Abstract
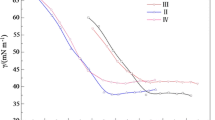
A series of novel cationic gemini surfactants were synthesized from corresponding amido-amines in a single step reaction. The amido-amines were obtained from long chain carboxylic acids and 3-N,N-dimethylamino-1-propyl-amine with excellent isolated yield (up to 95 %). All the synthesized quaternary ammonium compounds (QACs) were further investigated for surface active properties. The critical micelle concentration (CMC) and the effectiveness of surface tension reduction were determined. The surface tension measurements of newly synthesized gemini surfactants showed good water solubility, and low CMC values, had great efficiency in lowering the surface tension and a strong adsorption at the air/water interface than the corresponding monomeric surfactants. Further, the antibacterial activity of the synthesized QACs against both Gram-positive and Gram-negative bacteria was also investigated.



Similar content being viewed by others
References
Abbanat D, Macielag M, Bushrajesh K (2003) Novel antibacterial agents for the treatment of serious Gram-positive infection. Expert Opin Investig Drugs 12:379–399
Pootoolal J, Neu J, Wright GD (2002) Glycopeptide antibiotic resistance. Annu Rev Pharmacol Toxicol 42:381–408
Raad II, Hanna HA, Hachem RY, Dvorak T, Arbuckle RB, Chaiban G, Rice LB (2004) Clinical-use-associated decrease in susceptibility of vancomycin-resistant Enterococcus faecium to linezolid: comparison with quinupristin-dalfopristin. Antimicrob Agents Chemother 48:3583–3585
Goldstein FW (2007) Combating resistance in a challenging, changing environment. Clin Microbiol Infect 13:2–6
Fidler DF (1998) Legal issues associated with antimicrobial drug resistance. Emerg Infect Dis 4:169–177
Macchiarulo A, Constantino G, Fringuelli D, Vecchiarelli A, Schiaffella F, Fringuelli R (2002) 1,4-Benzothiazine and 1,4-benzoxazine imidazole derivatives with antifungal activity: a docking study. Bioorg Med Chem 10:3415–3423
Diz M, Manresa A, Pinazo A, Erra P, Infante MR (1994) Synthesis, surface active properties and antimicrobial activity of new bis quaternary ammonium compounds. J Chem Soc Perkin Trans 2:1871–1879
Kourai H, Yabuhara T, Shirai A, Maeda T, Nagamune H (2006) Synthesis and antimicrobial activities of a series of new bis-quaternary ammonium compounds. Eur J Med Chem 41:437–444
Menger FM, Littau CA (1993) Gemini surfactants: a new class of self-assembling molecules. J Am Chem Soc 115:10083–10090
Chunli X, Hailin Z, Tingting Z, Duanlin C, Zhiyong H (2011) Synthesis and properties of novel alkylbetaine zwitterionic gemini surfactants derived from cyanuric chloride. Colloid Surf A 375:141–146
Oda R, Hac I, Candau SJ (1997) Gemini surfactants, the effect of hydrophobic chain length and dissymmetry. Chem Commun 21:2105–2106
Garcia MY, Ribosa I, Guindulain T, Sanchez-Leal J, Vives-Rego J (2001) Fate and effect of monoalkyl quaternary ammonium surfactants in the aquatic environment. J. Environ Pollut 111:169–175
Perez L, Torres JL, Manresa A, Solans C, Infante MR (1996) Synthesis, aggregation, and biological properties of a new class of gemini cationic amphiphilic compounds from arginine, bis(Args). Langmuir 12:5296–5301
Weihs D, Danino D, Pinazo-Gassol A, Perez L, Franses EI, Talmon Y (2005) Self-aggregation in dimeric arginine-based cationic surfactants solutions. Colloid Surf A Physicochem Eng Asp 255:73–78
Fisicaro E, Compari C, Duce E, Donofrio G, Rozycka-Roszak B, Wozniak E (2005) Biologically active bisquaternary ammonium chlorides: physico–chemical properties of long chain amphiphiles and their evaluation as non-viral vectors for gene delivery. Biochim Biophys Acta 1722:224–233
Gaucheron J, Wong T, Wong KF, Maurer N, Cullis PR (2002) Synthesis and properties of novel tetraalkyl cationic lipids. Bioconjug Chem 1:671–675
Zana R (2002) Dimeric (Gemini) surfactants: effect of the spacer group on the association behavior in aqueous solution. J Colloid Interface Sci 248:203–220
Fielden ML, Perrin C, Kremer A, Bergsma M, Stuart MC, Camilleri Jan B, Engberts FN (2001) Sugar-based tertiary amino gemini surfactants with a vesicle-to-micelle transition in the endosomal pH range mediate efficient transfection in vitro. Eur J Biochem 268:1269–1279
Bell PC, Bergsma M, Dolbnya IP, Bras W, Stuart MCA, Rowan AE, Feiters MC, Engberts Jan BFN (2003) Transfection mediated by gemini surfactants: engineered escape from the endosomal compartment. J Am Chem Soc 125:1551–1558
Karlsson LV, Eijk MCP, Soderman O (2002) Interaction of bovine myelin basic protein with triphosphoinositide. J Colloid Interface Sci 252:290–296
Sharkawy HE (2007) Cycloalkylation reactions of fatty amines with α,ω-dihaloalkanes: role of bis-quaternary ammonium salts as phase-transfer catalysts. Cat Commun 8:855–860
Christine HF, Michael HC (1986) Comparative in vitro activities of ciprofloxacin and other 4-quinolones against mycobacterium tuberculosis and mycobacterium intracellular. Antimicrob Agents Chemother 29:386–388
Paulo A, Pires R, Omar A, Seoud E (2006) Surfactants with an amide group “spacer”: synthesis of 3-(acylaminopropyl) trimethylammonium chlorides and their aggregation in aqueous solutions. J Colloid Interface Sci 304:474–485
Cronin MTD, Aptula AO, Dearden JC, Duffy JC, Ntezeva TI, Patel H, Philip H, Rowe T, Wayne S, Andrew P, Worth, Konstantinos V, Gerrit S (2002) Structure-based classification of antibacterial activity. J Chem Inf Comput Sci 42:869–878
Acknowledgments
The author (AKG) is greatly thankful to the UGC (University Grant Commission, India) for providing a fellowship. The authors would also extend their special thanks to Mr. Sharad Gaikwad and Mr. Chetan Pawar, K. T. H. M. College, Nashik, Maharashtra, India for carrying out antibacterial activity analysis during this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Ghumare, A.K., Pawar, B.V. & Bhagwat, S.S. Synthesis and Antibacterial Activity of Novel Amido-Amine-Based Cationic Gemini Surfactants. J Surfact Deterg 16, 85–93 (2013). https://doi.org/10.1007/s11743-012-1406-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-012-1406-4