Abstract
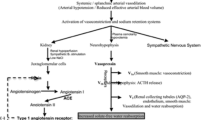
Hyponatremia is a common complication of patients with advanced cirrhosis that is associated with increased morbidity and mortality. Patients with cirrhosis may develop two types of hyponatremia: hypovolemic or hypervolemic hyponatremia. Hypervolemic hyponatremia is the most frequent type of hyponatremia that develops in patients with advanced liver disease and is the consequence of impairment in the renal capacity to eliminate solute-free water. The pathogenesis of these increased solute-free water retention involves several factors, but the most important one is a non-osmotic hypersecretion of vasopressin. The treatment of choice for hypervolemic hyponatremia is fluid restriction. Vaptans, drugs that are selective antagonists of vasopressin V2 receptors, emerged as the first pharmacological treatment of hypervolemic hyponatremia in cirrhosis with promising results. However, satavaptan was withdrawn from development for safety reasons and tolvaptan is not recommended in patients with liver disease. Therefore, currently there is no effective and safe pharmacological approach available for the management of hypervolemic hyponatremia in cirrhosis.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Angeli P, Wong F, Watson H, et al. Hyponatremia in cirrhosis: results of a patient population survey. Hepatology. 2006;44:1535–42.
Adrogue HJ, Madias NE. Hyponatremia. N Engl J Med. 2000;342:1581–9.
Ginès P, Berl T, Bernardi M, et al. Hyponatremia in cirrhosis: from pathogenesis to treatment. Hepatology. 1998;28:851–64.
Ginès P, Guevara M. Hyponatremia in cirrhosis: pathogenesis, clinical significance and management. Hepatology. 2008;48:1002–10.
Ginès P, Cardenas A, Schrier RW. Liver disease and the kidney. In: Schrier RW, editor. Diseases of the kidney and urinary tract, vol. 3. 8th ed. Philadelphia: Lippincott Williams & Wilkins; 2006. p. 2179–205.
Biggins S, Rodriguez HJ, Bachetti P, et al. Serum predicts mortality in patients listed for liver transplantation. Hepatology. 2005;41:32–9.
Londoño MC, Guevara M, Rimola A, et al. MELD score and serum sodium in the predictions of survival of patients with cirrhosis awaiting liver transplantation. Gut. 2007;56:1283–90.
Kim WR, Biggins SW, Kremers WK, et al. Hyponatremia and mortality among patients on the liver-transplant waiting list. N Engl J Med. 2008;359:1018–26.
Ishikawa S, Schrier RW. Pathogenesis of hyponatremia: the role of arginine vasopressin. In: Ginès P, Arroyo V, Rodes J, Schrier R, editors. Ascites and renal dysfunction in liver disease. 2nd ed. Oxford: Blackwell Publishing; 2005. p. 305–14.
Kwon TH, Hager H, Nejsum LN, et al. Physiology and pathophysiology of renal aquaporins. Semin Nephrol. 2001;21:231–8.
Nielsen S, Frokiaer J, Marples D, et al. Aquaporins in the kidney: from molecules to medicine. Physiol Rev. 2002;82:205–44.
Guevara M, Baccaro ME, Torre A, et al. Hyponatremia is a risk factor of hepatic encephalopathy in patients with cirrhosis: a prospective study with time-dependent analysis. Am J Gastroenterol. 2009;104:1382–9.
Riggio O, Angeloni S, Salvatori FM, et al. Incidence, natural history, and risk factors of hepatic encephalopathy after transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene-covered stent grafts. Am J Gastroenterol. 2008;103:2738–46.
Guevara M, Baccarro ME, Rios J, et al. Risk factors for hepatic encephalopathy in patients with cirrhosis and refractory Ascites: relevance of serum sodium concentration. Liver Int. 2010;30:1137–42.
Haussinger D, Schliess F. Pathogenetic mechanisms of hepatic encephalopathy. Gut. 2008;57:1156–65.
Córdoba J, García-Martinez R, Simón-Talero M. Hyponatremic and hepatic encephalopathies: similarities, differences and coexistence. Metab Brain Dis. 2010;25:73–80.
Pereira G, Guevara M, Fagundes C, et al. Renal failure and hiponatremia in patients with cirrosis and skin and soft tissue infection. A retrospective study. J Hepatol. 2012;56:1040–6.
Follo A, Llovet JM, Navasa M, et al. Renal impairment after spontaneous bacterial peritonitis in cirrhosis: course, predictive factors and prognosis. Hepatology. 1994;20:1495–501.
Ginès A, Escorsell A, Ginès P, et al. Incidence, predictive factors, and prognosis of hepatorenal syndrome in cirrhosis. Gastroenterology. 1993;105:229–36.
Solà E, Watson H, Graupera I, et al. Factors related to quality of life in patients with cirrhosis and Ascites: relevance of serum sodium concentration and leg edema. J Hepatol. 2012;57:1199–206. This study investigated the effects of serum sodium concentration in the impairment of health-related quality of life in a large population of patients with cirrhosis and ascites. Interestingly, results of this study showed that serum sodium concentration was an independent predictive factor of impairment of quality of life in patients with decompensated cirrhosis.
Verbalis JG, Goldsmith SR, Greenberg A, et al. Hyponatremia treatment guidelines 2007: expert panel recommendations. Am J Med. 2007;120(11 Suppl 1):S1–S21.
Ginès P, Angeli P, Lenz K, et al. EASL clinical practice guidelines on the management of Ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53:397–417.
Jalan R, Mookerjee R, Cheshire L, Williams R, Davies N. Albumin infusion for severe hyponatremia in patients with refractory Ascites: a randomized clinical trial (abstract). J Hepatol. 2007;46:232A.
McCormick PA, Mistry P, Kaye G, Burroughs AK, McIntyre N. Intravenous albumin infusion is an effective therapy for hyponatremia in cirrhotic patients with Ascites. Gut. 1990;31:204–7.
Decaux G, Soupart A, Vassart G. Non-peptide arginine-vasopressin antagonists: the vaptans. Lancet. 2008;371:1624–32.
Wong F, Blei AT, Blendis LM, Thuluvath PJ. A vasopressin receptor antagonist (VPA-985) improves serum sodium concentration in patients with hyponatremia: a multicenter, randomized, placebo-controlled trial. Hepatology. 2003;37:182–91.
Gerbes AL, Gulberg V, Ginès P, et al. VPA study group. Therapy of hyponatremia in cirrhosis with a vasopressin receptor antagonist: a randomized double-blind multicenter trial. Gastroenterology. 2003;124:933–9.
Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med. 2006;355:2099–112.
Ginès P, Wong F, Watson H, et al. Effects of satavaptan, a selective vasopressin V2 receptor antagonist, on Ascites and serum sodium in cirrhosis with hyponatremia: a randomized trial. Hepatology. 2008;48:204–13.
O’Leary JG, Davis GL. Conivaptan increases serum sodium in hyponatremic patients with end-stage liver disease. Liver Transpl. 2009;15:1325–9.
Cárdenas A, Ginès P, Marotta P, et al. The safety and efficacy of tolvaptan, an oral vasopressin antagonist in the treatment of hyponatremia in cirrhosis. J Hepatol. 2012;56:571–8. This is a subanalysis of patients with cirrhosis included in the Study of Ascending Levels of Tolvaptan (SALT) trial that investigated the efficacy and safety of tolvaptan in patients with hypervolemic hyponatremia. The results of this study showed that after one month of treatment, 33% of patients receiving tolvaptan normalized serum sodium concentration compared to 19% of patients in the placebo group. Moreover, results from this subanalysis showed that the correction of hyponatremia with tolvaptan improved quality of life.
Berl T, Quittnat-Pelletier F, Verbalis JG, Schrier RW, Bichet DG, Ouyang J, et al. Oral tolvaptan is safe and effective in chronic hyponatremia. J Am Soc Nephrol. 2010;21:705–12.
Ahluwalia V, Heuman DM, Feldman G, et al. Correction of hyponatremia improves cognition, quality of life, and brain oedema in cirrhosis. J Hepatol. 2015;62:75–82. This is another recent study that investigated the effects of tolvaptan not only on serum sodium concentration but also on quality of life and brain magnetic resonance imaging in 14 patients with cirrhosis and hypervolemic hyponatremia. The study showed that serum sodium concentration significantly increased with a mean final value of 133mEq/L. Moreover, the study showed that the correction of hyponatremia was associated with cognitive and health-related quality of life improvement.
Acknowledgments
Some of the work discussed in this review has been funded by the FIS PI12/00330 integrated in the Plan Nacional I+D+I and co-funded by ISCIII-Subdirección General de Evaluación and European Regional Development Fund (ERDF). CIBEREHD is funded by the Instituto de Salud Carlos III. Agència de Gestió d’Ajuts Universitaris i de Recerca (AGAUR) 2014 SGR 708. Pere Ginès is recipient of an ICREA Academia Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
Elsa Solà, Patricia Huelin, and Pere Ginès declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
With regard to the authors’ research cited in this paper, all procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
This article is part of the Topical Collection on Management of the Cirrhotic Patient
Rights and permissions
About this article
Cite this article
Solà, E., Huelin, P. & Ginès, P. The Management of Hyponatremia in Cirrhosis: Should it Be Pharmacologic?. Curr Hepatology Rep 15, 53–59 (2016). https://doi.org/10.1007/s11901-016-0293-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11901-016-0293-y