Abstract
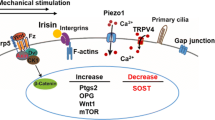
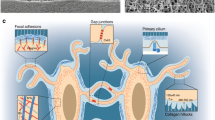
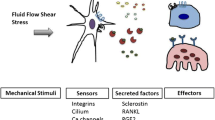
Bone adaptation to changes in mechanical stimuli occurs by adjusting bone formation and resorption by osteoblasts and osteoclasts, to maintain optimal bone mass. Osteocytes coordinate the actions of these cells on the bone surface by sensing mechanical forces and producing cytokines that increase or prevent osteoblast and osteoclast differentiation and function. Channels formed by connexins (Cxs) and, in particular, connexin 43 (Cx43) in osteoblasts and osteocytes are central part of this mechanism to control bone mass. Cx43 hemichannels are opened by fluid flow and mediate the anti-apoptotic effect of mechanical stimulation in vitro, suggesting that Cx43 participates in mechanotransduction. However, mice lacking Cx43 in osteoblasts and/or osteocytes show an increased anabolic response to loading and decreased catabolic response to unloading. This evidence suggests that Cx43 channels expressed in osteoblastic cells are not required for the response to mechanical stimulation, but mediate the consequence of lack thereof. The molecular basis of these unexpected responses to mechanical stimulation is currently under investigation.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bonewald LF, Johnson ML. Osteocytes, mechanosensing and Wnt signaling. Bone. 2008;42:606–15.
Schirrmacher K, Schmitz I, Winterhager E, et al. Characterization of gap junctions between osteoblast-like cells in culture. Calcif Tissue Int. 1992;51:285–90.
Civitelli R, Beyer EC, Warlow PM, et al. Connexin43 mediates direct intercellular communication in human osteoblastic cell networks. J Clin Invest. 1993;91:1888–96.
Kato Y, Windle JJ, Koop BA, et al. Establishment of an osteocyte-like cell line, MLO-Y4. J Bone Miner Res. 1997;12:2014–23.
Su M, Borke JL, Donahue HJ, et al. Expression of connexin 43 in rat mandibular bone and periodontal ligament (PDL) cells during experimental tooth movement. J Dent Res. 1997;76:1357–66.
Ilvesaro J, Väänänen K, Tuukkanen J. Bone-resorbing osteoclasts contain gap-junctional connexin-43. J Bone Miner Res. 2000;15:919–26.
Yellowley CE, Li Z, Zhou Z. Functional gap junctions between osteocytic and osteoblastic cells. J Bone Miner Res. 2000;15:209–17.
Stains JP, Civitelli R. Gap junctions in skeletal development and function. Biochim Biophys Acta. 2005;1719:69–81.
Pacheco-Costa R, Hassan I, Reginato RD. High bone mass in mice lacking Cx37 Due to defective osteoclast differentiation. J Biol Chem. 2014;289:8508–20.
Goodenough DA, Paul DL. Beyond the gap: functions of unpaired connexon channels. Nat Rev Mol Cell Biol. 2003;4:285–94.
Zhang Y, Paul EM, Sathyendra V, et al. Enhanced osteoclastic resorption and responsiveness to mechanical load in gap junction deficient bone. PLoS One. 2011;6:e23516. Reports the enhanced response to mechanical stimulation in mice with conditional deletion of Cx43 from osteoblasts and osteocytes.
Grimston SK, Watkins MP, Brodt MD, et al. Enhanced periosteal and endocortical responses to axial tibial compression loading in conditional connexin43 deficient mice. PLoS One. 2012;7:e44222. Reports the enhanced response to mechanical stimulation in mice with conditional deletion of Cx43 from osteochondroprogenitors.
Bivi N, Pacheco-Costa R, Brun LR, et al. Absence of Cx43 selectively from osteocytes enhances responsiveness to mechanical force in mice. J Orthop Res. 2013;31:1075–81. Reports the enhanced response to mechanical stimulation in mice with conditional deletion of Cx43 from osteocytes. This manuscript, together with reference 11 and 12 demonstrate the unexpected effect of Cx43 expression in osteoblastic cells blunting the anabolic effect of loading on the periosteal bone surface.
Lloyd SA, Lewis GS, Zhang Y, et al. Connexin 43 deficiency attenuates loss of trabecular bone and prevents suppression of cortical bone formation during unloading. J Bone Miner Res. 2012;27:2359–72.
Plotkin LI, Bellido T. Beyond gap junctions: Connexin43 and bone cell signaling. Bone. 2013;52:157–66.
Lloyd SA, Loiselle AE, Zhang Y, et al. Shifting paradigms on the role of Connexin43 in the skeletal response to mechanical load. J Bone Miner Res. 2014;29:275–86.
Cheng B, Zhao S, Luo J, Luo J, et al. Expression of functional gap junctions and regulation by fluid flow in osteocyte-like MLO-Y4 cells. J Bone Miner Res. 2001;16:249–59.
Ziambaras K, Lecanda F, Steinberg TH, et al. Cyclic stretch enhances gap junctional communication between osteoblastic cells. J Bone Miner Res. 1998;13:218–28.
Robinson JA, Chatterjee-Kishore M, Yaworsky PJ, et al. WNT/beta-catenin signaling is a normal physiological response to mechanical loading in bone. J Biol Chem. 2006;281:31720–8.
Tu X, Rhee Y, Condon KW, et al. Sost downregulation and local Wnt signaling are required for the osteogenic response to mechanical loading. Bone. 2012;50:209–17.
Alford AI, Jacobs CR, Donahue HJ. Oscillating fluid flow regulates gap junction communication in osteocytic MLO-Y4 cells by an ERK1/2 MAP kinase-dependent mechanism. Bone. 2003;33:64–70.
Saunders MM, You J, Trosko JE, et al. Gap junctions and fluid flow response in MC3T3–E1 cells. Am J Physiol Cell Physiol. 2001;281:C1917–25.
Keeting PE, Scott RE, Colvard DS, et al. Development and characterization of a rapidly proliferating, well-differentiated cell line derived from normal adult human osteoblast-like cells transfected with SV40 large T antigen. J Bone Miner Res. 1992;7:127–36.
Romanello M, D’Andrea P. Dual mechanism of intercellular communication in HOBIT osteoblastic cells: a role for gap-junctional hemichannels. J Bone Miner Res. 2001;16:1465–76.
Jorgensen NR, Henriksen Z, Brot C, et al. Human osteoblastic cells propagate intercellular calcium signals by two different mechanisms. J Bone Miner Res. 2000;15:1024–32.
Spray DC, Burt JM. Structure-activity relations of the cardiac gap junction channel. Am J Physiol. 1990;258:C195–205.
Krutovskikh VA, Yamasaki H, Tsuda H, et al. Inhibition of intrinsic gap-junction intercellular communication and enhancement of tumorigenicity of the rat bladder carcinoma cell line BC31 by a dominant-negative connexin 43 mutant. Mol Carcinog. 1998;23:254–61.
Thi MM, Islam S, Suadicani SO, et al. Connexin43 and pannexin1 channels in osteoblasts: who is the “hemichannel”? J Membr Biol. 2012;245:401–9.
Cherian PP, Siller-Jackson AJ, Gu S, et al. Mechanical strain opens connexin 43 hemichannels in osteocytes: a novel mechanism for the release of prostaglandin. Mol Biol Cell. 2005;16:3100–6.
Siller-Jackson AJ, Burra S, Gu S, et al. Adaptation of connexin 43-hemichannel prostaglandin release to mechanical loading. J Biol Chem. 2008;283:26374–82.
Cheng B, Kato Y, Zhao S, et al. PGE(2) is essential for gap junction-mediated intercellular communication between osteocyte-like MLO-Y4 cells in response to mechanical strain. Endocrinology. 2001;142:3464–73.
Cherian PP, Cheng B, Gu S, et al. Effects of mechanical strain on the function of gap junctions in osteocytes are mediated through the prostaglandin EP2 receptor. J Biol Chem. 2003;278:43146–56.
Plotkin LI, Bellido T. Bisphosphonate-induced, hemichannel-mediated, anti-apoptosis through the Src/ERK pathway: a gap junction-independent action of connexin43. Cell Adhes Commun. 2001;8:377–82.
Plotkin LI, Manolagas SC, Bellido T. Transduction of cell survival signals by connexin-43 hemichannels. J Biol Chem. 2002;277:8648–57.
Jiang JX, Cherian PP. Hemichannels formed by connexin 43 play an important role in the release of prostaglandin E(2) by osteocytes in response to mechanical strain. Cell Commun Adhes. 2003;10:259–64.
Burra S, Nicolella DP, Francis WL, et al. Dendritic processes of osteocytes are mechanotransducers that induce the opening of hemichannels. Proc NatlAcad Sci USA. 2010;107:13648–53.
Batra N, Burra S, Siller-Jackson AJ, et al. Mechanical stress-activated integrin alpha5beta1 induces opening of connexin 43 hemichannels. Proc Natl Acad Sci U S A. 2012;109:3359–64.
Batra N, Riquelme MA, Burra S, et al. 14-3-3theta facilitates plasma membrane delivery and function of mechanosensitive connexin 43 hemichannels. J Cell Sci. 2014;127:137–46.
Plotkin LI, Aguirre JI, Kousteni S, et al. Bisphosphonates and estrogens inhibit osteocyte apoptosis via distinct molecular mechanisms downstream of ERK activation. J Biol Chem. 2005;280:7317–25.
Genetos DC, Kephart CJ, Zhang Y. Oscillating fluid flow activation of gap junction hemichannels induces ATP release from MLO-Y4 osteocytes. J Cell Physiol. 2007;212:207–14.
Li J, Liu D, Ke HZ, et al. The P2X7 nucleotide receptor mediates skeletal mechanotransduction. J Biol Chem. 2005;280:42952–9.
Penuela S, Gehi R, Laird DW. The biochemistry and function of pannexin channels. Biochim Biophys Acta. 2013;1828:15–22.
Kitase Y, Barragan L, Jiang JX, et al. Mechanical induction of PGE(2) in osteocytes blocks glucocorticoid induced apoptosis through both the beta-catenin and PKA pathways. J Bone Miner Res. 2010;25:2657–68.
Plotkin LI, Mathov I, Aguirre JI, et al. Mechanical stimulation prevents osteocyte apoptosis: requirement of integrins, Src kinases and ERKs. Am J Physiol Cell Physiol. 2005;289:C633–43.
Weinger JM, Holtrop ME, et al. An ultrastructural study of bone cells: the occurrence of microtubules, microfilaments and tight junctions. Calcif Tissue Res. 1974;14:15–29.
Doty SB. Morphological evidence of gap junctions between bone cells. Calcif Tissue Int. 1981;33:509–12.
Palumbo C, Palazzini S, Marotti G. Morphological study of intercellular junctions during osteocyte differentiation. Bone. 1990;11:401–6.
Grimston SK, Brodt MD, Silva MJ, et al. Attenuated response to in vivo mechanical loading in mice with conditional osteoblast ablation of the Connexin43 gene (Gja1). J Bone Miner Res. 2008;23:879–86.
Ai Z, Fischer A, Spray DC, et al. Wnt-1 regulation of connexin43 in cardiac myocytes. J Clin Invest. 2000;105:161–71.
Talhouk RS, Mroue R, Mokalled M, et al. Heterocellular interaction enhances recruitment of alpha and beta-catenins and ZO-2 into functional gap-junction complexes and induces gap junction-dependant differentiation of mammary epithelial cells. Exp Cell Res. 2008;314:3275–91.
Dbouk HA, Mroue RM, El-Sabban ME, et al. Connexins: a myriad of functions extending beyond assembly of gap junction channels. Cell Commun Signal. 2009;7:4.
Glass DA, Bialek P, Ahn JD, et al. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev Cell. 2005;8:751–64.
Holmen SL, Zylstra CR, Mukherjee A, et al. Essential role of beta-catenin in postnatal bone acquisition. J Biol Chem. 2005;280:21162–8.
Spencer GJ, Utting JC, Etheridge SL, et al. Wnt signalling in osteoblasts regulates expression of the receptor activator of NFkappaB ligand and inhibits osteoclastogenesis in vitro. J Cell Sci. 2006;119:1283–96.
O’Brien CA, Nakashima T, Takayanagi H. Osteocyte control of osteoclastogenesis. Bone. 2013;54:258–63.
Javaheri B, Stern A, Lara N, et al. Deletion of a single beta-catenin allele in osteocytes abolishes the bone anabolic response to loading. J Bone Miner Res. 2013;29:705–15.
Bivi N, Condon KW, Allen MR, et al. Cell autonomous requirement of connexin 43 for osteocyte survival: consequences for endocortical resorption and periosteal bone formation. J Bone Miner Res. 2012;27:374–89.
Watkins M, Grimston SK, Norris JY, et al. Osteoblast Connexin43 modulates skeletal architecture by regulating both arms of bone remodeling. Mol Biol Cell. 2011;22:1240–51.
Lloyd SA, Loiselle AE, Zhang Y, et al. Connexin 43 deficiency desensitizes bone to the effects of mechanical unloading through modulation of both arms of bone remodeling. Bone. 2013;57:76–83.
Grimston SK, Goldberg DB, Watkins M, et al. Connexin43 deficiency reduces the sensitivity of cortical bone to the effects of muscle paralysis. J Bone Miner Res. 2011;26:2151–60.
Acknowledgments
This research was supported by National Institutes of Health (R01-AR053643) and by a Biomedical Research Grant and a Developing Diverse Researchers with InVestigative Expertise (DRIVE) Grant from Indiana University School of Medicine to LIP and R01AR068132 and R01AG13087 to HJD.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
LI Plotkin, TL Speacht, and HJ Donahue all declare no conflicts of interest.
Human and Animal Rights and Informed Consent
All studies by the authors involving animal and/or human subjects were performed after approval by the appropriate institutional review boards. When required, written informed consent was obtained from all participants.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Osteocytes
Rights and permissions
About this article
Cite this article
Plotkin, L.I., Speacht, T.L. & Donahue, H.J. Cx43 and Mechanotransduction in Bone. Curr Osteoporos Rep 13, 67–72 (2015). https://doi.org/10.1007/s11914-015-0255-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-015-0255-2