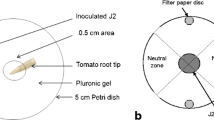
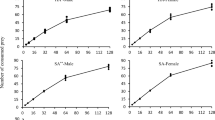
Abstract
Arthropod–plant interactions are vital in agriculture and pharmacology. However, enhancement and quantification of plant cell behavioural activity is still a challenge. Therefore, the finding of chitosan-induced anti-fungal activity in plants suggested a promising potential. Consequently, we treated autotroph and heterotroph Chenopodium rubrum L. cell cultures with chitosan to test behavioural activity with Tenebrio molitor L. larvae. We found chitosans to enhance repelling, depending on low molecular weight, partial degree of deacetylation, plant cell type and age. These effects of chitosans were compared with abiotic stresses induced by salt, osmotic changes and heat shock, collecting data of six plant cell samples, 18 plant cell treatments, 6,912 larvae and 8,424 analyses of regression and variance. Behavioural activity was quantified by linear, multivariate and nonlinear approaches, testing an exponential model in 78 segments of 5 h time kinetics. Thereby, we introduced a sensitive, low-cost bio-assay and a preference index constant as a measure of transient behaviour. Multivariate analyses revealed the five principal factors to enhance the behavioural activity in plant cells: (1) salt-osmo-sensor, (2) high molecular weight chitosan sensor, (3) acetic acid-antagonized low molecular weight chitosan sensor, (4) heat shock sensor and (5) mannitol-sensitive chitosan sensor. Thus, we suggest the independent factors (1)–(5) for the management of insect pests by autotroph and heterotroph plant materials.



Similar content being viewed by others
Abbreviations
- AAACS:
-
Acetic acid-antagonized chitosan sensor
- A30, A60, A90:
-
Autotroph cells 30, 60, 90 days after subculture
- b :
-
Time constant
- DD:
-
Degree of deacetylation
- H4, H11, H14:
-
Heterotroph cells 4, 11, 14 days after subculture
- HMWCS:
-
High molecular weight chitosan sensor
- HSS:
-
Heat shock sensor
- K PI :
-
Preference index constant
- MANCS:
-
Mannitol-sensitive chitosan sensor
- n +, n − :
-
Number of larvae in plant cell-containing, cell-free half of olfactometry field
- O70, O80, O90:
-
Oligomer chitosan 30, 45, 110 kDa, all DD > 90 %
- O70C, O80C, O90C:
-
Control for solvent of oligomer chitosan 50, 75, 183 μM acetic acid
- OS200, OS650:
-
Osmotic stress mannitol 200, 650 mM
- P, P(b), P(K PI), P(r):
-
Significance of variance analysis, b, K PI, r
- P70, P80, P90:
-
Polymer chitosan 400, 530, 450 kDa, DD 75–80, 80–90, > 90 %
- P70C, P80C, P90C:
-
Control for solvent of polymer chitosan 666, 881, 749 μM acetic acid
- PI:
-
Preference index, negative repelling, positive attracting
- PI-3, PI-5:
-
Average PI in 3 h, in 5 h
- r, r-3, r-5:
-
Pearson’s correlation coefficient, during 3 h, during 5 h
- RR:
-
Repelling ranking of plant cell treatment, 1 most repelling, 18 most attracting
- S100, S250:
-
Salt stress NaCl 100, 250 mM
- t :
-
Time
- SOS:
-
Salt-osmo-sensor
- T :
-
Period of exponential decay with significant b
- T22, T37:
-
Temperature treatment 5 min 22 °C, 37 °C
References
Berlin J, Sieg S, Strack D, Bokern M, Harms H (1986) Production of betalains by suspension cultures of Chenopodium rubrum L. Plant Cell, Tissue Organ Cult 5:163–174
Bruinsma M, van Loon JJA, Dicke M (2010) Increasing insight into induced defense mechanisms using elicitors and inhibitors. Plant Signal Behav 5:271–274
Cabrera JC, Messiaen J, Cambier P, Van Cutsem P (2006) Size, acetylation and concentration of chitooligosaccharide elicitors determine the switch from defense involving PAL activation to cell death and water peroxide production in Arabidopsis cell suspensions. Physiol Plant 127:44–56
Cavalli JF, Tomi F, Bernardini AF, Casanova J (2004) Combined analysis of the essential oil of Chenopodium ambrosioides by GC, GC-MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem Anal 15:275–279
Chaidee A, Pfeiffer W (2006) Parameters for cellular viability and membrane function in Chenopodium cells show a specific response of extracellular pH to heat shock with extreme Q10. Plant Biol 8:42–51
Chaidee A, Foissner I, Pfeiffer W (2008a) Cell-specific association of heat shock-induced proton flux with actin ring formation in Chenopodium cells: comparison of auto- and heterotroph cultures. Protoplasma 234:33–50
Chaidee A, Wongchai C, Pfeiffer W (2008b) Extracellular alkaline phosphatase is a sensitive marker for cellular stimulation and exocytosis in heterotroph cell cultures of Chenopodium rubrum. J Plant Physiol 165:1655–1666
Costanzo M, Baryshnikova A, Bellay J, Kim Y, Spear ED, Sevier CS, Ding H, Koh JLY, Toufighi K, Mostafavi S, Prinz J, St Onge RP, VanderSluis B, Makhnevych T, Vizeacoumar FJ, Alizadeh S, Bahr S, Brost RL, Chen Y, Cokol M, Deshpande R, Li Z, Lin Z-Y, Liang W, Marback M, Paw J, San Luis B-J, Shuteriqi E, Tong AHY, van Dyk N, Wallace IM, Whitney JA, Weirauch MT, Zhong G, Zhu H, Houry WA, Brudno M, Ragibizadeh S, Papp B, Pál C, Roth FP, Giaever G, Nislow C, Troyanskaya OG, Bussey H, Bader GD, Gingras A-C, Morris QD, Kim PM, Kaiser CA, Myers CL, Andrews BJ, Boone C (2010) The genetic landscape of a cell. Science 327:425–431
De Alwis R, Fujita K, Ashitani T, Kuroda K (2009) Volatile and non-volatile monoterpenes produced by elicitor-stimulated Cupressus lusitanica cultured cells. J Plant Physiol 166:720–728
Falcón AB, Cabrera JC, Costales D, Ramírez MA, Cabrera G, Toledo V, Martínez-Téllez MA (2008) The effect of size and acetylation degree of chitosan derivatives on tobacco plant protection against Phytophthora parasitica nicotianae. World J Microbiol Biotechnol 24:103–112
Gillij YG, Gleiser RM, Zygadlo JA (2008) Mosquito repellent activity of essential oils of aromatic plants growing in Argentina. Bioresource Technol 99:2507–2515
Harms H, Dehnen W, Mönch W (1977) Bezo(a)pyrene metabolites formed by plant cells. Z Naturforsch 32c:321–326
He P-Q, Tian L, Chen K-S, Hao L-H, Li G-Y (2006) Induction of volatile organic compounds of Lycopersicon esculentum Mill. and its resistance to botrytis cinerea Pers. by Burdock oligosaccharide. J Integr Plant Biol 48:550–557
Inbar M, Doostdar H, Sonoda RM, Leibee GL, Mayer RT (1998) Elicitors of plant defensive systems reduce insect densities and disease incidence. J Chem Ecol 24:135–149
Kaun KR, Azanchi R, Maung Z, Hirsh J, Heberlein U (2011) A Drosophila model for alcohol reward. Nat Neurosci 14:612–621
Koren G, Matsui D, Bailey B (2003) DEET-based insect repellents: safety implications for children and pregnant and lactating women. Can Med Assoc J 169:209–212
Limpanavech P, Chaiyasuta S, Vongpromek R, Pichyangkura R, Khunwasi C, Chadchawan S, Lotrakul P, Bunjongrat R, Chaidee A, Bangyeekhun T (2008) Chitosan effects on floral production, gene expression, and anatomical changes in the Dendrobium orchid. Sci Hort 116:65–72
Lin W, Hu X, Zhang W, Rogers WJ, Cai W (2005) Hydrogen peroxide mediates defence responses induced by chitosans of different molecular weight in rice. J Plant Physiol 162:937–944
Liu N, Chen X-G, Park H-J, Liu C-G, Liu C-S, Meng X-H, Yu L-J (2006) Effect of MW and concentration of chitosan on antibacterial activity of Escherichia coli. Carbohydr Polym 64:60–65
Maharaj R, Maharaj V, Newmarch M, Crouch NR, Bhagwandin N, Folb PI, Pillay P, Gayaram R (2010) Evaluation of selected South African ethnomedicinal plants as mosquito repellents against the Anopheles arabiensis mosquito in a rodent model. Malar J 9:301–308
Marella S, Fischler W, Kong P, Asgarian S, Rueckert E, Scott K (2006) Imaging taste responses in the fly brain reveals a functional map of taste category and behavior. Neuron 49:285–295
Masek P, Scott K (2010) Limited taste discrimination in Drosophila. Proc Natl Acad Sci USA 107:14833–14838
Nässel DR (1993) Neuropeptides in the insect brain: a review. Cell Tissue Res 273:1–29
Nation JL (2002) Insect physiology and biochemistry. CRC Press LLC, Boca Raton. ISBN 0-8493-1181-0
Pfeiffer W, Höftberger M (2001) Oxidative burst in Chenopodium rubrum suspension cells: induction by auxin and osmotic changes. Physiol Plant 111:144–150
Sosa ME, Tonn CE, Giordano OS (1994) Insect antifeedant activity of clerodane diterpenoids. J Nat Prod 57:1262–1265
Swierczek NA, Giles AC, Rankin CH, Kerr RA (2011) High-throughput behavioural analysis in C. elegans. Nat Meth 8:592–598
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5:283–291
Wegerhoff R, Breidbach O, Lobemeier M (1996) Development of Locustatachykinin immunopositive neurons in the central complex of the beetle Tenebrio molitor. J Comp Neurol 375:157–166
Wendt dos Santos AL, El Gueddari NE, Trombotto S, Moerschbacher BM (2008) Partially acetylated chitosan oligo- and polymers induce an oxidative burst in suspension cultured cells of the gymnosperm Araucaria angustifolia. Biomacromolecules 9:3411–3415
Wongchai C, Chaidee A, Pfeiffer W (2012) Multivariate analyses of salt stress and metabolite sensing in auto- and heterotroph Chenopodium cell suspensions. Plant Biol 14:129–141
Yarmolinsky DA, Zuker CS, Ryba NJ (2009) Common sense about taste: from mammals to insects. Cell 139:234–244
Zhang JS, Zhao NN, Liu QZ, Liu ZL, Du SS, Zhou L, Deng ZW (2011) Repellent constituents of essential oil of Cymbopogon distans aerial parts against two stored-product insects. J Agric Food Chem 59:9910–9915
Acknowledgments
We thank Assist. Prof. Rath Pichyangkura, the Center of Chitin-Chitosan Biomaterial, Metallurgy and Materials Research Institute, Chulalongkorn University, Bangkok, Thailand, for chitosans. This work was partially supported by the Higher Education Research Promotion and National Research University Project of Thailand (to A.C.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Guy Smagghe.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wongchai, C., Chaidee, A. & Pfeiffer, W. Enhancement and quantification of repellent activity in Chenopodium cells. Arthropod-Plant Interactions 7, 69–82 (2013). https://doi.org/10.1007/s11829-012-9221-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-012-9221-4