Abstract
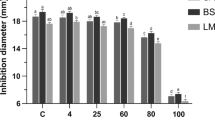
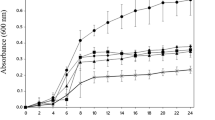
In the present work, the influence of pH and sodium chloride concentration on thermal stability of antimicrobial peptide P34 was evaluated under different time–temperature conditions by a 22 factorial design experiment. At sterilization conditions (121 °C for 20 min), maximum retention (36%) was obtained at pH between 5.5 and 8.5 and sodium chloride concentration between 0.4 and 0.75 mol/l. For boiling conditions (100 °C for 20 min), antimicrobial activity was about 100% combining pH between 6.0 and 8.0 and salt concentration in the range of 0.65 to 1 mol/l. At low temperature pasteurization conditions (30 min at 65 °C), antimicrobial activity was not affected within the pH range from 5.0 to 8.0. For the three time–temperature conditions tested, the antimicrobial activity was minimal at more acidic or alkaline pH. Sodium chloride concentration of 0.65 mol/l increased thermostability of the peptide P34. Combination of sodium chloride and slight alkaline pH may increase the stability of peptide P34, which is essential to the proper utilization of bacteriocins in food industry.


Similar content being viewed by others
References
Asaduzzaman, S. K., & Sonomoto, K. (2009). Lantibiotics: diverse activities and unique modes of action. Journal of Bioscience and Bioengineering, 107, 475–487.
Awuah, G. B., Ramaswamy, H. S., & Economides, A. (2007). Thermal processing and quality: principles and overview. Chemical Engineering and Processing, 46, 584–602.
Cladera-Olivera, F., Caron, G. R., & Brandelli, A. (2004). Bacteriocin production by Bacillus licheniformis strain P40 in cheese whey using response surface methodology. Biochemical Engineering Journal, 21, 53–58.
Cleveland, J., Montville, T. J., Nes, I. F., & Chikindas, M. L. (2001). Bacteriocins: safe, natural antimicrobials for food preservation. International Journal of Food Microbiology, 71, 1–20.
Deegan, L. H., Cotter, P. D., Hill, C., & Ross, R. P. (2006). Bacteriocins: biological tools for bio-preservation and shelf-life extension. International Dairy Journal, 16, 1058–1071.
Delves-Broughton, J., Blackburn, P., Evans, R. J., & Hugenholtz, J. (1996). Applications of the bacteriocin nisin. Antonie Van Leeuwenhoek, 69, 193–202.
Gálvez, A., Abriouel, H., López, R. L., & Omar, N. B. (2007). Bacteriocin-based strategies for food biopreservation. International Journal of Food Microbiology, 120, 51–70.
Gao, Y.-L., Ju, X.-R., & Jiang, H.-H. (2006). Studies on inactivation of Bacillus subtilis spores by high hydrostatic pressure and heat using design of experiments. Journal of Food Engineering, 77, 672–679.
Kwok, K. C., Liang, K. C., & Niranjan, K. (2002). Mathematical modelling of the heat inactivation of trypsin inhibitors in soymilk at 121–154 degrees C. Journal of the Science of Food and Agriculture, 82(3), 243–247.
Leães, F. L., Sant’Anna, V., Vannin, N. G., & Brandelli, A. (2011). Use of byproducts of food industry for production of antimicrobial activity by Bacillus sp. P11. Food and Bioprocess Technology, 4(5), 822–828. doi:10.1007/s11947.010.0410.9.
Makki, F., & Durance, T. D. (1996). Thermal inactivation of lysozyme as influenced by pH, sucrose and sodium chloride and inactivation and preservative effect in beer. Food Research International, 29, 635–645.
Marlow, G. E., Perkyns, J. S., & Pettitt, B. M. (1993). Salt effects in peptide solutions: theory and simulations. Chemical Reviews, 93, 2503–2521.
McAuliffe, O., Ryan, M. P., Ross, R. P., Hill, C., Breeuwer, P., & Abee, T. (1998). Lacticin 3147, a broad spectrum bacteriocin which selectively dissipates the membrane potential. Applied and Environmental Microbiology, 64, 439–445.
Montville, T. J., & Matthews, K. R. (2008). Food microbiology: an introduction (2nd ed.). Washington: ASM Press.
Motta, A. S., & Brandelli, A. (2002). Characterization of an antimicrobial peptide produced by Brevibacterium linens. Journal of Applied Microbiology, 92, 63–70.
Motta, A. S., & Brandelli, A. (2008). Evaluation of environmental conditions for production of bacteriocin-like substance by Bacillus sp. strain P34. World Journal of Microbiology and Biotechnology, 24, 641–646.
Motta, A. S., Cladera-Olivera, F., & Brandelli, A. (2004). Screening for antimicrobial activity among bacteria isolated from the Amazon basin. Brazilian Journal of Microbiology, 35, 307–310.
Motta, A. S., Cannavan, F. S., Tsai, S. M., & Brandelli, A. (2007a). Characterization of a broad range antibacterial substance from a new Bacillus species isolated from Amazon basin. Archives of Microbiology, 188, 367–375.
Motta, A. S., Lorenzini, D. M., & Brandelli, A. (2007b). Purification and partial characterization of an antimicrobial peptide produced by a novel Bacillus sp. isolated from the Amazon basin. Current Microbiology, 54, 282–286.
Myers, R. H., & Montgomery, R. C. (2002). Response surface methodology: process and product optimization using designed experiments. New York: Wiley.
Naim, F., Zareifard, M. R., Zhu, S., Huizing, R. H., Grabowski, S., & Marcotte, M. (2008). Combined effects of heat, nisin and acidification on the inactivation of Clostridium sporogenes spores in carrot-alginate particles: from kinetics to process validation. Food Microbiology, 25, 936–941.
Pedersen, P. B., Bjørnvad, M. E., Rasmussen, M. D., & Petersen, J. N. (2002). Cytotoxic potential of industrial strains of Bacillus spp. Regulatory Toxicology and Pharmacology, 36, 155–161.
Polydera, A. C., Stoforos, N. G., & Taoukis, P. S. (2003). Comparative shelf life study and vitamin C loss kinetics in pasteurised and high pressure processed reconstituted orange juice. Journal of Food Engineering, 60, 21–29.
Sant’Anna, V., Utpott, M., Cladera-Olivera, F., & Brandelli, A. (2010). Kinetic modeling of the thermal inactivation of bacteriocin-like inhibitory substance P34. Journal of Agricultural and Food Chemistry, 58, 3147–3152.
Sant’Anna, V., Utpott, M., Cladera-Olivera, F., & Brandelli, A. (2011a). Influence of pH and sodium chloride on kinetics of thermal inactivation of the bacteriocin-like substance P34. Journal of Applied Microbiology, 110, 156–162.
Sant’Anna, V., Malheiros, O. S., & Brandelli, A. (2011b). Lipossome encapsulation protects bacteriocin-like substance P34 against Maillard reaction products. Food Research International, 44, 326–330.
Sobrino-López, A., & Martín-Belloso, O. (2008). Use of nisin and other bacteriocins for preservation of dairy products. International Dairy Journal, 18, 329–343.
Wandling, L. R., Sheldon, B. W., & Foegeding, P. M. (1999). Nisin in milk sensitizes Bacillus spores to heat and prevents recovery of survivors. Journal of Food Protection, 62, 492–498.
Wirjantoro, T. I., Lewis, M. J., Grandison, A. S., Williams, G. C., & Delves-Broughton, J. (2001). The effect of nisin on the keeping quality of reduced heat-treated milks. Journal of Food Protection, 64, 213–219.
Acknowledgements
The authors thank the financial support of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil. M. Utpott received a fellowship from Fundação de Amparo a Pesquisa do Rio Grande do Sul (FAPERGS), Brazil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sant’Anna, V., Utpott, M., Cladera-Olivera, F. et al. Antimicrobial Activity of Peptide P34 During Thermal Processing. Food Bioprocess Technol 6, 73–79 (2013). https://doi.org/10.1007/s11947-011-0633-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-011-0633-4