, E. T. (1996). Animal Feed Science and Technology, 61, 361–368.
Ullah, A. H. J., & Gibson, D. M. (1987). Preparative Biochemistry, 17, 63–91.
Ushasree, M. V., Vidya, J., & Pandey, A. (2014). Process Biochemistry, 49, 1440–1447.
Augspurger, N. R., & Baker, D. H. (2004). Journal of Animal Sciences, 82, 1100–1107.
Joshi, M. D., Sidhu, G., Nielsen, J. E., Brayer, G. D., Withers, S. G., & McIntosh, L. P. (2001). Biochemistry, 40, 10115–10139.
Cockburn, D. W., & Clarke, A. J. (2011). Protein Engineering Design and Selection, 24, 429–437.
Tomschy, A., Brugger, R., Lehmann, M., Svendsen, A., Vogel, K., Kostrewa, D., et al. (2002). Applied Environmental Microbiology, 68, 1907–1913.
Kim, T., Mullaney, E. J., Porres, J. M., Roneker, K. R., Crowe, S., Rice, S., et al. (2006). Applied Environmental Microbiology, 72, 4397–4403.
Larkin, M. A., Blackshields, G., Brown, N. P., Chenna, R., McGettigan, P. A., McWilliam, H., Valentin, F., et al. (2007). Bioinformatics, 23, 2947–2948.
Petersen, B., Petersen, T. N., Andersen, P., Nielsen, M., & Lundegaard, C. (2009). BMC Structural Biology, 9, 6807–6809.
Bas, D. C., Rogers, D. M., & Jensen, J. H. (2008). Proteins, 73, 765–783.
Guex, N., & Peitsch, M. C. (1997). Electrophoresis, 18, 2714–2723.
Lin-Cereghino, J., Wong, W. W., Xiong, S., Giang, W., Luong, L. T., Vu, J., et al. (2005). Biotechniques, 38, 44–48.
Heinonen, J. K., & Lathi, R. J. (1981). Analytical Biochemistry, 113, 313–317.
Cheng, J., Randall, A., & Baldi, P. (2006). Proteins, 62, 1125–1132.
Capriotti, E., Fariselli, P., & Casadio, R. (2005). Nucleic Acid Research, 33, 306–310.
Yin, S., Ding, F., & Dokholyan, N. V. (2007). Nature Methods, 4, 466–467.
Ushasree, M. V., Vidya, J., & Pandey, A. (2014). Biotechnology Letters, 36, 85–91.
Kostrewa, D., Grueninger-Leitch, F., D'Arcy, A., Broger, C., Mitchell, D., Adolphus, P. G. M., et al. (1997). Nature Structural Biology, 4, 185–190.
Shimizu, M. (1993). Bioscience. Biotechnology and Biochemistry, 57, 1364–1365.
Pasamontes, L., Haiker, M., Wyss, M., Tessier, M., & van Loon, A. P. (1997). Applied Environmental Microbiology, 63, 1696–1700.
Ullah, A. H. J., & Sethumadhavan, K. (2003). Biochemical and Biophysical Research Communication., 303, 463–468.
Rodriguez, E., Han, Y., & Lei, X. G. (1999). Biochemical and Biophysical Research Communication, 257, 117–123.
Mullaney, E. J., Daly, C. B., Kim, T., Porres, J. M., Lei, X. G., & Sethumadhavan, K. (2002). Biochemical and Biophysical Research Communication, 297, 1016–1020.
Zhou, C., Xue, Y., & Ma, Y. (2010). Journal of Bioscience and Bioengineering, 110, 12–17.
Thiltgen, G., & Goldstein, R. A. (2012). PLoS ONE. doi:10.1371/ 0046084.
Acknowledgments
One of the authors (MVU) thank the Indian Council of Medical Research, New Delhi for the award of a Senior Research Fellowship to her. Financial support from the Department of Biotechnology, New Delhi (Grant No. GAP 1410) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ushasree, M.V., Vidya, J. & Pandey, A. Replacement P212H Altered the pH–Temperature Profile of Phytase from Aspergillus niger NII 08121. Appl Biochem Biotechnol 175, 3084–3092 (2015). https://doi.org/10.1007/s12010-015-1485-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1485-6