Abstract
Background
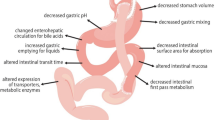
Data on the absorption of orally administered drugs following Roux-en-Y gastric bypass (RYGB) surgery in obese patients are limited and inconclusive. As it is difficult to predict changes in absorption, studies on frequently used drugs in this population are necessary. Acetylsalicylic acid (ASA) and omeprazole are two commonly prescribed drugs in obese patients.
Methods
In this repeated measures study, omeprazole and salicylic acid (SA) serum concentrations were measured before and after RYGB in 34 morbidly obese subjects. Time to maximum concentration (Tmax), lag time (Tlag), maximum concentration (Cmax), and area under the serum concentration versus time curve (AUC) were calculated for both drugs to determine possible differences in drug absorption after the procedure.
Results
For SA, Tmax significantly decreased after RYGB, while both Cmax and AUC0–24 significantly increased. For omeprazole, both Tmax and Tlag significantly decreased after RYGB, while Cmax significantly increased. Mean AUC0–12 significantly decreased post-surgery. The difference in AUC0–12 before and after surgery varied between subjects.
Conclusions
Our study shows a faster absorption of both ASA and omeprazole after RYGB. The exposure to ASA is higher post-surgery, but the standard dose of 80 mg does not need to be modified, considering its range in effective dose. The exposure to omeprazole is, on average, decreased after surgery. Clinicians should be aware to increase the dose of omeprazole if symptoms suggest inadequate response.





Similar content being viewed by others
References
World Health Organization. WHO factsheet Obesity and Overweight. 1-1-2015. 13-9-2015. Ref Type: Online Source http://www.who.int/mediacentre/factsheets/fs311/en/.
Canadian Agency for Drugs and Technologies in Health. Bariatric surgical procedures for obese and morbidly obese patients: a review of comparative clinical and cost-effectiveness, and guidelines. 24-4-2014. 13-9-2015. Ref Type: Online Source http://www.ncbi.nlm.nih.gov/books/NBK264224/.
Edwards A, Ensom MH. Pharmacokinetic effects of bariatric surgery. Ann Pharmacother. 2012;46(1):130–6.
Padwal R. A systematic review of drug absorption following bariatric surgery and its theoretical implications. Obes Rev. 2010;11(1):41–50.
Yska JP, van der Linde S, Tapper VV, Apers JA, Emous M, Totte ER, et al. Influence of bariatric surgery on the use and pharmacokinetics of some major drug classes. Obes Surg. 2013;23(6):819–25.
Titus R, Kastenmeier A, Otterson MF. Consequences of gastrointestinal surgery on drug absorption. Nutr Clin Pract. 2013;28(4):429–36.
Sawaya RA, Jaffe J, Friedenberg L, Friedenberg FK. Vitamin, mineral, and drug absorption following bariatric surgery. Curr Drug Metab. 2012;13(9):1345–55.
Fuller AK, Tingle D, DeVane CL, Scott JA, Stewart RB. Haloperidol pharmacokinetics following gastric bypass surgery. J Clin Psychopharmacol. 1986;6(6):376–8.
Hamad GG, Helsel JC, Perel JM, Kozak GM, McShea MC, Hughes C, et al. The effect of gastric bypass on the pharmacokinetics of serotonin reuptake inhibitors. Am J Psychiatry. 2012;169(3):256–63.
Magee SR, Shih G, Hume A. Malabsorption of oral antibiotics in pregnancy after gastric bypass surgery. J Am Board Fam Med. 2007;20(3):310–3.
Prince RA, Pincheira JC, Mason EE, Printen KJ. Influence of bariatric surgery on erythromycin absorption. J Clin Pharmacol. 1984;24(11–12):523–7.
Roerig JL, Steffen K, Zimmerman C, Mitchell JE, Crosby RD, Cao L. Preliminary comparison of sertraline levels in postbariatric surgery patients versus matched nonsurgical cohort. Surg Obes Relat Dis. 2012;8(1):62–6.
Skottheim IB, Stormark K, Christensen H, Jakobsen GS, Hjelmesaeth J, Jenssen T, et al. Significantly altered systemic exposure to atorvastatin acid following gastric bypass surgery in morbidly obese patients. Clin Pharmacol Ther. 2009;86(3):311–8.
Wills SM, Zekman R, Bestul D, Kuwajerwala N, Decker D. Tamoxifen malabsorption after Roux-en-Y gastric bypass surgery: case series and review of the literature. Pharmacotherapy. 2010;30(2):217.
Rogers CC, Alloway RR, Alexander JW, Cardi M, Trofe J, Vinks AA. Pharmacokinetics of mycophenolic acid, tacrolimus and sirolimus after gastric bypass surgery in end-stage renal disease and transplant patients: a pilot study. Clin Transplant. 2008;22(3):281–91.
Rubio IG, Galrao AL, Santo MA, Zanini AC, Medeiros-Neto G. Levothyroxine absorption in morbidly obese patients before and after Roux-En-Y gastric bypass (RYGB) surgery. Obes Surg. 2012;22(2):253–8.
Hamilton R, Thai XC, Ameri D, Pai MP. Oral bioavailability of linezolid before and after Roux-en-Y gastric bypass surgery: is dose modification necessary in obese subjects? J Antimicrob Chemother. 2013;68(3):666–73.
De Smet J, Colin P, De PP, Ruige J, Batens H, Van NY, et al. Oral bioavailability of moxifloxacin after Roux-en-Y gastric bypass surgery. J Antimicrob Chemother. 2012;67(1):226–9.
Koninklijke Nederlandse Maatschappij ter bevordering der Pharmacie. KNMP Kennisbank. 1-9-2015. 13-9-2015. Ref Type: Online Source https://kennisbank.knmp.nl/.
Stichting Farmaceutische Kengetallen. Data en Feiten 2015. 1-8-2015. 13-9-2015. Ref Type: Online Source https://www.sfk.nl/nieuws-publicaties/data-en-feiten/data-en-feiten-2015.
Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002;324(7329):71–86.
Hunfeld NGM. Clinical effects of proton pump inhibitors, Focus on pharmacogenetics, kinetics and dynamics. Rotterdam: Erasmus University Rotterdam; 2010.
D’Hondt MA, Pottel H, Devriendt D, Van RF, Vansteenkiste F. Can a short course of prophylactic low-dose proton pump inhibitor therapy prevent stomal ulceration after laparoscopic Roux-en-Y gastric bypass? Obes Surg. 2010;20(5):595–9.
Dallal RM, Bailey LA. Ulcer disease after gastric bypass surgery. Surg Obes Relat Dis. 2006;2(4):455–9.
Garrido Jr AB, Rossi M, Lima Jr SE, Brenner AS, Gomes Jr CA. Early marginal ulcer following Roux-en-Y gastric bypass under proton pump inhibitor treatment: prospective multicentric study. Arq Gastroenterol. 2010;47(2):130–4.
Kwaliteitsinstituut voor de gezondheidszorg C. NSAID-gebruik en preventie van maagschade. Van Zuiden communications BV; 2003. http://www.diliguide.nl/document/2188
van Schaik RH, Kok M, Sweep FC, van Vliet M, van Fessem M, Meijer-van Gelder ME, et al. The CYP2C19*2 genotype predicts tamoxifen treatment outcome in advanced breast cancer patients. Pharmacogenomics. 2011;12(8):1137–46.
Pharmachemie. SmPC Acetylsalicylzuur 80 mg. 3-10-2014. 13-9-2015. Ref Type: Online Source http://db.cbg-meb.nl/IB-teksten/h102651.pdf.
Tandra S, Chalasani N, Jones DR, Mattar S, Hall SD, Vuppalanchi R. Pharmacokinetic and pharmacodynamic alterations in the Roux-en-Y gastric bypass recipients. Ann Surg. 2013;258(2):262–9.
AstraZeneca. SmPC Losec MUPS 20 mg. 27-8-2014. 13-9-2015. Ref Type: Online Source http://db.cbg-meb.nl/IB-teksten/h21684.pdf.
Acknowledgments
The authors would like to thank Central Hospital Pharmacy, The Hague, Medical Centre Haaglanden, The Hague, and the Dutch Obesity Clinic West, The Hague for funding this study, Henk Trumpie for the analysis of the samples, and Anne Hanschke, Piet Sturm, Yolanda Ooms, and Caroline Wiersma for their contribution in performing the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Central Hospital Pharmacy, The Hague, Medical Centre Haaglanden, The Hague, and the Dutch Obesity Clinic West, The Hague.
Conflicts of Interest
All authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Approval for the study was obtained from the local ethical committee of The Hague (METCZWH, The Hague, the Netherlands). The study was registered in the Dutch Trial Register (NTR3939).
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
The ERY-PAO study was conducted at the department of clinical pharmacy, Central Hospital Pharmacy, Medical Centre The Hague, The Hague, The Netherlands and the Dutch Obesity Clinic West, The Hague, The Netherlands.
Rights and permissions
About this article
Cite this article
Mitrov-Winkelmolen, L., van Buul-Gast, MC.W., Swank, D.J. et al. The Effect of Roux-en-Y Gastric Bypass Surgery in Morbidly Obese Patients on Pharmacokinetics of (Acetyl)Salicylic Acid and Omeprazole: the ERY-PAO Study. OBES SURG 26, 2051–2058 (2016). https://doi.org/10.1007/s11695-016-2065-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-016-2065-8