Abstract
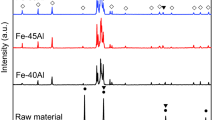
With its historically long popularity in major structural applications, the use of iron (Fe) has also recently begun to be explored as an advanced functional material. For this purpose, it is more advantageous to use Fe as a porous structure, simply because it can provide a greater surface area and a higher reaction rate. This study uses a freeze-casting method, which consists of simple and low-cost processing steps, to produce Fe foam with a mean pore size of 10 μm. We examine the influences of various parameters (i.e., mold bottom temperature, powder content, and sintering time) on the processing of Fe foam, along with its oxidation kinetics at 823 K (550 °C) with various heat-treatment times. We confirm that Fe2O3 and Fe3O4 oxide layers are successfully formed on the surface of Fe foam. With the Fe oxide layers as an active anode material, the Fe foam can potentially be used as a three-dimensional anode current collector for an advanced lithium-ion battery.









Similar content being viewed by others
References
B.H. Smith, S. Szyniszewski, J.F. Hajjar, B.W. Schafer, and S.R. Arwade: J. Constr. Steel Res., 2012, vol. 71, pp. 1–10.
P.A. Xu, G.M. Zeng, D.L. Huang, C.L. Feng, S. Hu, M.H. Zhao, C. Lai, Z. Wei, C. Huang, and G.X. Xie: Sci. Total Environ., 2012, vol. 424, pp. 1–10.
A.B. Cundy, L. Hopkinson, and R.L.D. Whitby: Sci. Total Environ., 2008, vol. 400, pp. 42–51.
J. Chen, L.N. Xu, W.Y. Li, and X.L. Gou: Adv. Mater., 2005, vol. 17, pp. 582–586.
S. Jin, H. Deng, D. Long,, X. Liu, L. Zhan, X. Liang, W. Qiao, and L. Ling: J. Power Sources, 2011, vol. 196, pp. 3887–3893.
J. Capek, and D. Vojtech: Mater. Sci. Eng., 2014, vol. 43, pp. 494–501.
M. Peustera, C. Hessea, T. Schlooa, C. Finkb, P. Beerbauma, and C.V. Schnakenburg: biomaterials, 2006, vol. 27, pp. 4955–4962.
M.F. Ashby, A.G. Evans, N.A. Fleck, L.J. Gibson, J.W. Hutchinson, and H.N.G. Wadley: Metal Foams: A Design Guide, Butterworth-Heinemann, Boston, 2000.
J. Banhart: Prog. Mater Sci., 2001, vol. 46, pp. 559–632.
L.P. Lefebvre, J. Banhart, and D.C. Dunand: Adv. Eng. Mater., 2008, vol. 10, pp. 775–787.
C. Park, and S.R. Nutt: Mater. Sci. Eng. A, 2001, vol. 229, pp. 68–74.
T. Murakami, K. Ohara, T. Narushima, and C. Ouchi: Mater. Trans., 2007, vol. 48, pp. 2937–2944.
T. Murakami, T. Akagi, and E. kasai: Proc. Mater. Sci., 2014, vol. 4, pp. 27–32.
S.T. Szyniszewski, B.H. Smith, J.F. Hajjar, B.W. Schafer, and S.R. Arwade: Mater. Des., 2014, vol. 54, pp. 1083–1094.
T. Ikeda, T. Aoki, and H Nakajima: Metall. Mater. Trans. A, 2005, vol. 36A, pp. 77–86.
M. Vesenjak, A. Kovacic, M. Tane, M. Borovinsek, H. Nakajima, and Z.R. Ren: Comput. Mater. Sci, 2012, vol. 65, pp. 37–43.
S. Deville: Adv. Eng. Mater., 2008, vol. 10, pp. 155–69.
S. Deville, E. Saiz, and A.P. Tomsia: Acta Mater., 2007, vol. 55, pp. 1965–74.
U.G.K. Wegst, M. schecter, A.E. Donius, and P.M. Hunger: Phil. Trans. R. Soc. A, 2010, vol. 368, pp. 2099–121.
Y. Chino, and D.C. Dunand: Acta Mater., 2008, vol. 56, pp. 105–113.
S. Deville, E. Saiz, and A.P. Tomsia: Biomaterials, 2006, vol. 27, pp. 5480–5489.
M.E. Launey, E. Munch, D.H. Alsem, H.B. Barth, E. Saiz, A.P. Tomsia, and R.O. Richie: Acta Mater., 2009, vol. 57, pp. 2919–2932.
S. Deville, E. Saiz, R.K. Nalla, and A.P. Tomsia: Science, 2006, vol. 311, pp. 515–518.
H.-Y. Lin, Y.-W. Chen, and C. Li: Thermochim. Acta, 2003, vol. 400, pp. 61–67.
W.K. Jozwiak, E. Kaczmarek, T.P. Maniecki, W. Ignaczak, and W. Maniukiewicz: Appl. Catal., A, 2007, vol. 326, pp. 17–27.
W.L. Li, K. Lu, J.Y. Walz: Int. Mater. Rev., 2012, vol. 57, pp. 37–60.
H. Park, M. Choi, H. Choe, and D.C. Dunand: Mater. Char., 2016, in review.
L.Y. Chen, J.Q. Xu, H. Choi, M. Pozuelo, X. Ma, S. Bhowmick, J.M. Yang, S. Mathaudhu, and X.C. Li: Nature, 2012, vol. 528, pp. 539–543.
N.O. Shanti, K. Araki, and J.W. Halloran: J. Am. Ceram. Soc., 2006, vol. 89, pp. 2444–2447.
H. Jo, M. Kim, H. Choi, Y. Sung, H. Choe, and D.C. Dunand: Metall. Mater. Trans. E, 2016, vol. 3, pp. 46–54.
Y.M. Lin, P.R. Abel, A. Heller, and C.B. Mullins: J. Phys. Chem. Lett., 2011, vol. 2, pp. 2885–2891.
J.H. Um, H. Park, Y.H. Cho, M. Glazer, D.C. Dunand, H. Choe, and Y.E. Sung: RSC Adv., 2014, vol. 4, pp. 58059–58063.
X. Xu, R. Cao, S. Jeong, and J. Cho: Nano Lett., 2012, vol. 12, pp. 4988–4991.
E.A. Brandes and G.B. Brook: Smithells metals reference book, 7th ed., Butterworth-Heinemann, USA, 1992.
H.J.T. Ellingham: J. Soc. Chem. Ind., 1944, vol. 63, pp. 125–160.
R.Y. Chen, and W.Y.D. Yuen: Oxid. Met., 2003, vol. 59, pp. 433–468.
N. Bertrandl, C. Desgranges, D. Gauvain, D. Monceau, and D. Poquillon: Mater. Sci. Forum., 2004, vol. 461–464, pp. 591–598.
G.B. Gibbs, M.R. Wootton, W.R. Price, and K.E. Hodgson: Oxid. Met., 1973, vol. 7, pp. 185–200.
Acknowledgments
This research was supported by the Priority Research Center Program through the National Research Foundation (NRF) of Korea (2012-0006680). HC also acknowledges the support from the Basic Science Research Program (2014R1A2A1A11052513).
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted September 15, 2015.
Rights and permissions
About this article
Cite this article
Park, H., Noh, Y., Choi, H. et al. Processing, Microstructure, and Oxidation Behavior of Iron Foams. Metall Mater Trans A 47, 4760–4766 (2016). https://doi.org/10.1007/s11661-016-3601-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11661-016-3601-9