Abstract
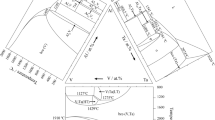
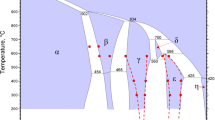
Phase equilibria in the SiO2-K2O-CaO system have been experimentally investigated in the SiO2-rich area. High-temperature equilibration, rapid quenching, and electron probe X-ray microanalysis (EPMA) techniques have been used in this study. K2O may vaporize during EPMA measurements causing significant uncertainties. In the present study, optimum EPMA operating conditions have been determined in order to accurately measure K2O concentrations in the quenched samples. The compositions of all phases present in the quenched sample were measured using EPMA with optimum operating parameters. The following primary phase fields were identified in the composition range investigated: SiO2, CaO·SiO2, 2CaO·SiO2, K2O·2CaO·2SiO2, and K2O·6CaO·4SiO2. The isotherms between 1273 K and 1473 K (1000 °C and 1200 °C) in these primary phase fields have been determined. The presence of the compounds K2O·2CaO·2SiO2 and K2O·6CaO·4SiO2 has been confirmed.






Similar content being viewed by others
References
W. Stern and Y. Gerber: Archaeometry, 2004, vol. 46, pp. 137-56.
S. V. Vassilev, D. Baxter, L. K. Andersen, and C. G. Vassileva: Fuel, 2010, vol. 89, pp. 913-33.
H. Hughes: Trans. Br. Ceram. Soc., 1966, vol. 65, pp. 661-79.
T. Akiyama, Y. Yao, and S. Matsuno: Jpn. J. Soil Sci. Plant Nutr., 2001, vol. 72, pp. 484-8.
C. Vitale-Brovarone and E. Verné: J. Mater. Sci. Mater. Med., 2005, vol. 16, pp. 863-71.
G. Morey, F. Kracek, and N. Bowen: J. Soc. Glass Technol., 1930, vol. 14, pp. 149-87.
G. Morey, F. Kracek, and N. Bowen: J. Soc. Glass Technol., 1931, vol. 14, pp. 57-8.
F. Kracek, N. Brown, and G. Morey: J. Phys. Chem., 1937, vol. 41, pp. 1183-93.
W. C. Taylor: J. Res. Natl. Bur Stand., 1941, vol. 27, pp. 311-23.
E. Arroyabe, R. Tessadri, D. M. Többens, and V. Kahlenberg: J. Am. Ceram. Soc., 2011, vol. 94, pp. 2652-5.
H. Ohsato, T. Sugimura, S. Hayashi, and T. Ogihara: Bull. Nagoya Inst. Technol., 1980, vol. 32, pp. 123-30.
E. Arroyabe, R. Kaindl, D. M. Többens, and V. Kahlenberg: J. Solid State Chem., 2009, vol. 182, pp. 3254-61.
L. Zhang, C. Schmetterer, and P. J. Masset: High Temp. Mater. Processes, 2013, vol. 32, pp. 223-8.
C. Bale, E. Bélisle, P. Chartrand, S. Degterov: Calphad, 2009, vol. 33, pp. 295-311.
M. Chen and B. Zhao: J. Am. Ceram. Soc., 2013, vol. 96, pp. 3631-6.
B. Zhao, P. C. Hayes, and E. Jak: Metall. Mater. Trans. B, 2010, vol. 41, pp. 374-85.
J. L. Lineweaver: J. Appl. Phys., 1963, vol. 34, pp. 1786-91.
Acknowledgments
The authors would like to thank Ms. Jie Yu for the lab assistance. The authors acknowledge the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy and Microanalysis, The University of Queensland, for providing facilities and scientific and technical assistance .
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted January 6, 2016.
Rights and permissions
About this article
Cite this article
Chen, M., Hou, X., Chen, J. et al. Phase Equilibria Studies in the SiO2-K2O-CaO System. Metall Mater Trans B 47, 1690–1696 (2016). https://doi.org/10.1007/s11663-016-0623-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0623-z