Abstract
Dengue virus is the most widespread geographically of the arboviruses and a major public health threat in the tropics and subtropics. Scientific advances in recent years have provided new insights about the pathogenesis of more severe disease and novel approaches into the development of antiviral compounds and dengue vaccines. Phylogenetic studies show an association between specific subtypes (within serotypes) and severity of dengue. The lack of association between maternal antibodies and development of severe dengue in infants in a recent study has called for the rethinking or refinement of the current antibody-dependent enhancement theory of dengue hemorrhagic syndrome in infancy. Such studies should stimulate new directions of research into mechanisms responsible for the development of severe dengue. The life cycle of dengue virus readily shows that virus entry and replication can be targeted by small molecules. Advances in a mouse model (AG 129 mice) have made it easier to test such antiviral compounds. The efforts to find specific dengue inhibitors are intensifying and the tools to evaluate the efficacy of new drugs are now in place for rapid translation into trials in humans. Furthermore, several dengue vaccine candidates are in development, of which the chimeric dengue/yellow fever vaccine has now entered phase 3 trials. Until the availability of a licensed vaccine, disease surveillance and vector population control remain the mainstay of dengue prevention.
Similar content being viewed by others
Introduction
Dengue viruses are the most widespread geographically of the arboviruses and are found in tropical and subtropical areas where 2.5 billion to 3 billion people are at risk of infection [1]. Dengue viruses are transmitted by mosquitoes of the genus Aedes, subgenus Stegomyia (eg, Aedes aegypti and A. albopictus). A. aegypti is well established in much of the tropical and subtropical world [1]. As a peridomiciliary mosquito, A. aegypti is well adapted to urban life and typically breeds in clean, stagnant water (eg, rain water) and thrives on human proximity. Coupled with the epidemic potential of its blood meal–seeking behavior through feeding on multiple human hosts during a single blood meal, A. aegypti is the main epidemic vector of dengue virus [2]. A. albopictus, often regarded as a secondary vector although it has been implicated in several dengue outbreaks such as that in Hawaii in 2001 [3], is continuing its geographic expansion into tropical and temperate climates. However, the scale of epidemic mediated by A. albopictus has been much smaller relative to A. aegypti [1].
Dengue is also the most rapidly spreading mosquito-borne viral disease in the world. In the past 50 years, the incidence of dengue has increased 30-fold with increasing geographic expansion to new countries and, in the present decade, from urban to rural settings [4••]. An estimated 50 million to 100 million dengue infections occur annually. The World Health Organization (WHO) South-East Asian and Western Pacific Regions bear nearly 75% of the current global disease burden of dengue [4••]. Dengue inflicts a significant health, economic, and social burden on the populations of endemic areas. Globally, the number of disability-adjusted life years (DALYs) per million population lost to dengue is estimated to be between 528 and 621 per million population [4••]. With an annual average of 574,000 cases reported, the aggregate annual economic cost of dengue for eight study countries in the Americas and Asia is at least $587 million [5]. Preliminary adjustment for under-reporting could raise this total to $1.8 billion, and incorporating costs of dengue surveillance and vector control would raise the amount further. Dengue imposes substantial costs on both the health sector and the overall economy [5].
The presence of all four serotypes of dengue virus (or hyperendemicity) throughout the tropical and subtropical world since the 1970s also raises the likelihood of increased incidence of severe dengue disease [1]. Furthermore, hyperendemicity also increases the probability of secondary infection in the population, which is epidemiologically associated with severe dengue [6].
As a consequence of the expanding geographical distribution of both the virus and the mosquito vector, increased frequency of epidemics, and the emergence of dengue hemorrhagic fever in new areas, WHO has classified dengue as a major international public health concern. The reasons for the resurgence are complex, and are likely a combination of multiple factors, including population growth associated with rapid uncontrolled urbanization, increased movement of viruses in people among countries and regions via international travel, demographic changes, poor vector control, genetic changes in circulating or introduced viruses, and modulating climatic factors [7•].
This paper provides an update on recent trends in dengue epidemiology activity, new insights into dengue virology, and novel approaches for dengue control with a special focus on vaccine development.
Recent Trends in Epidemiology
The number of cases reported annually to WHO ranged from 0.4 to 1.3 million in the decade from 1996 to 2005 [4••]. The number of cases varies substantially from year to year, with epidemics occurring every 3 to 5 years. The underlying reason for this cyclical trend is poorly understood but is perhaps best explained by demographic, immunologic, and environmental changes combined [7•, 8–10]. Climatic influences, such as the El Nino southern oscillation (ENSO) and global warming, have been suggested as other factors contributing to the cyclical pattern of dengue activity [7•, 11]. Furthermore, dengue activity is seasonal within a year in most endemic countries related to seasonal rainfall and temperature changes [8, 12, 13]. The greatest burden of dengue in endemic countries is in children. However, there has been an increasing trend of adult infection in certain countries [14, 15•]. Most travelers with dengue have been adults [16].
South-East Asia and Western Pacific
The countries of the region have been divided into four distinct climatic zones with different dengue transmission potential. Epidemic dengue is a major public health problem in Indonesia, Malaysia, Myanmar, Sri Lanka, Thailand, Cambodia, Philippines, Laos, Viet Nam, and Timor-Leste, which are in the tropical monsoon and equatorial zone where A. aegypti is widespread in urban and less so in rural areas, with multiple virus serotypes circulating, and where dengue is a leading cause of hospitalization and death in children [7•]. Cyclical epidemics are increasing in frequency and in-country geographic expansion is occurring in Bangladesh, India, and Maldives—countries in the deciduous dry and wet climatic zone with multiple virus serotypes circulating [4••]. In 2004, Nepal and Bhutan reported the country’s first dengue outbreak [4••]. In 2005, WHO’s Global Outbreak Alert and Response Network (GOARN) reported an outbreak with a high case-fatality rate (3.55%) in Timor-Leste [4••]. Dengue has also spread throughout the Pacific Islands. Between 2001 and 2008, the three most affected Pacific island countries were French Polynesia, New Caledonia, and Cook Islands according to notifications to WHO [4••].
Middle East
Recent outbreaks (2005–2006) of dengue have been recorded in Pakistan, Saudi Arabia, Sudan, and Yemen [17]. In Pakistan, the first confirmed outbreak of dengue hemorrhagic fever (DHF) occurred in 1994 [4••]. Since then, the expansion of dengue infections with increasing frequency and severity has been reported from large cities in Pakistan as far north as the North-West Frontier Province.
Africa
Despite poor surveillance for dengue in Africa, it is clear that epidemic dengue fever caused by all four dengue serotypes has increased dramatically since 1980, with most epidemics occurring in eastern Africa, and to a smaller extent in western Africa [1, 4••, 18]. Likewise, travelers returning from Africa with dengue have been documented [12, 19], which, along with the observed seroprevalence rate in Africa [20], suggests that the true incidence of dengue in Africa is likely to be much larger than thought. Although dengue may not appear to be a major public health problem in Africa compared to the widespread incidence of malaria and HIV/AIDS, the increasing frequency and severity of dengue epidemics worldwide calls for a better understanding of the epidemiology of dengue infections with regard to the susceptibility of African populations to dengue and the interference between dengue and the other major communicable diseases of the continent.
Americas
From 2001 to 2007, more than 30 countries of the Americas notified a total of 4,332,731 cases of dengue, with 2007 being the worst year on record since 1985, with 918,495 cases of dengue in the Americas [4••].
North America, Europe, Australia, New Zealand
The majority of cases in Western industrialized countries are imported [16, 21]. Nevertheless, indigenous outbreaks of dengue in Hawaii have been reported [3], and sporadic outbreaks with local transmission occurred in Texas at the border with Mexico [22]. TropNetEurope reports hundreds of dengue cases imported to Europe every year [23]. Australia has the vector for dengue and indigenous outbreaks have occurred in Northern Australia (Queensland) as a result of importation [24, 25].
Recent reports indicate an increasing trend in both dengue incidence and geographic distribution. The reported cases probably represent only the tip of the iceberg of actual dengue virus activity and the true dengue incidence; hence, the disease burden is likely to be much larger than indicated here.
The Virus
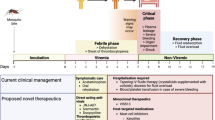
Dengue viruses (DENV) belong to the family of Flaviviridae. The spherical, enveloped virus contains three structural proteins and seven nonstructural proteins. The membrane precursor, prM, is believed to aid in the folding of the envelope (E) glycoprotein. The E glycoprotein mediates cell attachment and fusion and is also the major target of protective antibodies [26]. DENV has four serologically distinct serotypes (DENV-1, DENV-2, DENV-3, DENV-4). Infection with one serotype confers long-term immunity to that serotype, but not to the others [27]. DENV serotypes can be distinguished by virus-neutralizing antibodies if the patient has had only one flavivirus infection, but cross-reactive antibodies against the E protein and nonstructural proteins such as NS1 and NS3 are produced against the other serotypes after secondary infection, making serotype-specific diagnosis more difficult in secondary or multiple infections [28]. All four virus serotypes cause a spectrum of illness ranging from asymptomatic or mild febrile illness to classical dengue fever (DF) and to more severe disease manifested as DHF and dengue shock syndrome (DSS). The mechanisms for developing severe dengue are not fully understood. Severity of disease is likely to depend on various factors, including the strain and serotype of the infecting virus, age, genetic background of the patient and the degree of viremia [28, 29]. Many prospective, population-based cohort and clinical studies have established secondary infections with a heterotypic dengue virus serotype as a major risk factor for severe disease. Antibody-dependent enhancement (ADE) describes the boosting of virus replication in Fc-receptor bearing cells (especially monocytes and macrophages) by pre-existing, nonprotective levels of cross-reactive dengue antibodies [30]. It is postulated that ADE results in an amplified cascade of cytokines and complement activation causing endothelial dysfunction, platelet destruction, and consumption of coagulation factors, which result in plasma leakage and hemorrhagic manifestations [30]. These studies suggest that the immune response to a second heterotypic infection worsen rather than mitigate disease, although such events are not frequent.
However, not all dengue viruses can be enhanced, suggesting that the strain of virus is a major determining factor in virulence [31]. A recent prospective, nested, case-control study of primary DENV3 infections during infancy showed that infants exhibit a full range of disease severity after primary DENV infections [32••]. This study did not find an association between enhancing maternal antibodies and the development of DHF in infants [32••].
The evolution of dengue viruses has had a major impact on their virulence for humans and on the epidemiology of dengue disease around the world [33]. The RNA genome of the virus is susceptible to random mutations due to the lack of proofreading capacity of the RNA-dependent RNA polymerase, which could thus give rise to more virulent strains following increased levels of infection in a population [34]. Although antigenic and genetic differences in virus strains have become evident, the lack of animal models for severe dengue has made it difficult to study variation in virulence among dengue viruses. However, phylogenetic studies of many different dengue virus samples have led to the association between specific subtypes (within serotypes) and the presentation of more or less severe disease [33]. Currently, dengue viruses can be classified as being of epidemiologically low, medium, or high impact [33]. Subtypes within the American genotype of DENV-2 and genotype IV of DENV-3 for example are less virulent with a reduced ability to grow in cell cultures and mosquitoes compared to the Asian genotypes of DENV-2 and DENV-3. Analysis of envelope protein amino acid changes predicted to have accompanied endemic/epidemic emergence suggested a role for domain III in adaptation to new mosquito and/or human hosts [35]. Studies have suggested that specific viral structures may contribute to increased replication in human target cells and to increased transmission by the mosquito vector. In addition, mutations to the nonstructural genes could also play an important role in mediating epidemic transmission and increase disease severity [36]. Phylogenetic and epidemiological analyses suggest that the genotypes and subtypes with greater epidemic potential are now displacing those that have lower epidemiological impact [7•].
Although substantial progress has been made in understanding viral structure and viral interaction with host cells [37], much remains to be learned about the involvement of the immune system in disease pathogenesis. Although viral factors likely influence replication efficiency and hence epidemic transmission and virulence, host factors could also play a significant role in the degree of viremia. Given their compact genomes, dengue viruses probably require an extensive number of host factors; however, only a limited number of human, and an even smaller number of insect host factors, have been identified. A genome-wide RNA interference screen in Drosophila melanogaster cells using a well-established double-stranded RNA library was recently conducted [38••]. This screen identified 116 candidate dengue virus host factors (DVHFs). Although some were previously associated with flavivirus (eg, V-ATPases and α-glucosidases), most of the DVHFs were newly implicated in dengue virus propagation [38••]. The authors describe a notable conservation of required factors between dipteran and human hosts. This work suggests new approaches to control infection in the insect vector and the mammalian host.
Antiviral Drugs
Currently, no specific antiviral therapy is available for dengue, and treatment remains only supportive. The rationale for dengue antivirals arises from clinical studies that have noted that the quantity of virus circulating in the blood of patients who develop severe dengue (eg, DHF and DSS) is higher by around 1–2 logs compared with patients suffering from mild dengue disease [4••, 29]. Similar differences in viral load have been observed in animal models of ADE [39]. This observation suggests the potential benefit of antiviral therapy such as safe small-molecule compounds that can reduce viral load in the acute phase of the disease. An animal model—AG129 mice that are deficient for the interferon-α/β and interferon-γ receptors—is now being used for testing anti-dengue drugs [40••]. As a proof of concept, Schul et al. [40••] demonstrated that a clinical isolate of dengue virus can be used to infect AG129 mice, and antiviral compounds that block viral replication can clear viremia in a dose-dependent manner, even after delayed treatment, and suppress proinflammatory host responses. The life cycle of dengue virus readily shows that the steps involved in virus entry, membrane fusion, RNA genome replication, assembly, and ultimate release from the infected cell can be targeted by small molecules [41, 42•]. The entry of virus into host cells is mediated by the E protein and is the target for neutralizing antibodies that limit viral replication. Several small-molecular inhibitors have been shown to target viral entry [43, 44]. Currently, the most advanced targets are the NS2B/NS3 protease and the NS5 RNA-dependent RNA polymerase, and several in-silico and high-throughput screens yielding several lead compounds have been reported [45–47]. New targets—including E, NS3 helicase, and NS5 methyltransferase—are being explored [43, 48–50]. Further extensive characterization of the AG129 mouse model has demonstrated it to be one of the only models that permits infection by all four serotypes of dengue virus and allows antibody-mediated protection and enhancement of DENV infection [51]. In summary, the efforts to find specific dengue inhibitors are intensifying and the tools to evaluate the efficacy of new drugs are in place for rapid translation to human patients.
Dengue Vaccine Development
The development of a dengue vaccine has been a priority of WHO for three decades. The challenges in dengue vaccine development are summarized in Table 1. The main challenges have been the fear of ADE, the lack of a good animal model, and a reliable surrogate marker of immunity. Mice are often used as a small-animal model for the initial evaluation of the ability of candidate vaccines to induce a protective immune response. However, the results are not always predictive of what will happen in nonhuman primates and humans (ie, a candidate vaccine that protects mice may not be as effective in nonhuman primates and humans) [4••]. The second animal model is the nonhuman primate, and a variety of species have been used as models to evaluate candidate dengue vaccines. Unfortunately, although nonhuman primates demonstrate viremia, they do not present clinical disease and do not accurately predict attenuation in humans. Despite formidable challenges to developing tetravalent dengue vaccines, significant progress has been made in recent years and the pace towards clinical efficacy trials has accelerated substantially.
Four types of dengue vaccines are in development: live attenuated vaccines, chimeric live attenuated vaccines, inactivated or subunit vaccines, and nucleic acid–based vaccines.
Live Attenuated Vaccines
Live attenuated vaccines (LAVs) can induce durable humoral and cellular immune responses because they most closely mimic a natural infection. Several parameters are crucial for LAVs: the viruses must be sufficiently attenuated and must have low viremia, low reactogenicity, and high immunogenicity. The viremia level must also be low so that no transmission of the viruses by mosquitoes can occur.
The mutations that confer the attenuation phenotype should be stable and not revert to wild type or other virulent forms. Managing viral interference and balancing attenuation to produce acceptable tetravalent immunogenicity with minimal reactogenicity is another challenge [52]. Each of the four components of the vaccine must induce a balanced neutralizing antibody response.
Chimeric Live Attenuated Vaccines
The dengue vaccine with the most advanced development is the dengue/yellow fever chimeric vaccine. This vaccine uses the 17D yellow fever vaccine virus as its genetic backbone and replaces the yellow fever envelope (E) and prM genes with those from each of the four dengue viruses. This vaccine was shown to be attenuated, efficacious, safe, and highly unlikely to be transmitted by arthropod vectors [53]. ChimeriVax-Dengue (Sanofi Pasteur, Lyon, France) elicits antibodies only to dengue [54]. Current requirements for the development of live viral vaccines (including yellow fever 17D) produced from potentially neurotropic wild-type viruses, include tests for neurovirulence in nonhuman primates [55]. Neurovirulence in mice and monkeys was reduced compared to the yellow fever 17D vaccine virus [55]. These results suggest that rare neurotropic disease seen with the yellow fever vaccine [56] are less likely to occur with ChimeriVax-Dengue, although rare events can only be excluded with very large sample sizes. ChimeriVax-Dengue has also been tested in subjects with preexisting yellow fever immunity. The results showed that preexisting immunity to yellow fever virus did not interfere with immunization, and long-lasting and cross-neutralizing antibody responses to all four dengue serotypes were documented [57]. Currently, a large phase 2b trial on safety and immunogenicity is taking place in Singapore.
Another chimeric live vaccine uses the PDK-53 DEN-2 Mahidol vaccine candidate as a backbone. This virus was attenuated by passage in primary dog kidney cells. Similar to the concept of the yellow fever/dengue chimeric vaccine, the prM and E genes of DENV-2 are replaced with those of DENV-1, DENV-3, and DENV-4. Phase 1 safety trials are underway in the United States and Colombia. The three attenuating mutations of the PDK-53 DEN-2 are located outside the structural protein genes of and appear to be quite stable. The tetravalent vaccine produced by combining the four chimeric dengue viruses is protective when administered to mice and monkeys [58, 59]. Phase 1 trials in humans are planned.
A third chimeric live vaccine uses a DENV-4 attenuated by a ∆30 deletion of the 3′-untranslated region as the backbone. The prM and E proteins are replaced as above. Tetravalent formulations are being developed incorporating the into wild-type DENV-4, DENV-2, and DENV-3 viruses resulting in rDEN1/4∆30, rDEN2/4∆30, and rDEN3/4∆30 [60].
Inactivated Vaccines
Whole-virus inactivated vaccines have two major advantages over live attenuated virus vaccines. First, it is not possible for inactivated vaccines to revert to a more pathogenic phenotype; second, induction of a balanced antibody response is easier to attain [26]. The requirement for multiple dosing and the relatively shorter term of immunity make this approach less attractive compared to the others, although both limitations could be overcome through the development of novel adjuvants. However, such developments are likely to be expensive and would not meet the needs of dengue in endemic countries, which mostly have developing economies.
DNA Vaccines
DNA shuffling and screening technologies have been used to construct DNA expression vectors encoding the epitopes of the four dengue serotypes. Shuffled DNA vaccines have shown immunogenicity, and phase 1 monovalent dengue vaccine studies are currently underway [61]. DNA vaccines afford advantages in terms of ease of production, stability, and transport at room temperature, decreased likelihood of replication interference, and the possibility to vaccinate against multiple pathogens in a single vaccination [26]. However, DNA vaccines necessitate multiple dosing and experimental adjuvants, and they are unlikely to be economical.
Conclusions
Phylogenetic studies of many different dengue virus samples have led to the association between specific subtypes (within serotypes) and severity of dengue. Dengue genotypes and host factors play a more significant role in the development of more severe disease than previously thought. A recent study by Libraty et al. [32••] even suggests that rethinking or refinement of the current ADE pathogenesis model for infant DHF is needed, and this study should stimulate new directions of research into mechanisms responsible for the development of DHF. The life cycle of dengue virus readily shows that the steps involved in virus entry, RNA genome replication, and ultimate release from the infected cell can be targeted by small molecules. Advances in a mouse model (AG 129 mice) have made it easier to test antiviral compounds. The efforts to find specific dengue inhibitors are intensifying and the tools to evaluate the efficacy of new drugs are now in place for rapid translation to human patients.
Recent years have seen a rapid acceleration of the development of dengue vaccines. Various candidates are in development, of which the chimeric dengue/yellow fever vaccine has now entered phase 3 trials. Until the availability of a licensed vaccine, disease surveillance and vector population control remain the mainstay of dengue prevention.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Gubler DJ: The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res 2002, 33:330–342.
Halstead SB: Dengue virus-mosquito interactions. Annu Rev Entomol 2008, 53:273–291.
Effler PV, Pang L, Kitsutani P, et al.: Dengue fever, Hawaii, 2001–2002. Emerg Infect Dis 2005, 11:742–749.
•• Dengue: guidelines for diagnosis, treatment, prevention and control. Geneva: World Health Organization; 2009. These revised and updated WHO guidelines provide updates on epidemiology, the new classification of clinical dengue, and new developments in vaccines and antiviral compounds.
Suaya JA, Shepard DS, Siqueira JB, et al.: Cost of dengue cases in eight countries in the Americas and Asia: a prospective study. Am J Trop Med Hyg 2009, 80:846–855.
Halstead SB, Udomsakdi S, Simasthien P, et al.: Observations related to pathogenesis of dengue hemorrhagic fever. I. Experience with classification of dengue viruses. Yale J Biol Med 1970, 42:261–275.
• Wilder-Smith A, Gubler DJ: Geographic expansion of dengue: the impact of international travel. Med Clin North Am 2008, 92:1377–1390. This review article provides a critical discussion about the reasons for the geographic expansion of dengue in recent years.
Ooi E, Gubler DJ: Global spread of epidemic dengue: the influence of environmental change. Future Virol 2009, 4:571–580.
Cheng S, Kalkstein LS, Focks DA, et al.: New procedures to estimate water temperatures and water depths for application in climate-dengue modeling. J Med Entomol 1998, 35:646–652.
Egger JR, Ooi EE, Kelly DW, et al.: Reconstructing historical changes in the force of infection of dengue fever in Singapore: implications for surveillance and control. Bull World Health Organ 2008, 86:187–196.
Hales S, Weinstein P, Souares Y, et al.: El Nino and the dynamics of vectorborne disease transmission. Environ Health Perspect 1999, 107:99–102.
Schwartz E, Weld LH, Wilder-Smith A, et al.: Seasonality, annual trends, and characteristics of dengue among ill returned travelers, 1997–2006. Emerg Infect Dis 2008, 14:1081–1088.
Bartley LM, Donnelly CA, Garnett GP: The seasonal pattern of dengue in endemic areas: mathematical models of mechanisms. Trans R Soc Trop Med Hyg 2002, 96:387–397.
Ooi EE, Goh KT, Gubler DJ: Dengue prevention and 35 years of vector control in Singapore. Emerg Infect Dis 2006, 12:887–893.
• Cummings DA, Iamsirithaworn S, Lessler JT, et al.: The impact of the demographic transition on dengue in Thailand: insights from a statistical analysis and mathematical modeling. PLoS Med 2009, 6:e1000139. This article highlights the demographic changes in the presentation of dengue.
Wilder-Smith A, Schwartz E: Dengue in travelers. N Engl J Med 2005, 353:924–932.
WHO/EMRO World Health Organization, Regional Office for the Eastern Mediterranean, Division of Communicable Disease Control, Newsletter. 2005, 6:7–8.
Leroy EM, Nkoghe D, Ollomo B, et al.: Concurrent chikungunya and dengue virus infections during simultaneous outbreaks, Gabon, 2007. Emerg Infect Dis 2009, 15:591–593.
Ninove L, Parola P, Baronti C, et al.: Dengue virus type 3 infection in traveler returning from west Africa. Emerg Infect Dis 2009, 15:1871–1872.
Kuniholm MH, Wolfe ND, Huang CY, et al.: Seroprevalence and distribution of Flaviviridae, Togaviridae, and Bunyaviridae arboviral infections in rural Cameroonian adults. Am J Trop Med Hyg 2006, 74:1078–1083.
Freedman DO, Weld LH, Kozarsky PE, et al.: Spectrum of disease and relation to place of exposure among ill returned travelers. N Engl J Med 2006, 354:119–130.
Ramos MM, Mohammed H, Zielinski-Gutierrez E, et al.: Epidemic dengue and dengue hemorrhagic fever at the Texas-Mexico Border: results of a household-based seroepidemiologic survey, December 2005. Am J Trop Med Hyg 2008, 78:364–369.
Jelinek T, Mühlberger N, Harms G, et al.: Epidemiology and clinical features of imported dengue fever in Europe: sentinel surveillance data from TropNetEurop. Clin Infect Dis 2002, 35:1047–1052.
Kitchener S, Leggat PA, Brennan L, McCall B: Importation of dengue by soldiers returning from East Timor to north Queensland, Australia. J Travel Med 2002, 9:180–183.
Beebe NW, Cooper RD, Mottram P, et al.: Australia’s dengue risk driven by human adaptation to climate change. PLoS Negl Trop Dis 2009, 3:e429.
Whitehead SS, Blaney JE, Durbin AP, et al.: Prospects for a dengue virus vaccine. Nat Rev Microbiol 2007, 5:518–528.
Gubler DJ: Epidemic dengue/dengue hemorrhagic fever as a public health, social and economic problem in the 21st century. Trends Microbiol 2002, 10:100–103.
Guzman MG, Kouri G: Dengue: an update. Lancet Infect Dis 2002, 2:33–42.
Vaughn DW, Green S, Kalayanarooj S, et al.: Dengue viremia titer, antibody response pattern, and virus serotype correlate with disease severity. J Infect Dis 2000, 181:2–9.
Halstead SB: In vivo enhancement of dengue virus infection in rhesus monkeys by passively transferred antibody. J Infect Dis 1979, 140:527–533.
Gubler DJ: Dengue and dengue hemorrhagic fever. Clin Microbiol Rev 1998, 11:480–496.
•• Libraty DH, Acosta LP, Tallo V, et al.: A prospective nested case-control study of Dengue in infants: rethinking and refining the antibody-dependent enhancement dengue hemorrhagic fever model. PLoS Med 2009, 6:e1000171. This study suggests that the ADE theory for DHF in infancy needs to be rethought.
Rico-Hesse R: Microevolution and virulence of dengue viruses. Adv Virus Res 2003, 59:315–341.
Gubler DJ, Trent DW: Emergence of epidemic dengue/dengue hemorrhagic fever as a public health problem in the Americas. Infect Agents Dis 1993, 2:383–393.
Wang E, Ni H, Xu R, et al.: Evolutionary relationships of endemic/epidemic and sylvatic dengue viruses. J Virol 2000, 74:3227–3234.
Bennett SN, Holmes EC, Chirivella M, et al.: Selection-driven evolution of emergent dengue virus. Mol Biol Evol 2003, 20:1650–1658.
Kuhn RJ, Zhang W, Rossmann MG, et al.: Structure of dengue virus: implications for flavivirus organization, maturation, and fusion. Cell 2002, 108:717–725.
•• Sessions OM, Barrows NJ, Souza-Neto JA, et al.: Discovery of insect and human dengue virus host factors. Nature 2009, 458:1047–1050. This study identified 116 candidate dengue virus host factors; indicating notable conservation of required factors between dipteran and human hosts. This work suggests new approaches to control infection in the insect vector and the mammalian host.
Goncalvez AP, Engle RE, St Claire M, et al.: Monoclonal antibody-mediated enhancement of dengue virus infection in vitro and in vivo and strategies for prevention. Proc Natl Acad Sci U S A 2007, 104:9422–9427.
•• Schul W, Liu W, Xu HY, et al.: A dengue fever viremia model in mice shows reduction in viral replication and suppression of the inflammatory response after treatment with antiviral drugs. J Infect Dis 2007, 195:665–674. This article highlights the advantages of a new mouse model.
Keller TH, Chen YL, Knox JE, et al.: Finding new medicines for flaviviral targets. Novartis Found Symp 2006, 277:102–114; discussion 14–9, 251–253.
• Noble CG, Chen YL, Dong H, et al.: Strategies for development of dengue virus inhibitors. Antiviral Res 2010, 85:450–462. This article provides a comprehensive update on recent developments in antiviral compounds for dengue virus inhibition.
Wang QY, Patel SJ, Vangrevelinghe E, et al.: A small-molecule dengue virus entry inhibitor. Antimicrob Agents Chemother 2009, 53:1823–1831.
Li Z, Khaliq M, Zhou Z, et al.: Design, synthesis, and biological evaluation of antiviral agents targeting flavivirus envelope proteins. J Med Chem 2008, 51:4660–4671.
Yin Z, Chen YL, Kondreddi RR, et al.: N-sulfonylanthranilic acid derivatives as allosteric inhibitors of dengue viral RNA-dependent RNA polymerase. J Med Chem 2009, 52:7934–7937.
Yin Z, Chen YL, Schul W, et al.: An adenosine nucleoside inhibitor of dengue virus. Proc Natl Acad Sci U S A 2009;106:20435–20439.
Johnston PA, Phillips J, Shun TY, et al.: HTS identifies novel and specific uncompetitive inhibitors of the two-component NS2B-NS3 proteinase of West Nile virus. Assay Drug Dev Technol 2007, 5:737–750.
Luzhkov VB, Selisko B, Nordqvist A, et al.: Virtual screening and bioassay study of novel inhibitors for dengue virus mRNA cap (nucleoside-2′O)-methyltransferase. Bioorg Med Chem 2007, 15:7795–7802.
Li J, Lim SP, Beer D, et al.: Functional profiling of recombinant NS3 proteases from all four serotypes of dengue virus using tetrapeptide and octapeptide substrate libraries. J Biol Chem 2005, 280:28766–28774.
Xu T, Sampath A, Chao A, et al.: Towards the design of flavivirus helicase/NTPase inhibitors: crystallographic and mutagenesis studies of the dengue virus NS3 helicase catalytic domain. Novartis Found Symp 2006, 277:87–97; discussion 101, 251–253.
Williams KL, Zompi S, Beatty PR, et al.: A mouse model for studying dengue virus pathogenesis and immune response. Ann N Y Acad Sci 2009, 1171(Suppl 1):E12–E23.
Kitchener S, Nissen M, Nasveld P, et al.: Immunogenicity and safety of two live-attenuated tetravalent dengue vaccine formulations in healthy Australian adults. Vaccine 2006, 24:1238–1241.
McGee CE, Lewis MG, Claire MS, et al.: Recombinant chimeric virus with wild-type dengue 4 virus premembrane and envelope and virulent yellow fever virus asibi backbone sequences is dramatically attenuated in nonhuman primates. J Infect Dis 2008, 197:693–697.
Monath TP: Dengue and yellow fever--challenges for the development and use of vaccines. N Engl J Med 2007, 357:2222–2225.
Monath TP, Myers GA, Beck RA, et al.: Safety testing for neurovirulence of novel live, attenuated flavivirus vaccines: infant mice provide an accurate surrogate for the test in monkeys. Biologicals 2005, 33:131–144.
Khromava AY, Eidex RB, Weld LH, et al.: Yellow fever vaccine: an updated assessment of advanced age as a risk factor for serious adverse events. Vaccine 2005, 23:3256–3263.
Guirakhoo F, Kitchener S, Morrison D, et al.: Live attenuated chimeric yellow fever dengue type 2 (ChimeriVax-DEN2) vaccine: phase I clinical trial for safety and immunogenicity: effect of yellow fever pre-immunity in induction of cross neutralizing antibody responses to all 4 dengue serotypes. Hum Vaccin 2006, 2:60–67.
Huang CY, Butrapet S, Tsuchiya KR, et al.: Dengue 2 PDK-53 virus as a chimeric carrier for tetravalent dengue vaccine development. J Virol 2003, 77:11436–11447.
Rabablert J, Wasi C, Kinney R, et al.: Attenuating characteristics of DEN-2 PDK53 in flavivirus-naive peripheral blood mononuclear cells. Vaccine 2007, 25:3896–3905.
Halstead SV, Vaughn DW: Dengue vaccines. In Vaccines. Edited by Plotkin SA, Orenstein WA, Offit PA. Oxford: Elsevier; 2008:1155–1161.
Hombach J: Vaccines against dengue: a review of current candidate vaccines at advanced development stages. Rev Panam Salud Publica 2007, 21:254–260.
Disclosure
Dr. Wilder-Smith is the principal investigator of a Sanofi Pasteur dengue vaccine trial; received speaker honoraria and reimbursements for travel to conferences by GlaxoSmithKline, Novartis, and Sanofi Pasteur; and has served on the Novartis advisory board for travel vaccines. Dr. Gubler has received honoraria from Novartis Institute of Tropical Diseases, Invivogen, and Hawaii Biotech. No other potential conflict of interest relevant to this article was reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wilder-Smith, A., Ooi, EE., Vasudevan, S.G. et al. Update on Dengue: Epidemiology, Virus Evolution, Antiviral Drugs, and Vaccine Development. Curr Infect Dis Rep 12, 157–164 (2010). https://doi.org/10.1007/s11908-010-0102-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11908-010-0102-7