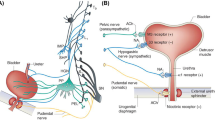
Abstract
The traditional approach to the treatment of overactive bladder (OAB) consists of behavioral therapies and pharmacologic interventions, both of which are effective in the short term but have failed to demonstrate durability over time. Patients who fail these interventions for OAB have other more invasive surgical options available that carry their own attendant risks. Herein we provide an up-to-date overview of contemporary research being performed in the treatment of OAB. Namely, we discuss the risks and benefits of pharmacogenomic medicine, whereby we can study the genetic differences in metabolic pathways that affect an individual’s response to certain drugs. We also look at molecular medicine and the use of viral vectors to deliver gene therapy to target cells. Finally, we provide an overview of stem cells and the purported therapeutic benefits as well as the challenges faced by researchers in the use of these emerging treatment modalities.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology. 2003;61:37–49.
Milsom I, Abrams P, Cardozo L, et al. How widespread are the symptoms of an overactive bladder and how are they managed? a population-based prevalence study. BJU Int. 2001;87:760–6.
Stewart WF, Van Rooyen JB, Cundiff GW, et al. Prevalence and burden of overactive bladder in the United States. World J Urol. 2003;20(6):327–36.
Coyne KS, Sexton CC, Kopp ZS, et al. The impact of overactive bladder on mental health, work productivity and health-related quality of life in the UK and Sweden: results from EpiLUTS. BJU Int. March 2011, In press.
Epstein LB, Goldstein RP. The overactive bladder and quality of life. Int J Fertil Women Med. 2005;50:30–6.
Hu-T W. TH: health-related consequences of overactive bladder: an economic perspective. BJU Int. 2005;96 Suppl 1:43–5.
Milne JL. Behavioral therapies for overactive bladder: making sense of the evidence. J Wound Ostomy Continence Nurs. 2008;35(1):93–101.
Tran K, Levin RM, Mousa SA. Behavioral intervention versus pharmacotherapy or their combinations in the management of overactive bladder dysfunction. Adv Urol. 2009;345324.
Benner JS, Nichol MB, Rovner ES, et al. Patient-reported reasons for discontinuing overactive bladder medication. BJU Int. 2010;105(9):1276–82.
• Dmochowski RR, Gomelsky A. Update on the treatment of overactive bladder. Curr Opin Urol. 2011;21(4):286–90. This comprehensive review article summarizes the current therapies directed at the treatment of OAB.
Hudson KL. Genomics, healthcare and society. N Engl J Med. 2011;365(11):1033–41.
Chancellor MB. New frontiers in the treatment of overactive bladder and incontinence. Rev Urol. 2002;4 Suppl 4:S50–6.
Hristov KL, Chen M, Kellett WF, Rovner ES, Petkov GV. Large-conductance voltage- and Ca2+−activated K+channels regulate human detrusor smooth muscle function. Am J Physiol Cell Physiol. 2011;301(4):C903–12.
Christ GJ, Day NS, Day M, et al. Bladder injection of “naked” hSlo/pcDNA3 ameliorates detrusor hyperactivity in obstructed rats in vivo. Am J Physiol Regul Integr Comp Physiol. 2001;281:R1699–709.
Goins WF, Yoshimura N, Phelan MW, et al. Gene therapy using herpes simplex virus mediated nerve growth factor expression in bladder and afferent neurons: potential treatment for diabetic bladder dysfunction. J Urol. 2001;165:1748–54.
Yoshimura N, Franks ME, Sasaki K, et al. Gene therapy of bladder pain with herpes simplex virus (HSV) vectors expressing preproenkephalin (PPE). Urology. 2001;57(6 Suppl 1):116.
• Yamamoto H, Shah S, Masters JR, Popert RJ, Dasgupta P. Stem cells in regenerative urology of the bladder. BJU Int. 2009;104(9):1183–4. This is a concise review on SCs and regenerative medicine and their applications to urologic diseases of the urinary bladder.
Yokoyama T, Yoshimura N, Dhir R, et al. Persistence and survival of autologous muscle derived cells versus bovine collagen as potential treatment of stress urinary incontinence. J Urol. 2001;165:271–6.
•• Lin CS. Advances in stem cell therapy for the lower urinary tract. World J Stem Cells. 2010;2(1):1–4. This is an excellent and comprehensive review of current research in SC therapy as it applies to diseases of the urinary bladder.
Usas A, Huard J. Muscle-derived stem cells for tissue engineering and regenerative therapy. Biomaterials. 2007;28:5401–6.
Lin CS, Xin ZC, Deng CH, et al. Defining adipose-tissue-derived stem cells in tissue and in culture. Histol Histopathol. 2010;25(6):807–15.
Nishijima S, Sugaya K, Miyazato M, et al. Restoration of bladder contraction by bone marrow transplantation in rats with underactive bladder. Biomed Res. 2007;28:275–80.
Woo LL, Tanaka ST, Anumanthan G, et al. Mesenchymal stem cell recruitment and improved bladder function following bladder outlet obstruction: preliminary data. J Urol. 2011;185(3):1132–8.
Huang YC, Shindel AW, Ning H, et al. Adipose derived stem cells ameliorate hyperlipidemia associated detrusor over activity in a rat model. J Urol. 2010;183:1232–40.
Lin CS. Stem cell therapy for the bladder—where do we stand? J Urol. 2011;185(3):779–80.
De Coppi P, Callegari A, Chiavegato A, et al. Amniotic fluid and bone marrow derived mesenchymal stem cells can be converted to smooth muscle cells in the cryoinjured rat bladder and prevent compensatory hypertrophy of surviving smooth muscle cells. J Urol. 2007;177:369–76.
Vricella GJ, Tao M, Altuntas CZ, et al. Expression of monocyte chemotactic protein 3 following simulated birth trauma in a murine model of obesity. Urology. 2010;76(6):1517.e12–7.
Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol. 2007;213(2):341–7.
Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98(5):1076–84.
Condic ML, Rao M. Regulatory issues for personalized pluripotent cells. Stem Cells. 2008;26(11):2753–8.
Bongso A, Fong CY, Gauthaman K. Taking stem cells to the clinic: major challenges. J Cell Biochem. 2008;105(6):1352–60.
Schuldiner M, Eiges R, Eden A, et al. Induced neuronal differentiation of human embryonic stem cells. Brain Res. 2001;913:201–5.
Fujikawa T, Oh SH, Pi L, et al. Teratoma formation leads to failure of treatment for type 1 diabetes using embryonic stem cell-derived insulin-producing cells. Am J Pathol. 2005;166:1781–91.
Cao F, Lin S, Xiaoyan X, et al. In vivo visualization of embryonic stem cell survival, proliferation and migration after cardiac delivery. Circulation. 2006;113:1005–14.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vricella, G.J., Hijaz, A.K. Is There a Role for Stem Cell Therapy for Overactive Bladder?. Curr Bladder Dysfunct Rep 7, 14–18 (2012). https://doi.org/10.1007/s11884-011-0115-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11884-011-0115-6