Abstract
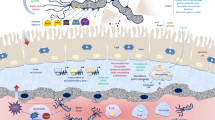
Colorectal cancer is one of the commonest forms of cancer worldwide. Although the molecular pathogenesis of colorectal cancer shares many characteristics with that of other cancers, the tissue environment is unique in that the intestinal mucosal surface is continuously exposed to a vast community of microorganisms. It is increasingly recognized that the intestinal microbiota is a critical component of the tumor environment that contributes to the development of colorectal cancer, and certain members of the commensal microbiota have been identified as critical elements in intestinal carcinogenesis. As sensors of the presence of microbes at mucosal surfaces, pattern-recognition receptors of the innate immune system are equally involved in this process. This review summarizes our current knowledge of the role of the microbiota in colorectal cancer development and provides an overview of the mechanisms involved in the cross talk between intestinal microbial colonization and tumorigenesis.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Kuper H, Adami HO, Trichopoulos D. Infections as a major preventable cause of human cancer. J Intern Med. 2000;248:171–83.
Hu WY, Bushman FD, Siva AC. RNA interference against retroviruses. Virus Res. 2004;102:59–64.
Russell W. An address on a characteristic organism of cancer. Br Med J. 1890;2:1356–60.
Wuerthele-Caspe V et al. Cultural properties and pathogenicity of certain microorganisms obtained from various proliferative and neoplastic diseases. Am J Med Sci. 1950;220:638–46.
Livingston VW, Alexander-Jackson E. An experimental biologic approach to the treatment of neoplastic disease; determination of actinomycin in urine and cultures as an aid to diagnosis and prognosis. J Am Med Womens Assoc. 1965;20:858–66.
Unproven methods of cancer management. Livingston-Wheeler therapy. CA Cancer J Clin. 1991;41:A7–12.
Polk DB, Peek Jr RM. Helicobacter pylori: gastric cancer and beyond. Nat Rev Cancer. 2010;10:403–14.
Samaras V, Rafailidis PI, Mourtzoukou EG, Peppas G, Falagas ME. Chronic bacterial and parasitic infections and cancer: a review. J Infect Dev Ctries. 2010;4:267–81.
Berg RD. The indigenous gastrointestinal microflora. Trends Microbiol. 1996;4:430–5.
Gordon JI. Honor thy gut symbionts redux. Science. 2012;336:1251–3.
Sonnenburg JL et al. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science. 2005;307:1955–9.
Candela M et al. Interaction of probiotic Lactobacillus and Bifidobacterium strains with human intestinal epithelial cells: adhesion properties, competition against enteropathogens and modulation of IL-8 production. Int J Food Microbiol. 2008;125:286–92.
Fukuda S et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature. 2011;469:543–7.
Hosokawa T, Kikuchi Y, Nikoh N, Shimada M, Fukatsu T. Strict host-symbiont cospeciation and reductive genome evolution in insect gut bacteria. PLoS Biol. 2006;4:e337.
Hooper LV, Littman DR, Macpherson AJ. Interactions between the microbiota and the immune system. Science. 2012;336:1268–73.
Yatsunenko T et al. Human gut microbiome viewed across age and geography. Nature. 2012;486:222–7.
Dominguez-Bello MG et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci U S A. 2010;107:11971–5.
Eckburg PB et al. Diversity of the human intestinal microbial flora. Science. 2005;308:1635–8.
Spor A, Koren O, Ley R. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat Rev Microbiol. 2011;9:279–90.
Lupp C et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe. 2007;2:204.
Tenesa A, Dunlop MG. New insights into the aetiology of colorectal cancer from genome-wide association studies. Nat Rev Genet. 2009;10:353–8.
Peek Jr RM, Blaser MJ. Helicobacter pylori and gastrointestinal tract adenocarcinomas. Nat Rev Cancer. 2002;2:28–37.
Gold JS, Bayar S, Salem RR. Association of Streptococcus bovis bacteremia with colonic neoplasia and extracolonic malignancy. Arch Surg. 2004;139:760–5.
Nakamura J et al. Comparison of four microbial enzymes in Clostridia and Bacteroides isolated from human feces. Microbiol Immunol. 2002;46:487–90.
Ellmerich S, Djouder N, Scholler M, Klein JP. Production of cytokines by monocytes, epithelial and endothelial cells activated by Streptococcus bovis. Cytokine. 2000;12:26–31.
Castellarin M et al. Fusobacterium nucleatum infection is prevalent in human colorectal carcinoma. Genome Res. 2012;22:299–306.
Arthur JC et al. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science. 2012;338:120–3. This article demonstrates the impact of intestinal inflammation on the outgrowth of a bacterial strain that exerts genotoxic effects on the host.
Sobhani I et al. Microbial dysbiosis in colorectal cancer (CRC) patients. PLoS One. 2011;6:e16393.
Chiba T, Marusawa H, Ushijima T. Inflammation-associated cancer development in digestive organs: mechanisms and roles for genetic and epigenetic modulation. Gastroenterology. 2012;143:550–63.
Scanlan PD et al. Culture-independent analysis of the gut microbiota in colorectal cancer and polyposis. Environ Microbiol. 2008;10:789–98.
Wu N et al. Dysbiosis signature of fecal microbiota in colorectal cancer patients. Microb Ecol. 2013;66:462–70.
Wang T et al. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. ISME J. 2012;6:320–9.
Pryde SE, Duncan SH, Hold GL, Stewart CS, Flint HJ. The microbiology of butyrate formation in the human colon. FEMS Microbiol Lett. 2002;217:133–9.
Rose DJ, DeMeo MT, Keshavarzian A, Hamaker BR. Influence of dietary fiber on inflammatory bowel disease and colon cancer: importance of fermentation pattern. Nutr Rev. 2007;65:51–62.
Martin HM et al. Enhanced Escherichia coli adherence and invasion in Crohn’s disease and colon cancer. Gastroenterology. 2004;127:80–93.
Maddocks OD, Short AJ, Donnenberg MS, Bader S, Harrison DJ. Attaching and effacing Escherichia coli downregulate DNA mismatch repair protein in vitro and are associated with colorectal adenocarcinomas in humans. PLoS One. 2009;4:e5517.
Reddy BS, Mastromarino A, Wynder EL. Further leads on metabolic epidemiology of large bowel cancer. Cancer Res. 1975;35:3403–6.
Kim SC et al. Variable phenotypes of enterocolitis in interleukin 10-deficient mice monoassociated with two different commensal bacteria. Gastroenterology. 2005;128:891–906.
Fasano A. Cellular microbiology: can we learn cell physiology from microorganisms? Am J Physiol. 1999;276:C765–76.
Rhee KJ et al. Induction of persistent colitis by a human commensal, enterotoxigenic Bacteroides fragilis, in wild-type C57BL/6 mice. Infect Immun. 2009;77:1708–18.
Wu S et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat Med. 2009;15:1016–22.
Jess T, Rungoe C, Peyrin-Biroulet L. Risk of colorectal cancer in patients with ulcerative colitis: a meta-analysis of population-based cohort studies. Clin Gastroenterol Hepatol. 2012;10:639–45.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–35.
Ullman TA, Itzkowitz SH. Intestinal inflammation and cancer. Gastroenterology. 2011;140:1807–16.
Hu B et al. Microbiota-induced activation of epithelial IL-6 signaling links inflammasome-driven inflammation with transmissible cancer. Proc Natl Acad Sci U S A. 2013;110:9862–7. This report demonstrates that susceptibility to CRC development is transmissible through the transfer of dysbiotic microbiota.
Qin J et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464:59–65.
Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–45.
Darfeuille-Michaud A et al. Presence of adherent Escherichia coli strains in ileal mucosa of patients with Crohn’s disease. Gastroenterology. 1998;115:1405–13.
Swidsinski A et al. Association between intraepithelial Escherichia coli and colorectal cancer. Gastroenterology. 1998;115:281–6.
Cuevas-Ramos G et al. Escherichia coli induces DNA damage in vivo and triggers genomic instability in mammalian cells. Proc Natl Acad Sci U S A. 2010;107:11537–42.
Nougayrede JP et al. Escherichia coli induces DNA double-strand breaks in eukaryotic cells. Science. 2006;313:848–51.
Putze J et al. Genetic structure and distribution of the colibactin genomic island among members of the family Enterobacteriaceae. Infect Immun. 2009;77:4696–703.
Geijtenbeek TB, van Vliet SJ, Engering A, ’t Hart BA, van Kooyk Y. Self- and nonself-recognition by C-type lectins on dendritic cells. Annu Rev Immunol. 2004;22:33–54.
Medzhitov R, Janeway Jr CA. Decoding the patterns of self and nonself by the innate immune system. Science. 2002;296:298–300.
Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell. 2002;10:417–26.
Martinon F, Mayor A, Tschopp J. The inflammasomes: guardians of the body. Annu Rev Immunol. 2009;27:229–65.
Elinav E, Strowig T, Henao-Mejia J, Flavell RA. Regulation of the antimicrobial response by NLR proteins. Immunity. 2011;34:665–79.
Chen GY, Shaw MH, Redondo G, Nunez G. The innate immune receptor Nod1 protects the intestine from inflammation-induced tumorigenesis. Cancer Res. 2008;68:10060–7.
Rakoff-Nahoum S, Medzhitov R. Regulation of spontaneous intestinal tumorigenesis through the adaptor protein MyD88. Science. 2007;317:124–7.
Salcedo R et al. MyD88-mediated signaling prevents development of adenocarcinomas of the colon: role of interleukin 18. J Exp Med. 2010;207:1625–36.
Fukata M et al. Toll-like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology. 2007;133:1869–81.
Lowe EL et al. Toll-like receptor 2 signaling protects mice from tumor development in a mouse model of colitis-induced cancer. PLoS One. 2010;5:e13027.
Dupaul-Chicoine J et al. Control of intestinal homeostasis, colitis, and colitis-associated colorectal cancer by the inflammatory caspases. Immunity. 2010;32:367–78.
Elinav E et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell. 2011;145:745–57. This article shows the involvement of the NLRP6 inflammasome in the prevention of dysbiosis development. Absence of NLRP6 leads to the outgrowth of colitogenic microbiota.
Allen IC et al. The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. J Exp Med. 2010;207:1045–56.
Zaki MH et al. The NLRP3 inflammasome protects against loss of epithelial integrity and mortality during experimental colitis. Immunity. 2010;32:379–91.
Bauer C et al. Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome. Gut. 2010;59:1192–9.
Hu B et al. Inflammation-induced tumorigenesis in the colon is regulated by caspase-1 and NLRC4. Proc Natl Acad Sci U S A. 2010;107:21635–40.
Chen GY, Liu M, Wang F, Bertin J, Nunez G. A functional role for Nlrp6 in intestinal inflammation and tumorigenesis. J Immunol. 2011;186:7187–94.
Allen IC et al. NLRP12 suppresses colon inflammation and tumorigenesis through the negative regulation of noncanonical NF-κB signaling. Immunity. 2012;36:742–54.
Zaki MH et al. The NOD-like receptor NLRP12 attenuates colon inflammation and tumorigenesis. Cancer Cell. 2011;20:649–60.
Zhu Y, Michelle Luo T, Jobin C, Young HA. Gut microbiota and probiotics in colon tumorigenesis. Cancer Lett. 2011;309:119–27.
Pala V et al. Yogurt consumption and risk of colorectal cancer in the Italian European prospective investigation into cancer and nutrition cohort. Int J Cancer. 2011;129:2712–9.
Goncharova GI, Dorofeichuk VG, Smolianskaia AZ, Sokolova K. [Microbial ecology of the intestines in health and in pathology]. Antibiot Khimioter. 1989;34:462–6.
Arumugam M et al. Enterotypes of the human gut microbiome. Nature. 2011;473:174–80.
Corthesy B, Gaskins HR, Mercenier A. Cross-talk between probiotic bacteria and the host immune system. J Nutr. 2007;137:781S–90.
Orlando A, Messa C, Linsalata M, Cavallini A, Russo F. Effects of Lactobacillus rhamnosus GG on proliferation and polyamine metabolism in HGC-27 human gastric and DLD-1 colonic cancer cell lines. Immunopharmacol Immunotoxicol. 2009;31:108–16.
Lee NK, Park JS, Park E, Paik HD. Adherence and anticarcinogenic effects of Bacillus polyfermenticus SCD in the large intestine. Lett Appl Microbiol. 2007;44:274–8.
Kim Y et al. Inhibition of proliferation in colon cancer cell lines and harmful enzyme activity of colon bacteria by Bifidobacterium adolescentis SPM0212. Arch Pharm Res. 2008;31:468–73.
Le Leu RK et al. A synbiotic combination of resistant starch and Bifidobacterium lactis facilitates apoptotic deletion of carcinogen-damaged cells in rat colon. J Nutr. 2005;135:996–1001.
Pool-Zobel BL et al. Lactobacillus- and bifidobacterium-mediated antigenotoxicity in the colon of rats. Nutr Cancer. 1996;26:365–80.
Borody TJ, Paramsothy S, Agrawal G. Fecal microbiota transplantation: indications, methods, evidence, and future directions. Curr Gastroenterol Rep. 2013;15:337.
van Nood E et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407–15. This study presents the successful use of fecal transplantation to cure disease.
Acknowledgments
We thank the members of the Elinav laboratory for fruitful discussions. We apologize to authors whose relevant work was not included in this review owing to space constraints. Eran Elinav is supported by grants provided by the Abisch-Frenkel Foundation, the Kenneth Rainin Foundation, the Leona M. and Harry B. Helmsley Charitable Trust, the Israel Science Foundation, and the German-Israel Foundation, and a Marie Curie Career Integration Grant Christoph Thaiss is funded by a Boehringer Ingelheim Fonds PhD Fellowship. We thank A. Levy and Z. Levy for excellent support.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Maayan Levy, Christoph A. Thaiss, and Eran Elinav declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Maayan Levy and Christoph A. Thaiss are equal contributors.
Rights and permissions
About this article
Cite this article
Levy, M., Thaiss, C.A. & Elinav, E. The Microbiota: A New Player in the Etiology of Colorectal Cancer. Curr Colorectal Cancer Rep 10, 1–8 (2014). https://doi.org/10.1007/s11888-013-0196-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-013-0196-z