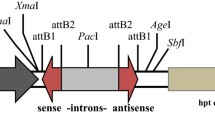
Abstract
RNA interference (RNAi) occurs naturally in plants and is involved in developmental gene regulation as well as in transposon silencing and defence against viruses. RNAi is initiated by small double-stranded RNAs (dsRNAs) of different origins, eventually leading to the silencing of genes with complementary sequences. Besides its use as a basic tool to study gene function, RNAi has widely been exploited for the generation of pathogen resistant crops and for modifying plant metabolism. For the elucidation of gene functions, silencing-inducing dsRNA is often delivered transiently by means of agroinfiltration. This method is independent of plant regeneration and allows rapid testing of multiple silencing constructs. To get more insight into the applicability of transient gene silencing, we compared a variety of different transient and stable approaches to induce silencing of the β-glucuronidase (gus) transgene by the expression of gus hairpin (hpGus) transcripts. While stable expression of the hairpin always resulted in specific gene silencing, transiently expressed hpGus transcripts could only trigger silencing when the hairpin construct was introduced simultaneously with or prior to the target gene.






Similar content being viewed by others
References
Andrieu A, Breitler JC, Sire C, Meynard D, Gantet P, Guiderdoni E (2012) An in planta, Agrobacterium‐mediated transient gene expression method for inducing gene silencing in rice (Oryza sativa L.) leaves. Rice (N Y) 5:23
Bernstein E, Caudy AA, Hammond SM, Hannon GJ (2001) Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409:363–366
Bhaskar PB, Venkateshwaran M, Wu L, Ane JM, Jiang JM (2009) Agrobacterium-mediated transient gene expression and silencing: a rapid tool for functional gene assay in potato. PLoS One 4:e5812
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Braunstein TH, Moury B, Johannessen M, Albrechtsen M (2002) Specific degradation of 3′ regions of GUS mRNA in posttranscriptionally silenced tobacco lines may be related to 5′–3′ spreading of silencing. RNA 8:1034–1044
Brodersen P, Voinnet O (2006) The diversity of RNA silencing pathways in plants. Trends Genet 22:268–280
Brodersen P, Sakvarelidze-Achard L, Bruun-Rasmussen M, Dunoyer P, Yamamoto YY, Sieburth L, Voinnet O (2008) Widespread translational inhibition by plant miRNAs and siRNAs. Science 320:1185–1190
Bruun-Rasmussen M, Madsen CT, Jessing S, Albrechtsen M (2007) Stability of Barley stripe mosaic virus-induced gene silencing in barley. Mol Plant Microbe Interact 20:1323–1331
Burch-Smith TM, Anderson JC, Martin GB, Dinesh-Kumar SP (2004) Applications and advantages of virus-induced gene silencing for gene function studies in plants. Plant J 39:734–746
Castel SE, Martienssen RA (2013) RNA interference in the nucleus: roles for small RNAs in transcription, epigenetics and beyond. Nat Rev Genet 14:100–112
Dodo HW, Konan KN, Chen FC, Egnin M, Viquez OM (2008) Alleviating peanut allergy using genetic engineering: the silencing of the immunodominant allergen Ara h 2 leads to its significant reduction and a decrease in peanut allergenicity. Plant Biotechnol J 6:135–145
English JJ, Mueller E, Baulcombe DC (1996) Suppression of virus accumulation in transgenic plants exhibiting silencing of nuclear genes. Plant Cell 8:179–188
Enrique R, Siciliano F, Favaro MA, Gerhardt N, Roeschlin R, Rigano L, Sendin L, Castagnaro A, Vojnov A, Marano MR (2011) Novel demonstration of RNAi in citrus reveals importance of citrus callose synthase in defence against Xanthomonas citri subsp. citri. Plant Biotechnol 9:394–407
Gilissen LJ, Bolhaar ST, Matos CI, Rouwendal GJ, Boone MJ, Krens FA, Zuidmeer L, Van Leeuwen A, Akkerdaas J, Hoffmann-Sommergruber K, Knulst AC, Bosch D, Van de Weg WE, Van Ree R (2005) Silencing the major apple allergen Mal d 1 by using the RNA interference approach. J Allergy Clin Immunol 115:364–369
Guidarelli M, Baraldi E (2015) Transient transformation meets gene function discovery: the strawberry fruit case. Front Plant Sci 6:444
Hamilton AJ, Baulcombe DC (1999) A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 286:950–952
Hammond SM, Bernstein E, Beach D, Hannon GJ (2000) An RNA-directed nuclease mediates post-transcriptional gene silencing in Drosophila cells. Nature 404:293–296
Hausmann L, Töpfer R (1999) Entwicklung von Plasmid-Vektoren. Vortr Pflanzenzücht 45:155–172
Helm JM, Dadami E, Kalantidis K (2011) Local RNA silencing mediated by agroinfiltration. Methods Mol Biol 744:97–108
Hoffmann T, Kalinowski G, Schwab W (2006) RNAi-induced silencing of gene expression in strawberry fruit (Fragaria × ananassa) by agroinfiltration: a rapid assay for gene function analysis. Plant J 48:818–826
Holzberg S, Brosio P, Gross C, Pogue GP (2002) Barley stripe mosaic virus-induced gene silencing in a monocot plant. Plant J 30:315–327
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes to plants. Science 227:1229–1231
Houmard NM, Mainville JL, Bonin CP, Huang S, Luethy MH, Malvar TM (2007) High-lysine corn generated by endosperm-specific suppression of lysine catabolism using RNAi. Plant Biotechnol J 5:605–614
Huang C, Qian Y, Li Z, Zhou X (2012) Virus-induced gene silencing and its application in plant functional genomics. Sci China Life Sci 55:99–108
Hutvágner G, Mlynárová L, Nap JP (2000) Detailed characterization of the posttranscriptional gene-silencing-related small RNA in a GUS gene-silenced tobacco. RNA 6:1445–1454
Ibrahim AB, Aragão FJ (2015) RNAi-mediated resistance to viruses in genetically engineered plants. Methods Mol Biol 1287:81–92
Jacobs JJMR, Litière K, Van Dijk V, Van Eldik GJ, Van Montagu M, Cornelissen M (1997) Post-transcriptional β-1,3-glucanase gene silencing involves increased transcript turnover that is translation-independent. Plant J 12:885–893
Jefferson RA, Burgess SM, Hirsh D (1986) Beta-glucuronidase from Escherichia coli as a gene-fusion marker. Proc Natl Acad Sci USA 83:8447–8451
Johansen LK, Carrington JC (2001) Silencing on the spot. Induction and suppression of RNA silencing in the Agrobacterium-mediated transient expression system. Plant Physiol 126:930–938
Koch A, Kogel K (2014) New wind in the sails: improving the agronomic value of crop plants through RNAi-mediated gene silencing. Plant Biotechnol J 12:821–831
Lacomme C (2011) Milestones in the development and applications of plant virus vector as gene silencing platforms. Curr Top Microbiol Immunol 375:89–105
Landsmann J, Llewellyn D, Dennis ES, Peacock WJ (1988) Organ regulated expression of Parasponia andersonii haemoglobin gene in transgenic tobacco plants. Mol Gen Genet 214:68–73
Levin JS, Thompson WF, Csinos AS, Stephenson MG, Weissinger AK (2005) Matrix attachment regions increase the efficiency and stability of RNA-mediated resistance to Tomato Spotted Wilt Virus in transgenic tobacco. Transgenic Res 14:193–206
Liu Q, Wu M, Zhang B, Shrestha P, Petrie J, Green AG, Singh SP (2016) Genetic enhancement of palmitic acid accumulation in cotton seed oil through RNAi down-regulation of ghKAS2 encoding β-ketoacyl-ACP synthase II (KASII). Plant Biotechnol J. doi:10.1111/pbi.12598
Lo C, Wang N, Lam E (2005) Inducible double-stranded RNA expression activates reversible transcript turnover and stable translational suppression of a target gene in transgenic tobacco. FEBS Lett 579:1498–1502
Lu S, Shi R, Tsao CC, Yi X, Li L, Chiang VL (2004) RNA silencing in plants by the expression of siRNA duplexes. Nucleic Acids Res 32:e171
Matzke MA, Kanno T, Matzke AJM (2015) RNA-directed DNA methylation: the evolution of a complex epigenetic pathway in flowering plants. Annu Rev Plant Biol 66:243–267
Napoli C, Lemieux C, Jorgensen R (1990) Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes in trans. Plant Cell 2:279–289
Ratanasut K, Monmai C, Piluk P (2015) Transient hairpin RNAi-induced silencing in floral tissues of Dendrobium Sonia ‘Earsakul’ by agroinfiltration for rapid assay of flower colour modification. Plant Cell Tissue Organ Cult 120:643–654
Regina A, Kosar-Hashemi B, Ling S, Li ZY, Rahman S, Morell M (2010) Control of starch branching in barley defined through differential RNAi suppression of starch branching enzyme IIa and IIb. J Exp Bot 61:1469–1482
Schubert D, Lechtenberg B, Forsbach A, Gils M, Bahadur S, Schmidt R (2004) Silencing in Arabidopsis T-DNA transformants: the predominant role of a gene-specific RNA sensing mechanism versus position effects. Plant Cell 16:2561–2572
Scofield SR, Huang L, Brandt AS, Gill BS (2005) Development of a virus-induced gene-silencing system for hexaploid wheat and its use in functional analysis of the Lr21-mediated leaf rust resistance pathway. Plant Physiol 138:2165–2173
Sun L, Yuan B, Zhang M, Wang L, Cui M, Wang Q, Leng P (2012) Fruit-specific RNAi-mediated suppression of SlNCED1 increases both lycopene and beta-carotene contents in tomato fruit. J Exp Bot 63:3097–3108
Sunilkumar G, Campbell LM, Puckhaber L, Stipanovic RD, Rathore KS (2006) Engineering cottonseed for use in human nutrition by tissue-specific reduction of toxic gossypol. Proc Natl Acad Sci USA 103:18054–18059
Tuschl T, Zamore PD, Lehmann R, Bartel DP, Sharp PA (1999) Targeted mRNA degradation by double-stranded RNA in vitro. Genes Dev 13:3191–3197
Urso S, Zottini M, Ruberti C, Lo Schiavo F, Stanca AM, Cattivelli L, Valè G (2013) An Agrobacterium tumefaciens-mediated gene silencing system for functional analysis in grapevine. Plant Cell Tissue Organ Cult 114:49–60
Vancanneyt G, Schmidt R, O’Connor-Sanchez A, Willmitzer L, Rocha-Sosa M (1990) Construction of an intron-containing marker gene: splicing of the intron in transgenic plants and its use in monitoring early events in Agrobacterium-mediated plant transformation. Mol Gen Genet 220:245–250
Vaucheret H (2006) Post-transcriptional small RNA pathways in plants: mechanisms and regulations. Genes Dev 20:759–771
Waterhouse PM, Helliwell CA (2003) Exploring plant genomes by RNA-induced gene silencing. Nat Rev Genet 4:29–38
Weinmann P, Gossen M, Hillen W, Bujard H, Gatz C (1994) A chimeric transactivator allows tetracycline-responsive gene expression in whole plants. Plant J 5:559–569
Wesley SV, Helliwell CA, Smith NA, Wang M, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, Robinson SP, Gleave AP, Green AG, Waterhouse PM (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27:581–590
Zhang BH (2015) MicroRNA: a new target for improving plant tolerance to abiotic stress. J Exp Bot 66:1749–1761
Acknowledgements
We thank Tobias Wille, Birgit Effinghausen-Büssing, and Gudrun Püster for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Manske, U., Landsmann, J. & Dietz-Pfeilstetter, A. Comparison of different methods for the establishment of RNA silencing in plants. Plant Biotechnol Rep 11, 115–125 (2017). https://doi.org/10.1007/s11816-017-0436-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-017-0436-9