Abstract
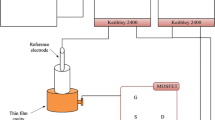
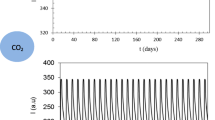
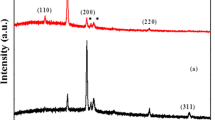
Many studies have been conducted on membranes implemented as pH sensors for measuring pH sensitivity; nevertheless, the effect of metal oxide semiconductor field effect transistors was not taken into account. Hence, this study was conducted to measure the sensitivity of the CuS membrane with and without a field effect transistor. The CuS membrane was deposited onto a glass substrate using the spray pyrolysis technique. The sensitivity and linearity in the absence of the field effect transistor were measured to be 22.86 mV/pH and 95.62%, whereas the presence of the field effect showed slightly higher sensitivity and linearity of 24 mV/pH and 98.18%, respectively. The CuS membrane synthesized in the presence of the field effect transistor also showed higher stability because the metal oxide semiconductor was not immersed in a buffer solution. Furthermore, the hysteresis of the CuS membrane, measured for 5 min, yielded a value of 12.8 mV. The structural characteristics of the membrane confirmed the formation of a single, pure CuS phase, whereas the morphological characteristics showed porous agglomerations of square nanocrystals.








Similar content being viewed by others
References
E.M. Guerra and M. Mulato, J. Sol-Gel. Sci. Technol. 52, 315 (2009).
P. Bergveld, IEEE Trans. Biomed. Eng BME 17, 70 (1970).
L.-L. Chi, J.-C. Chou, W.-Y. Chung, T.-P. Sun, and S.-K. Hsiung, Mater. Chem. Phys. 63, 19 (2000).
T. Matsuo and M. Esashi, Sensor Actuat. 1, 77 (1981).
J.-L. Wang, P.-Y. Yang, T.-Y. Hsieh, C.-C. Hwang, and M.-H. Juang, J. Nanomater. Article ID 152079, 7 (2013).
S.X. Chen, S.P. Chang, and S.J. Chang, Dig J. Nanomater. Biostruct. 9, 1505 (2014).
A. Das, D.H. Ko, C.-Hsin Chen, L.-Be Chang, C.-Sung Lai, F.-Chuan Chu, L. Chow, and R.-Ming Lin, Sensor Actuat. B-Chem. 205, 199 (2014).
H.-H. Lu, W.-S. Dai, J.-C. Chou, and H.-C. Cheng, IEEE Electron. Device Lett. 33, 1495 (2012).
K. Anuar, W.T. Tan, N. Saravanan, L.K. Khor, and S.M. Ho, J. Nepal Chem. Soc. 25, 2 (2010).
M. Lee and K. Yong, IOP Publish. Nanotech. 23, 194014 (2012).
A.D. Dhondge, S.R. Gosavi, N.M. Gosavi, C.P. Sawant, A.M. Patil, A.R. Shelke, and N.G. Deshpande, WJCMP 5, 1 (2015).
X.L. Yu, Y. Wang, H.L.W. Chan, and C.B. Cao, Micropor. Mesopor. Mater. 118, 423 (2009).
K.D. Yuan, J.J. Wu, M.L. Liu, L.L. Zhang, F.F. Xu, L.D. Chen, and F.Q. Huang, Appl. Phys. Lett. 93, 132106 (2008).
R. Das and S. Sarkar, Mater. Chem. Phys. 167, 97 (2015).
A.K. Zak, W.H.A. Majid, M.E. Abrishami, and R. Yousefi, Solid State Sci. 13, 251 (2011).
S.A.-Hilli, M. Willander, Sensors 9, 7445 (2009).
N.H. Al-Hardan, M.A.A. Hamid, N.M. Ahmed, A. Jalar, R. Shamsudin, O.K. Othman, L.K. Keng, W. Chiu, and H.N. Al-Rawi, Sensors 16, 839 (2016).
D.E. Yates, S. Levine, and T.W. Healy, J. Chem. Soc., Faraday Trans. 70, 1807 (1974).
P.-Y. Chen, L.-T. Yin, T.-H. Cho, Life Sci. J. 11, 871 (2014).
L.-T. Yin, J.-C. Chou, W.-Y. Chung, T.-P. Sun, S.-K. Hsiung, Mater. Chem. Phys. 70, 12 (2001).
C.-W. Pan, J.-C. Chou, T.-P. Sun, S.-K. Hsiung, Sensor. Actuat. B-Chem. 108, 863 (2005).
S.S.K. Hsiung, J.-C. Chou, T.-P. Sun, W.-Y. Chung, Y.-L. Chin, L. Z. Ce, Patent Application Publication US, 2004/0185591 A1 (2004).
J.L. Chiang, S.S. Jan. J.C. Chou, Y.C. Chen, Sensor Actuat. B-Chem. 76, 624 (2001).
P.-C. Yao, J.-L. Chiang, M.-C. Lee, Solid State Sci. 28, 47 (2014).
Acknowledgement
The authors gratefully acknowledge the financial support of the University Sains Malaysia and the RU Top- Down Grant 1001/CINOR/870019.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sabah, F.A., Ahmed, N.M., Hassan, Z. et al. Sensitivity of CuS Membrane pH Sensor With and Without MOSFET. JOM 69, 1134–1142 (2017). https://doi.org/10.1007/s11837-016-2165-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-016-2165-x