Abstract
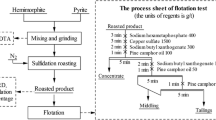
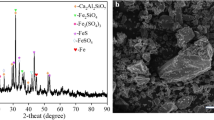
The selective sulfidation of lead smelter slag with pyrite in the presence of carbon and Na salts, and the flotation behavior of synthetic ZnS were studied. The effects of temperature, time, pyrite dosage, Na salts, and carbon additions were investigated based on thermodynamic calculation, and correspondingly, the growth mechanism of ZnS particles was studied at high temperatures. The results indicated that the zinc in lead smelter slag was selectively converted into zinc sulfides by sulfidation roasting. The sulfidation degree of zinc was increased until the temperature, time, pyrite, and carbon dosages reached their optimum values, under which it was more than 95 pct. The growth of ZnS particles largely depended upon roasting temperature, and the ZnS grains were significantly increased above 1373 K (1100 °C) due to the formation of a liquid phase. After the roasting, the zinc sulfides generated had a good floatability, and 88.34 pct of zinc was recovered by conventional flotation.









Similar content being viewed by others
References
F. Huang, Y. Liao, J. Zhou, Y. Wang, and H. Li: Sep. Purif. Technol., 2015, vol. 156, pp. 572-581.
[2] J. Han, W. Liu, W. Qin, B. Peng, K. Yang, and Y. Zheng: J. Ind. Eng. Chem., 2015, vol. 22, pp. 272-279.
[3] J. Han, W. Liu, W. Qin, Y. Zheng, and H. Luo: Metall. Mater. Trans. B, 2015, vol. 47, pp. 686-693.
[4] N.M. Piatak, M.B. Parsons, and R.R. Seal Ii: Appl. Geochem., 2015, vol. 57, pp. 236-266.
[5] J. Han, W. Liu, W. Qin, K. Yang, D. Wang, and H. Luo: Sep. Purif. Technol., 2015, vol. 154, pp. 263-270.
[6] F. Cihangir, B. Ercikdi, A. Kesimal, H. Deveci, and F. Erdemir: Miner. Eng., 2015, vol. 83, pp. 117-127.
M. Albitar, M.S. Mohamed, P. Visintin, and M. Drechsler: Constr. Build. Mater., 2015, vol. 83, pp. 128-135.
L.R.P. de Andrade and L.A. Bernardez: J. Hazard. Mater., 2011, vol. 189, pp. 692-699.
[9] D. Kuchar, T. Fukuta, M.S. Onyango, and H. Matsuda: J J. Hazard. Mater., 2006, vol. 137, pp. 185-191.
[10] D. Kuchar, T. Fukuta, M.S. Onyango, and H. Matsuda: Chemosphere, 2007, vol. 67, pp. 1518-1525.
[11] D. Wu, S. Wen, J. Deng, J. Liu, and Y. Mao: Appl. Surf. Sci., 2015, vol. 329, pp. 315-320.
[12] W. Yuan, J. Li, Q. Zhang, and F. Saito: Powder Technol., 2012, vol. 230, pp. 63-66.
[13] Y. Liang, L. Chai, H. Liu, X. Min, Q. Mahmood, H. Zhang, and Y. Ke: Miner. Eng., 2012, vol. 25, pp. 14-19.
[14] Y. Liang, L. Chai, X. Min, C. Tang, H. Zhang, Y. Ke, and X. Xie: J. Hazard. Mater., 2012, vols. 217–218, pp. 307-314.
[15] L. Chai, Y. Ke, X. Min, B. Zhou, K. Xue, and J. Chen: Sep. Purif. Technol., 2015, vol. 154, pp. 76-81.
[16] J. Wang, J. Lu, Q. Zhang, and F. Saito: Ind. Eng. Chem. Res., 2003, vol. 42, pp. 5813-5818.
[17] C.T. Harris, J.G. Peacey, and C.A. Pickles: Miner. Eng., 2011, vol. 24, pp. 651-660.
[18] C.T. Harris, J.G. Peacey, and C.A. Pickles: Miner. Eng., 2013. vol. 54, pp. 21-31.
[19] C.A. Pickles, C.T. Harris, J. Peacey, and J. Forster: Miner. Eng., 2013, vol. 54, pp. 52-62.
[20] J. Han, W. Liu, D. Wang, F. Jiao, and W. Qin: Metall. Mater. Trans. B, 2015, vol. 47, pp. 344-354.
[21] Y. Li, J. Wang, C. Wei, C. Liu, J. Jiang, and F. Wang: Miner. Eng., 2010, vol. 23, pp. 563-566.
[22] Y. Zheng, W. Liu, W. Qin, J. Han, K. Yang, H. Luo, and D. Wang: Can. Metall. Quart., 2014, vol. 54, pp. 92-100.
[23] Y. Zheng, W. Liu, W. Qin, Y. Kong, H. Luo, and J. Han: Sep. Sci. Technol., 2014, vol. 49, pp. 783-791.
[24] R.M. German, P. Suri, and S.J. Park: J. Mater. Sci., 2008, vol. 44, pp. 1-39.
[25] V.T. Golovchan: J. Superhard Mater., 2014, vol. 36, pp. 89-95.
[26] A. Sarrafi, B. Rahmati, H.R. Hassani, and H.H.A. Shirazi: Miner. Eng., 2004, vol. 17, pp. 457-459.
[27] W.J. Bruckard, M. Somerville, and F. Hao: Miner. Eng., 2004, vol. 17, pp. 495-504.
[28] N. Karimi, R. Vaghar, M.R.T. Mohammadi, and S.A. Hashemi: J. Inst. Eng. India Ser. D, 2013, vol. 94, pp. 43-50.
[29] C.A. Pickles: J. Hazard. Mater., 2009, vol. 166, pp. 1030-1042.
[30] C.A. Pickles: Sep. Purif. Technol., 2008, vol. 59, pp. 115-128.
[31] G. Li, T. Shi, M. Rao, T. Jiang, and Y. Zhang: Miner. Eng., 2012, vol. 32, pp. 19-26.
[32] M. Jiang, T. Sun, Z. Liu, J. Kou, N. Liu, and S. Zhang: Int. J. Miner. Process., 2013, vol. 123, pp. 32-38.
Acknowledgments
The authors would like to thank the Innovation Project for Postgraduates of Central South University (2015zzts090) and National Natural Science Foundation of China (51204210) for funding this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted March 6, 2016.
Rights and permissions
About this article
Cite this article
Han, J., Liu, W., Wang, D. et al. Selective Sulfidation of Lead Smelter Slag with Pyrite and Flotation Behavior of Synthetic ZnS. Metall Mater Trans B 47, 2400–2410 (2016). https://doi.org/10.1007/s11663-016-0693-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0693-y