Abstract
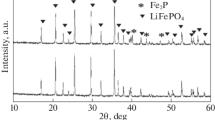
With the aim of investigating the influence of pH value on the properties of NH4Fe2(OH)(PO4)2·2H2O precursor and LiFePO4/C composite, we prepared a series of (NH4)Fe2(OH)(PO4)2·2H2O by adjusting the pH value in the chemical precipitation process. In addition, the LiFePO4/C composite was fabricated by using (NH4)Fe2(OH)(PO4)2·2H2O as precursor. The images of scanning electron microscopy demonstrate that the precursor particle size increases as the pH value increases, which facilitates the tuning of the properties of the LiFePO4/C composite. A further investigation indicates that an increase of viscosity of the precipitate slurry in the chemical precipitation process is responsible for the change of precursor particle size. In addition, the electrochemical performance of the LiFePO4/C composite is also governed by pH value. LiFePO4/C composite prepared at the pH value of 4.0 delivers a discharge capacity of 91 mAh g−1 and 114 mAh cm−3 at 10 C, showing a capacity retention of 99.48% after 100 cycles, which confirms the excellent rate capability and cycling stability.
Similar content being viewed by others
References
C.-T. Hsieh, I.L. Chen, W.-Y. Chen, and J.-P. Wang, Electrochim. Acta 83, 202 (2012).
J.W. Fergus, J. Power Sources 195, 939 (2010).
A.K. Padhi, K.S. Nanjundaswamy, and J.B. Goodenough, J. Electrochem. Soc. 144, 1188 (1997).
D. Kim, J. Lim, V. Mathew, B. Koo, Y. Paik, D. Ahn, S.-M. Paek, and J. Kim, J. Mater. Chem. 22, 2624 (2012).
D.-H. Kim and J. Kim, Electrochem. Solid-State Lett. 9, A439 (2006).
D.-H. Kim and J. Kim, J. Phys. Chem. Solids 68, 734 (2007).
S. Ju, H. Peng, G. Li, and K. Chen, Mater. Lett. 74, 22 (2012).
K. Saravanan, M.V. Reddy, P. Balaya, H. Gong, B.V.R. Chowdari, and J.J. Vittal, J. Mater. Chem. 19, 605 (2009).
S. Yu, S. Dan, G. Luo, W. Liu, Y. Luo, X. Yu, and Y. Fang, J. Solid State Electrochem. 16, 1675 (2012).
Z. Liu, X. Zhang, and L. Hong, J. Appl. Electrochem. 39, 2433 (2009).
J. Su, X.-L. Wu, C.-P. Yang, J.-S. Lee, J. Kim, and Y.-G. Guo, J. Phys. Chem. C 116, 5019 (2012).
X.-M. Liu, P. Yan, Y.-Y. Xie, H. Yang, X.-D. Shen, and Z.-F. Ma, Chem. Commun. 49, 5396 (2013).
S.-W. Cao, Y.-J. Zhu, and J.-B. Cui, J. Solid State Chem. 183, 1704 (2010).
P. Yan, L. Lu, X.-M. Liu, Y. Cao, Z.-P. Zhang, H. Yang, and X.-D. Shen, J. Mater. Chem. A 1, 10429 (2013).
K. Tang, X. Yu, J. Sun, H. Li, and X. Huang, Electrochim. Acta 56, 4869 (2011).
L. Qian, Y. Xia, W. Zhang, H. Huang, Y. Gan, H. Zeng, and X. Tao, Microporous Mesoporous Mater. 152, 128 (2012).
F. Cheng, W. Wan, Z. Tan, Y. Huang, H. Zhou, J. Chen, and X. Zhang, Electrochim. Acta 56, 2999 (2011).
A.Y. Shenouda and H.K. Liu, J. Alloy Compd. 477, 498 (2009).
C. Gao, H. Liu, G. Liu, J. Zhang, and W. Wang, Mater. Sci. Eng. B 178, 272 (2013).
Y. Xia, W. Zhang, H. Huang, Y. Gan, C. Li, and X. Tao, Mater. Sci. Eng. B 176, 633 (2011).
Acknowledgement
This work was financially supported by the Science and technology support program in Sichuan province (No. 14ZC2237).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, XB., Liu, GB., Wang, Y. et al. Influence of pH Value on the Properties of NH4Fe2(OH)- (PO4)2·2H2O Precursor and LiFePO4/C Composite. J. Electron. Mater. 44, 1008–1014 (2015). https://doi.org/10.1007/s11664-014-3613-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-014-3613-4